📋 IB Content Statements (R1.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- R1.1.1: Chemical reactions involve a transfer of energy between the system and the surroundings. Reactions are described as exothermic or endothermic depending on the direction of the energy transfer.
- R1.1.2: The enthalpy change of a reaction ($\Delta H$) is the heat energy exchanged with the surroundings under constant pressure. This can be measured experimentally by calorimetry.
- R1.1.3: Standard enthalpy changes are measured under standard conditions (pressure of 100 kPa, temperature of 298 K, concentration of 1 mol dm⁻³).
- R1.1.4: The enthalpy of combustion ($\Delta H_c^\theta$) is the enthalpy change when one mole of a substance undergoes complete combustion under standard conditions.
Additional Syllabus Notes
- Interpretation and drawing of reaction coordinate (enthalpy profile) diagrams
- Calorimetry experiments: solution calorimetry and combustion calorimetry
- Sources of error in calorimetry and their effect on experimental values
- Use of the formula $q = mc\Delta T$ and $\Delta H = \frac{-q}{n}$
🔹 Energy Changes in Reactions
📺 R1.1.1 — Temperature vs Heat
Key Definition: Enthalpy ($H$)
Enthalpy is the total heat content of a system at constant pressure. We cannot measure $H$ directly, only the change in enthalpy ($\Delta H$). $\Delta H$ is the heat energy exchanged between the system and the surroundings at constant pressure.
$$\Delta H = H_{products} - H_{reactants}$$
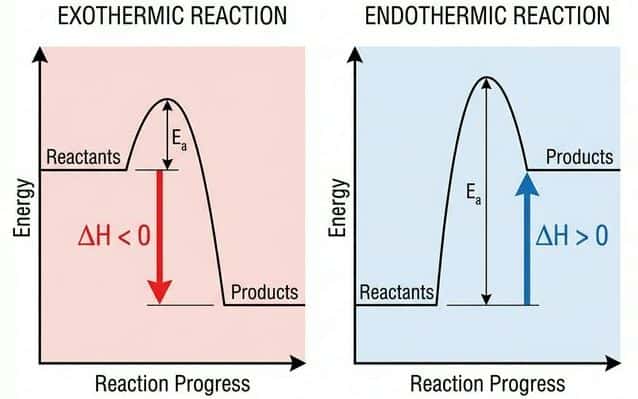
Exothermic vs Endothermic
🔥 Exothermic ($\Delta H < 0$)
Heat is released to the surroundings.
- Temperature of surroundings increases
- Products are more stable (lower energy) than reactants
- Energy released in forming new bonds > energy required to break old bonds
Examples: Combustion, neutralization, respiration, bond formation
❄️ Endothermic ($\Delta H > 0$)
Heat is absorbed from the surroundings.
- Temperature of surroundings decreases
- Products are less stable (higher energy) than reactants
- Energy required to break old bonds > energy released in forming new bonds
Examples: Thermal decomposition, photosynthesis, dissolving NH₄NO₃, melting ice
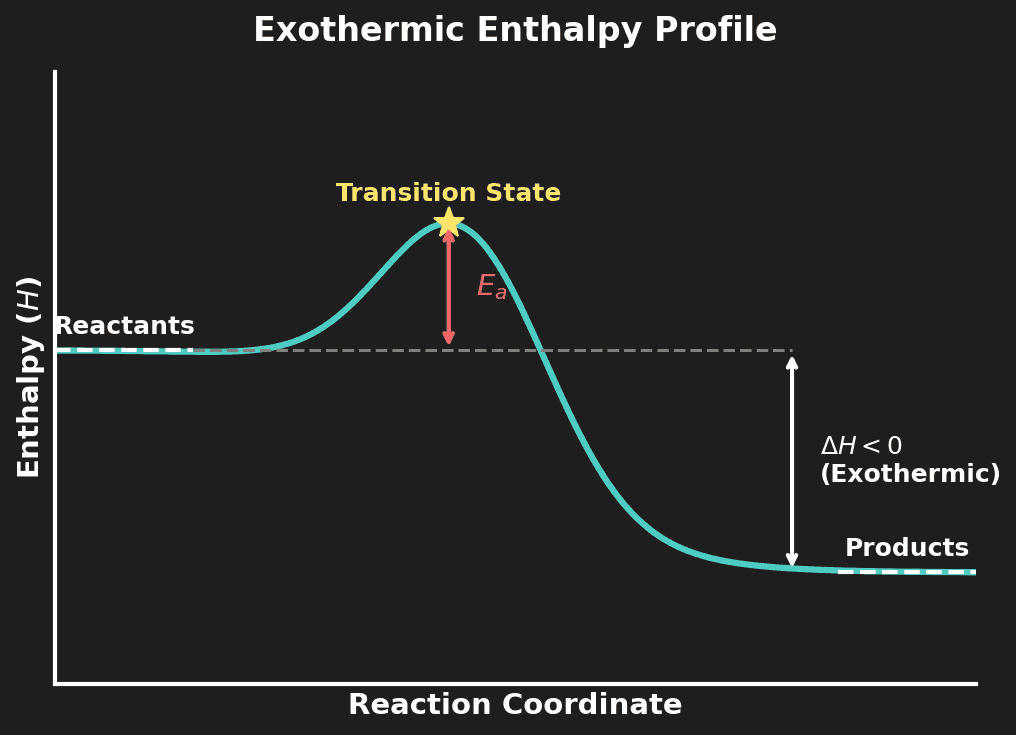
Enthalpy Profile Diagrams
Enthalpy profile (reaction coordinate) diagrams show the energy changes during a reaction:
- Y-axis: Enthalpy ($H$)
- X-axis: Reaction coordinate (progress of reaction)
- $\Delta H$: Difference between products and reactants energy levels
- Activation energy ($E_a$): The energy barrier that must be overcome for the reaction to proceed — shown as the height of the "hump" above the reactants
- Transition state: The peak of the energy profile — an unstable intermediate at maximum energy
📖 Standard Enthalpy Definitions
📺 R1.1 — Introduction to Bond Energies
These definitions are frequently tested in IB exams. Students must know them precisely — every word matters for marks.
| Term | Symbol | Definition |
|---|---|---|
| Standard Enthalpy of Combustion | $\Delta H_c^\theta$ | The enthalpy change when one mole of a substance is burned completely in excess oxygen under standard conditions |
| Standard Enthalpy of Formation | $\Delta H_f^\theta$ | The enthalpy change when one mole of a compound is formed from its elements in their standard states under standard conditions |
| Standard Enthalpy of Neutralization | $\Delta H_{neut}^\theta$ | The enthalpy change when an acid and base react to form one mole of water under standard conditions |
| Standard Enthalpy of Solution | $\Delta H_{sol}^\theta$ | The enthalpy change when one mole of a solute is dissolved in excess solvent to form an infinitely dilute solution under standard conditions |
⚠️ Exam Tip: The key phrase in every definition is "one mole". If you write "when a substance burns" without specifying "one mole", you will lose marks. Also, for combustion, always include "excess oxygen" and "complete" combustion.
🌡️ Calorimetry
Key Definition
Calorimetry is the experimental technique of measuring heat changes in chemical reactions by monitoring the temperature change of a known mass of water (or solution).
The Key Equations
Heat Energy Calculation
$$q = mc\Delta T$$
- $q$ = Heat energy transferred (J)
- $m$ = Mass of water or solution heated (g)
- $c$ = Specific heat capacity ($4.18 \ J \ g^{-1} \ K^{-1}$ for water)
- $\Delta T$ = Temperature change ($T_{final} - T_{initial}$) in K or °C
Enthalpy Change per Mole
$$\Delta H = \frac{-q}{n}$$
- $n$ = Moles of limiting reagent
- Negative sign = If surroundings gain heat (ΔT positive), the reaction lost heat → exothermic → ΔH is negative
Types of Calorimetry
| Feature | Solution Calorimetry (Coffee Cup) | Combustion Calorimetry (Spirit Burner) |
|---|---|---|
| What it measures | $\Delta H_{neut}$, $\Delta H_{sol}$, displacement | $\Delta H_c$ (enthalpy of combustion) |
| Setup | Polystyrene cup, lid, thermometer, stirrer | Spirit burner, metal can/beaker of water, thermometer |
| Mass (m) | Mass of solution in cup | Mass of water heated above the burner |
| Moles (n) | Moles of limiting reagent added | Moles of fuel burned (from mass loss of burner) |
| Key assumption | Density of solution = 1 g/cm³, c = 4.18 | All heat goes to water (major source of error) |
🔍 Sources of Error in Calorimetry
Experimental calorimetry values are always less exothermic (closer to zero) than literature values. Understanding why is critical for exam answers on evaluating experiments.
| Source of Error | Effect | Improvement |
|---|---|---|
| Heat loss to surroundings | $\Delta T$ measured is less than actual → $q$ too small | Better insulation (polystyrene), lid, draught shields |
| Incomplete combustion | Not all fuel reacts; less heat produced | Ensure excess oxygen supply; blue flame |
| Evaporation of fuel | Some fuel evaporates without burning, increasing apparent mass loss | Cap burner immediately after use; minimize time |
| Heat absorbed by apparatus | Some heat heats the container, not the water | Use a container with low heat capacity (polystyrene) |
| Non-standard conditions | Lab temperature ≠ 298 K exactly | Record and report actual conditions |
Graphical correction for heat loss: Plot temperature vs time. Extrapolate the cooling section back to the time of mixing to find the "true" maximum temperature. This technique compensates for heat lost during the experiment.
🧪 Interactive Virtual Labs
Experiment 1: Solution Calorimetry
AvailableCoffee Cup Calorimeter: Measure enthalpy changes in solution (e.g., Neutralization, Displacement). Calculate $\Delta H$ accounting for heat loss.
→ Launch Solution LabExperiment 2: Combustion Calorimetry
AvailableSpirit Burner: Determine the enthalpy of combustion of different alcohols. Observe the effect of chain length on energy release.
→ Launch Combustion Lab🧠 Memory Aids & Mnemonics
🔤 Exo vs Endo
"Exo = Exit" (energy exits the system) "Endo = Enter" (energy enters the system)
Exothermic: heat goes OUT → surroundings get warmer → $\Delta H$ is negative. Endothermic: heat comes IN → surroundings get cooler → $\Delta H$ is positive.
🔤 Sign Convention
"Hot Hand = Negative" → If you can feel the heat (exothermic), ΔH is negative
A reaction that warms up the solution has a negative $\Delta H$ (exothermic). A reaction that cools down the solution has a positive $\Delta H$ (endothermic). The sign tells you the system's perspective, not the surroundings'.
🔤 Definition Key Words
"1 mole + standard + specific reaction" = full marks
Every enthalpy definition starts with "the enthalpy change when ONE MOLE of..." and ends with "...under standard conditions." Missing either loses marks.
🔤 The q=mcΔT Formula
"Queen Mary Catches Dashing Thieves" → q = m × c × ΔT
Remember: $m$ is the mass of water (not the reagent), $c$ is always 4.18 for water, and $\Delta T$ must be positive (use absolute value if needed for $q$, then apply the sign in $\Delta H$).
🌍 Real-World Applications
🤲 Hand Warmers & Cold Packs
Context: Disposable hand warmers and instant cold packs are direct, everyday applications of enthalpy changes. They work without electricity — just chemistry.
Science: Hand warmers use the exothermic oxidation of iron powder: $4Fe(s) + 3O_2(g) \rightarrow 2Fe_2O_3(s)$, $\Delta H = -1648 \ kJ \ mol^{-1}$. Exposing the packet to air starts the reaction; salt and water act as catalysts. The reaction releases heat for 8-10 hours. Cold packs use the endothermic dissolution of ammonium nitrate: $NH_4NO_3(s) \rightarrow NH_4^+(aq) + NO_3^-(aq)$, $\Delta H_{sol} = +25.7 \ kJ \ mol^{-1}$. Breaking the inner bag mixes the salt with water — the dissolving absorbs heat from surroundings.
Impact: Athletes use cold packs for immediate first aid (not ice, which is slower). Hand warmers keep outdoor workers warm in extreme cold. Both illustrate that chemistry provides energy solutions without electricity.
🔢 Nutrition Labels: Bomb Calorimetry
Context: How do food companies know that a chocolate bar contains 230 kcal? They use a technique directly related to combustion calorimetry.
Science: A bomb calorimeter is a steel container surrounded by water. A weighed food sample is placed inside and ignited electrically in pure oxygen (ensuring complete combustion). The temperature rise of the surrounding water is measured, and $q = mc \Delta T$ gives the energy content. The Atwater system then assigns approximate values: 4 kcal/g for protein and carbohydrates, 9 kcal/g for fat, and 7 kcal/g for alcohol.
Impact: Every nutrition label worldwide is based on calorimetry. The accuracy of these values affects dietary planning, medical nutrition therapy for diabetes patients, and athletic performance optimization.
🔥 Combustion: Comparing Fuels
Context: Why do we use octane (petrol) for cars and not methanol? The answer is enthalpy of combustion per gram.
Science: Different fuels release different amounts of energy per mole and per gram:
| Fuel | $\Delta H_c^\theta$ (kJ/mol) | Energy per gram (kJ/g) |
|---|---|---|
| Hydrogen ($H_2$) | −286 | −143 |
| Methanol ($CH_3OH$) | −726 | −22.7 |
| Ethanol ($C_2H_5OH$) | −1367 | −29.7 |
| Octane ($C_8H_{18}$) | −5471 | −47.9 |
Impact: Hydrogen has the highest energy per gram (143 kJ/g) but is a gas at room temperature (storage is challenging). Octane has high energy density as a liquid, making it practical for vehicles. Biofuels (ethanol) have lower energy density but are renewable.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Getting the sign of ΔH wrong: If the temperature of the solution increases, the reaction is exothermic ($\Delta H$ is negative). Students often write positive because "the number went up." Remember: the negative sign comes from the $\frac{-q}{n}$ formula.
- Using the mass of the reagent instead of the mass of water: In $q = mc \Delta T$, $m$ is the mass of water (or solution) being heated, NOT the mass of the solid or fuel added.
- Forgetting to convert kJ to J or vice versa: $q$ from the formula gives Joules. Many students forget to divide by 1000 when reporting ΔH in kJ mol⁻¹.
- Incomplete definitions: Writing "enthalpy of combustion is when a substance burns" without specifying "one mole", "completely", "excess oxygen", and "standard conditions" loses marks.
- Not identifying the limiting reagent: The moles ($n$) in $\Delta H = \frac{-q}{n}$ must be the limiting reagent, not just any reagent. In a neutralization, calculate moles of both acid and base to find the limiting one.
- Ignoring heat loss reasoning in evaluations: When asked why the experimental value differs from the literature value, "heat loss" alone is not enough. State that heat is lost to surroundings, so $\Delta T$ is smaller than expected, making $q$ smaller and $\Delta H$ less exothermic.
📝 IB-Style Exam Questions
Question 1: Calculate the heat energy required to raise the temperature of 150g of water from 25.0°C to 45.0°C. [2 marks]
Mark Scheme:
- [1 mark] Use $q = mc\Delta T$: $q = 150 \times 4.18 \times (45.0 - 25.0)$
- [1 mark] $q = 150 \times 4.18 \times 20.0 = 12\,540 \ J$ or $12.5 \ kJ$
Question 2: In a combustion experiment, 0.50g of methanol ($M_r = 32.04$) raised the temperature of 100g water by 15.0°C. Calculate $\Delta H_c$. [3 marks]
Mark Scheme:
- [1 mark] Calculate $q$: $100 \times 4.18 \times 15.0 = 6270 \ J = 6.27 \ kJ$
- [1 mark] Calculate moles of methanol: $n = 0.50 / 32.04 = 0.0156 \ mol$
- [1 mark] Calculate $\Delta H$: $-6.27 / 0.0156 = -402 \ kJ \ mol^{-1}$ (sign must be negative)
Question 3: State one systematic error that causes the experimental value for enthalpy of combustion to be less exothermic than the literature value. [1 mark]
Mark Scheme:
- [1 mark] Heat loss to the surroundings / Incomplete combustion / Evaporation of fuel / Heat absorbed by apparatus.
Question 4: Distinguish between the terms 'exothermic' and 'endothermic'. [2 marks]
Mark Scheme:
- [1 mark] Exothermic: Heat is released to the surroundings ($\Delta H$ is negative).
- [1 mark] Endothermic: Heat is absorbed from the surroundings ($\Delta H$ is positive).
Question 5: Define the term 'Standard Enthalpy of Combustion'. [2 marks]
Mark Scheme:
- [1 mark] The enthalpy change when one mole of a substance is burned completely in excess oxygen...
- [1 mark] ...under standard conditions ($100 \ kPa, 298 \ K$).
Question 6: 50.0 cm³ of 1.00 mol dm⁻³ HCl is added to 50.0 cm³ of 1.00 mol dm⁻³ NaOH in a polystyrene cup. The temperature rises from 22.0°C to 28.8°C. Calculate the enthalpy of neutralization. [3 marks]
Mark Scheme:
- [1 mark] Total mass = 100 g (assuming density = 1 g/cm³). $q = 100 \times 4.18 \times 6.8 = 2842 \ J = 2.84 \ kJ$
- [1 mark] Moles of HCl (or NaOH) = $0.050 \times 1.00 = 0.050 \ mol$
- [1 mark] $\Delta H = \frac{-2.84}{0.050} = -56.8 \ kJ \ mol^{-1}$ (negative sign required)
Question 7: Explain why the experimental value for the enthalpy of combustion of ethanol is less exothermic than the literature value of −1367 kJ mol⁻¹. [2 marks]
Mark Scheme:
- [1 mark] Heat is lost to the surroundings (not all heat is transferred to the water), so the measured temperature rise is lower than the actual value.
- [1 mark] Incomplete combustion occurs, producing CO or C instead of CO₂, so less energy is released per mole.
Question 8: Sketch and label an enthalpy profile diagram for an exothermic reaction. Include: reactants, products, $\Delta H$, activation energy ($E_a$), and the transition state. [3 marks]
Mark Scheme:
- [1 mark] Reactants at higher energy level than products, with $\Delta H$ shown as a downward arrow between them (negative).
- [1 mark] A "hump" above the reactant level representing the transition state; $E_a$ labelled as the distance from reactants to the peak.
- [1 mark] Axes labelled: y-axis = Enthalpy ($H$), x-axis = Reaction coordinate (progress of reaction).