📋 IB Content Statements (R1.2)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- R1.2.1: Hess's law states that the enthalpy change for a reaction is independent of the pathway taken.
- R1.2.2: Enthalpy changes of reactions can be calculated from standard enthalpy changes of formation or combustion using Hess's law.
- R1.2.3: Bond enthalpy is the energy needed to break one mole of a particular bond in gaseous molecules. Average bond enthalpies are used because the same bond can have different energies in different molecules.
- R1.2.4: $\Delta H$ for a reaction can be estimated using average bond enthalpies: $\Delta H = \Sigma E(\text{bonds broken}) - \Sigma E(\text{bonds formed})$.
HL Extension
- R1.2.5 (HL): Born-Haber cycles can be used to calculate lattice enthalpies for ionic compounds.
- R1.2.6 (HL): Entropy ($S$) is a measure of the number of possible microstates. $\Delta S$ can be calculated from standard entropy values.
- R1.2.7 (HL): Gibbs free energy: $\Delta G = \Delta H - T\Delta S$. A reaction is spontaneous when $\Delta G < 0$.
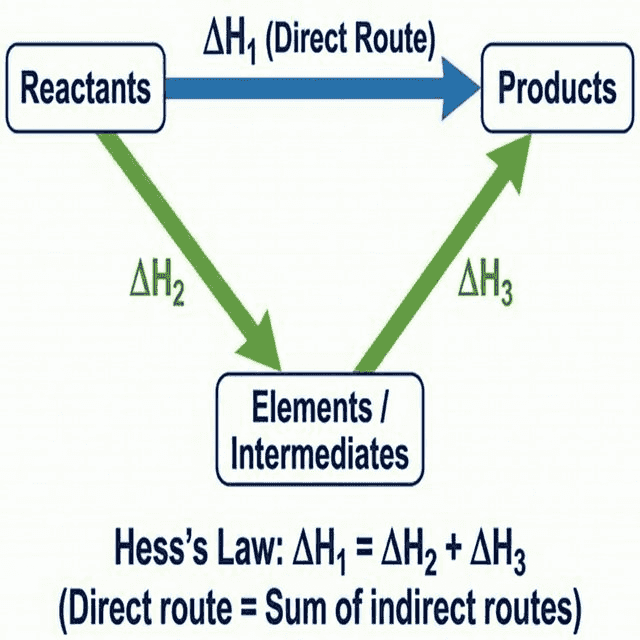
🔹 Hess's Law
Statement
The total enthalpy change for a chemical reaction is independent of the route taken, provided the initial and final conditions are the same.
This is a direct consequence of the law of conservation of energy.
Using Formation Data
$$\Delta H_{rxn} = \Sigma \Delta H_f^{\circ}(\text{products}) - \Sigma \Delta H_f^{\circ}(\text{reactants})$$
Key rule: The standard enthalpy of formation ($\Delta H_f^{\circ}$) of an element in its standard state is zero by definition. Example: $\Delta H_f^{\circ}[O_2(g)] = 0$, $\Delta H_f^{\circ}[C(graphite)] = 0$.
Using Combustion Data
$$\Delta H_{rxn} = \Sigma \Delta H_c^{\circ}(\text{reactants}) - \Sigma \Delta H_c^{\circ}(\text{products})$$
⚠️ Notice: For combustion data, it's reactants minus products (the reverse of formation). This is because the cycle goes "down then up" through combustion products as intermediates.
⚡ Bond Enthalpies
Definition
Bond enthalpy is the energy required to break one mole of a particular covalent bond in gaseous molecules under standard conditions. All bond enthalpy values are positive (endothermic).
Calculating ΔH from Bond Enthalpies
$$\Delta H = \Sigma E(\text{bonds broken}) - \Sigma E(\text{bonds formed})$$
| Bond | Average BE (kJ/mol) | Bond | Average BE (kJ/mol) |
|---|---|---|---|
| $C-H$ | 414 | $O=O$ | 498 |
| $C-C$ | 346 | $C=O$ (in $CO_2$) | 804 |
| $C=C$ | 614 | $O-H$ | 463 |
| $C-O$ | 358 | $N≡N$ | 945 |
⚠️ Why "average"? The same bond can have different strengths in different molecules. For example, $O-H$ in water (459 kJ/mol for the first, 428 for the second) differs from $O-H$ in ethanol. Therefore, bond enthalpy calculations give estimates, not exact values.
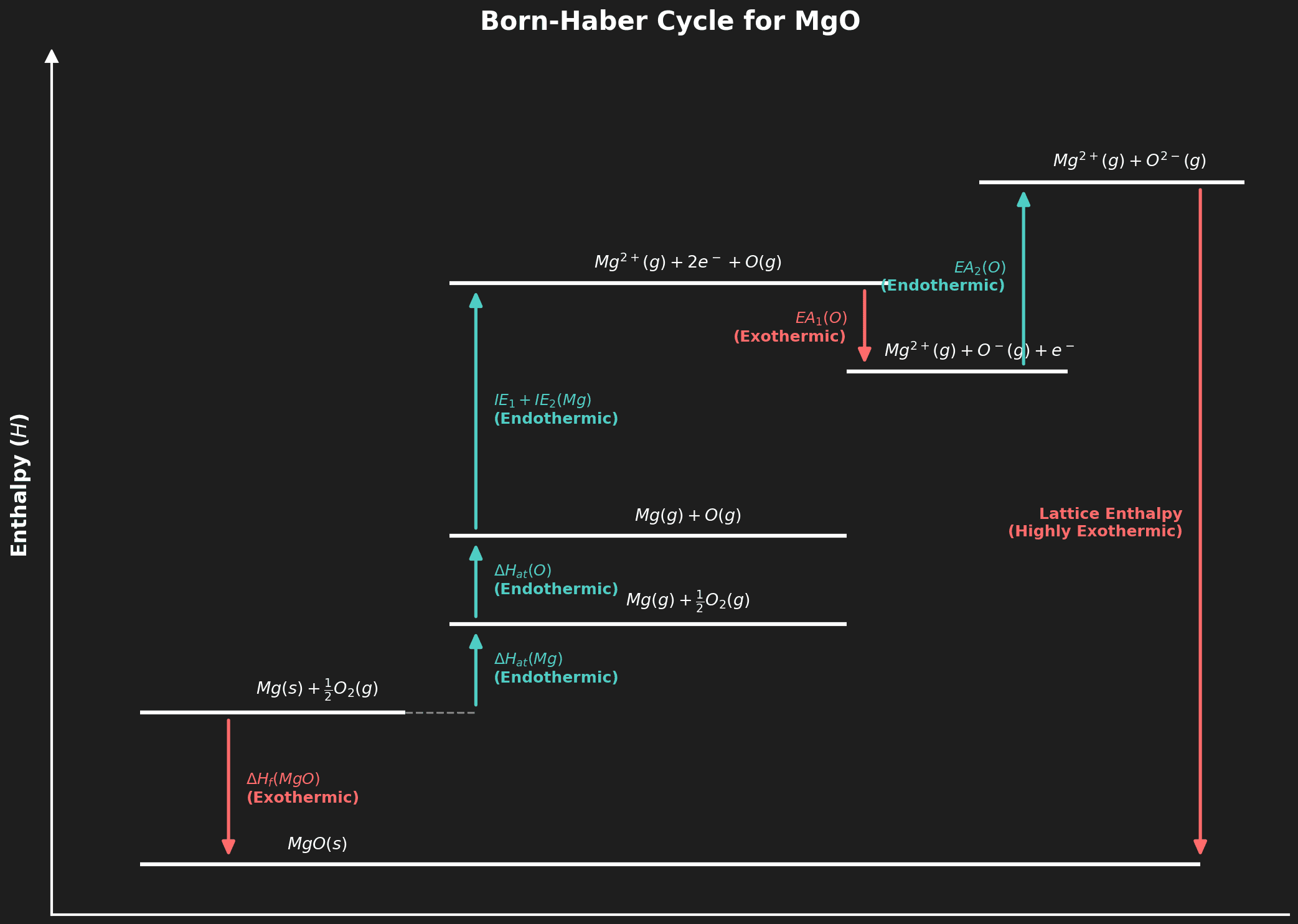
🏗️ Born-Haber Cycles (HL)
What is a Born-Haber Cycle?
A special application of Hess's law used to calculate the lattice enthalpy of an ionic compound — the enthalpy change when one mole of an ionic compound is formed from its gaseous ions under standard conditions.
Steps in a Born-Haber Cycle (for NaCl)
| Step | Process | Term | Sign |
|---|---|---|---|
| 1 | $Na(s) \rightarrow Na(g)$ | Atomization of Na | Endothermic (+) |
| 2 | $\frac{1}{2}Cl_2(g) \rightarrow Cl(g)$ | Atomization of Cl / ½ Bond dissociation | Endothermic (+) |
| 3 | $Na(g) \rightarrow Na^+(g) + e^-$ | 1st Ionization Energy | Endothermic (+) |
| 4 | $Cl(g) + e^- \rightarrow Cl^-(g)$ | 1st Electron Affinity | Exothermic (−) |
| 5 | $Na^+(g) + Cl^-(g) \rightarrow NaCl(s)$ | Lattice Enthalpy | Exothermic (−) |
$$\Delta H_f = \Delta H_{at}(Na) + \Delta H_{at}(Cl) + IE_1(Na) + EA_1(Cl) + \Delta H_{lat}$$
🌀 Entropy & Gibbs Free Energy (HL)
📺 R1.4 — Standard Entropy Change (HL)
📺 R1.4.1 — Predicting Entropy Changes (HL)
📺 R1.4.2 — How Temperature Changes Delta G (HL)
📺 R1.4.3 — Sign of Delta G (HL)
Entropy ($S$)
Entropy is a measure of the disorder (number of possible microstates) in a system. Higher entropy = more ways to arrange the particles.
$$\Delta S_{rxn} = \Sigma S^{\circ}(\text{products}) - \Sigma S^{\circ}(\text{reactants})$$
| Change | ΔS | Example |
|---|---|---|
| Solid → Liquid | Positive (+) | Ice melting |
| Liquid → Gas | Large Positive (++) | Water boiling |
| Fewer → More moles of gas | Positive (+) | $CaCO_3(s) → CaO(s) + CO_2(g)$ |
| More → Fewer moles of gas | Negative (−) | $N_2 + 3H_2 → 2NH_3$ |
Gibbs Free Energy
$$\Delta G = \Delta H - T\Delta S$$
| $\Delta H$ | $\Delta S$ | $\Delta G$ | Spontaneity |
|---|---|---|---|
| Negative (−) | Positive (+) | Always negative | Always spontaneous |
| Positive (+) | Negative (−) | Always positive | Never spontaneous |
| Negative (−) | Negative (−) | Depends on T | Spontaneous at low T |
| Positive (+) | Positive (+) | Depends on T | Spontaneous at high T |
🧠 Memory Aids
🔤 Bond Enthalpies — "Break = Buy, Form = Free"
Breaking bonds costs energy (endothermic, positive). Forming bonds releases energy (exothermic, negative). $\Delta H = \text{Break} - \text{Form}$.
🔤 Hess's Law Routes — "FPC" (Formation/Products first, Combustion/Products last)
- Formation: $\Delta H = \Sigma \Delta H_f(\text{Products}) - \Sigma \Delta H_f(\text{Reactants})$
- Combustion: $\Delta H = \Sigma \Delta H_c(\text{Reactants}) - \Sigma \Delta H_c(\text{Products})$
For Formation: Products first (P minus R). For Combustion: Reactants first (R minus P).
🔤 Gibbs — "GHiTS" ($\Delta G = \Delta H - T \Delta S$)
Gibbs = Heat minus Temperature times Scatter (entropy). Negative G = spontaneous (Go!).
🔤 Born-Haber Steps — "AAIIE + Lattice"
Atomize metal + Atomize non-metal + Ionize metal + Ionize non-metal (electron affinity) + Ends at lattice enthalpy.
🌍 Real-World Applications
🔋 Hand Warmers — Exothermic Crystallization
Context: Reusable hand warmers contain supersaturated sodium acetate. Clicking a metal disc triggers crystallization.
Science: The crystallization is exothermic ($\Delta H < 0$) and has negative $\Delta S$ (solid is more ordered). Using $\Delta G=\Delta H - T\Delta S$: both terms contribute to negative $\Delta G$ at room temperature → spontaneous.
Impact: Understanding Gibbs helps explain why some endothermic reactions can still occur spontaneously (at high T) when $T\Delta S$ overcomes $\Delta H$.
🧊 Instant Cold Packs — Entropy-Driven
Context: Instant cold packs dissolve ammonium nitrate ($NH_4NO_3$) in water. The pack feels cold.
Science: Dissolving is endothermic ($\Delta H > 0$), but the large increase in entropy ($\Delta S >> 0$) as the ordered crystal dissolves makes $T\Delta S > \Delta H$ → $\Delta G < 0$ → spontaneous despite being endothermic.
Impact: This is a classic example of an entropy-driven endothermic process. First-aid kits rely on this thermodynamics principle.
🏭 Haber Process — Gibbs Compromise
Context: $N_2 + 3H_2 \rightleftharpoons 2NH_3$ ($\Delta H = -92$ kJ/mol).
Science: The reaction is exothermic but $\Delta S < 0$ (4 mol gas → 2 mol gas). At low T, $\Delta G < 0$ (spontaneous) but the rate is too slow. At high T, $T\Delta S$ term dominates and $\Delta G$ becomes positive. The compromise: 450°C with an iron catalyst (kinetics) at 200 atm (Le Chatelier).
Impact: Produces 150 million tonnes of ammonia per year for fertilizers — feeding roughly half the world's population.
⚠️ Common Mistakes
- ❌ Swapping the formula for formation vs combustion → ✅ Formation: Products − Reactants. Combustion: Reactants − Products. Draw the cycle if unsure.
- ❌ "Breaking bonds releases energy" → ✅ Breaking bonds requires energy (endothermic). Only forming bonds releases energy.
- ❌ Forgetting to account for stoichiometric coefficients → ✅ Multiply bond/formation/combustion enthalpies by their coefficients in the balanced equation.
- ❌ "Spontaneous means fast" → ✅ Spontaneous ($\Delta G < 0$) means thermodynamically favorable — it says nothing about rate. Diamond → graphite is spontaneous but infinitely slow.
- ❌ Using °C instead of K for Gibbs → ✅ Temperature in $\Delta G = \Delta H - T\Delta S$ must be in Kelvin. Also, if $\Delta H$ is in kJ, convert $\Delta S$ from J/K to kJ/K.
📝 Exam-Style Questions
Question 1: State Hess's Law. [1 mark]
Mark Scheme:
- [1 mark] The enthalpy change for a reaction is independent of the pathway / route taken (provided initial and final conditions are the same).
Question 2: Using bond enthalpies, estimate $\Delta H$ for the combustion of methane: $CH_4 + 2O_2 → CO_2 + 2H_2O$. [3 marks]
Mark Scheme:
- [1 mark] Bonds broken: 4(C-H) + 2(O=O) = 4(414) + 2(498) = 2652 kJ.
- [1 mark] Bonds formed: 2(C=O in CO₂) + 4(O-H) = 2(804) + 4(463) = 3460 kJ.
- [1 mark] $\Delta H = 2652 - 3460 = -808$ kJ/mol.
Question 3: Explain why bond enthalpy calculations give only estimates. [2 marks]
Mark Scheme:
- [1 mark] Average bond enthalpies are used, not exact values for specific molecules.
- [1 mark] The same bond type has slightly different energies in different molecular environments.
Question 4 (HL): Predict the sign of $\Delta S$ for: $CaCO_3(s) \rightarrow CaO(s) + CO_2(g)$. [1 mark]
Mark Scheme:
- [1 mark] Positive. A gas is produced from a solid → increase in disorder / number of microstates.
Question 5 (HL): Calculate $\Delta G$ at 298 K if $\Delta H = -100$ kJ/mol and $\Delta S = +50$ J/(K·mol). [2 marks]
Mark Scheme:
- [1 mark] Convert: $\Delta S = 0.050$ kJ/(K·mol).
- [1 mark] $\Delta G = -100 - (298)(0.050) = -100 - 14.9 = -114.9$ kJ/mol. Spontaneous.
Question 6 (HL): Determine the temperature at which a reaction becomes spontaneous if $\Delta H = +50$ kJ/mol and $\Delta S = +100$ J/(K·mol). [2 marks]
Mark Scheme:
- [1 mark] At equilibrium: $\Delta G = 0$, so $T = \Delta H / \Delta S = 50000 / 100 = 500$ K (227°C).
- [1 mark] Spontaneous above 500 K.
Question 7: Calculate $\Delta H$ for the reaction $C_2H_4 + H_2 → C_2H_6$ using formation data: $\Delta H_f[C_2H_4] = +52$, $\Delta H_f[C_2H_6] = -85$ kJ/mol. [2 marks]
Mark Scheme:
- [1 mark] $\Delta H_f[H_2] = 0$ (element in standard state).
- [1 mark] $\Delta H = (-85) - (52 + 0) = -137$ kJ/mol.
Question 8 (HL): List the steps in a Born-Haber cycle for $MgO$ and state whether each is endothermic or exothermic. [3 marks]
Mark Scheme:
- [1 mark] Atomization of Mg and O (both endothermic).
- [1 mark] IE₁ + IE₂ of Mg (endothermic), EA₁ + EA₂ of O (EA₁ exothermic, EA₂ endothermic).
- [1 mark] Lattice enthalpy (exothermic — ions come together).