📋 IB Content Statements (R1.3)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- R1.3.1: Fossil fuels include coal, oil, and natural gas. Their combustion releases energy and produces $CO_2$ and $H_2O$.
- R1.3.2: Incomplete combustion produces $CO$ (toxic) and/or $C$ (soot).
- R1.3.3: Biofuels (e.g., ethanol, biodiesel) are renewable alternatives to fossil fuels. They can be carbon-neutral if managed sustainably.
- R1.3.4: Hydrogen can be used as a fuel. It produces only water on combustion, making it a clean fuel.
- R1.3.5: A hydrogen fuel cell converts chemical energy directly to electrical energy with high efficiency.
🔥 Combustion of Fuels
Complete vs Incomplete Combustion
Combustion reactions are exothermic ($\Delta H < 0$). The products depend on the availability of oxygen.
| Type | Oxygen Supply | Products | Issue |
|---|---|---|---|
| Complete | Excess $O_2$ | $CO_2 + H_2O$ | Maximum energy released; $CO_2$ greenhouse gas |
| Incomplete | Limited $O_2$ | $CO + H_2O$ or $C + H_2O$ | Less energy; $CO$ is toxic (binds to haemoglobin) |
Example: Combustion of Methane
Complete: $CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O$ ($\Delta H = -890$ kJ/mol)
Incomplete: $2CH_4 + 3O_2 \rightarrow 2CO + 4H_2O$
Specific Energy & Energy Density
| Fuel | Formula | $\Delta H_c$ (kJ/mol) | Specific Energy (kJ/g) |
|---|---|---|---|
| Hydrogen | $H_2$ | −286 | 143 (highest!) |
| Methane | $CH_4$ | −890 | 55.6 |
| Ethanol | $C_2H_5OH$ | −1367 | 29.7 |
| Octane | $C_8H_{18}$ | −5471 | 47.9 |
Specific energy = energy per gram (kJ/g). To calculate: $\text{Specific energy} = \frac{|\Delta H_c|}{M_r}$ where $M_r$ is the molar mass.
🛢️ Fossil Fuels & Environmental Impact
The Problem with Fossil Fuels
Fossil fuels are non-renewable (formed over millions of years from ancient organisms). Burning them releases stored carbon as $CO_2$, increasing atmospheric concentrations and driving climate change.
| Problem | Cause | Effect |
|---|---|---|
| Global warming | $CO_2$ and $CH_4$ trap IR radiation (greenhouse effect) | Rising temperatures, ice melt, sea level rise |
| Acid rain | $SO_2$ and $NO_x$ from S/N impurities in fuels | Damages buildings, kills aquatic life, destroys forests |
| Smog | $CO$, particulates, unburned hydrocarbons | Respiratory diseases, reduced visibility |
🌱 Biofuels & Renewable Fuels
| Biofuel | Source | Advantage | Limitation |
|---|---|---|---|
| Bioethanol | Fermentation of sugarcane / corn | Carbon neutral (in theory); renewable | Uses agricultural land; lower energy density than petrol |
| Biodiesel | Transesterification of vegetable oils | Can use waste cooking oil; renewable | Limited supply; can increase food prices |
| Biogas | Anaerobic digestion of organic waste | Uses waste products; produces $CH_4$ | Low energy density; needs large digesters |
Carbon Neutral?
Biofuels are described as "carbon neutral" because the $CO_2$ released during combustion was originally absorbed by the plant during photosynthesis. However, this ignores energy used in farming, transport, and processing — so in practice, biofuels are not truly carbon neutral.
⚡ Hydrogen Fuel Cells
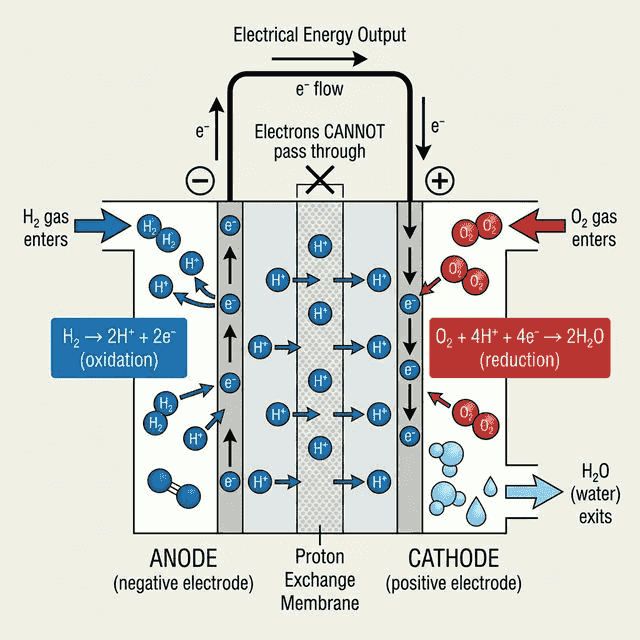
How a Hydrogen Fuel Cell Works
A fuel cell converts chemical energy directly to electrical energy, bypassing the heat engine step. This makes them much more efficient than combustion engines.
Overall reaction: $2H_2(g) + O_2(g) \rightarrow 2H_2O(l)$
| Electrode | Half-Reaction | Process |
|---|---|---|
| Anode (−) | $H_2 \rightarrow 2H^+ + 2e^-$ | Oxidation (hydrogen loses electrons) |
| Cathode (+) | $O_2 + 4H^+ + 4e^- \rightarrow 2H_2O$ | Reduction (oxygen gains electrons) |
Advantages & Disadvantages of Hydrogen Fuel Cells
| Advantages | Disadvantages |
|---|---|
| Only product is water → zero emissions at point of use | Hydrogen production often uses fossil fuels (steam reforming) |
| High efficiency (>60% vs ~25% for combustion engines) | Hydrogen is difficult to store (very low density, needs high pressure or cryogenic cooling) |
| Hydrogen has highest specific energy (143 kJ/g) | Infrastructure (refueling stations) is limited |
| Quiet operation, no moving parts | Platinum catalyst is expensive and rare |
⚙️ Energy Efficiency
Comparing Energy Sources
When evaluating fuels, we must consider:
- Specific energy (kJ/g) — energy per unit mass
- Energy density (kJ/L) — energy per unit volume
- Carbon footprint — total $CO_2$ produced per unit energy
- Availability & cost
- Conversion efficiency — what fraction of chemical energy becomes useful work
| Energy Source | Efficiency | $CO_2$ Emissions |
|---|---|---|
| Combustion engine (petrol) | ~25% | High |
| Coal power plant | ~33% | Very high |
| Hydrogen fuel cell | ~60% | Zero (at point of use) |
| Solar panel | ~20% | Zero (operational) |
🧠 Memory Aids
🔤 Complete vs Incomplete — "Full O₂ = Full Burn"
Full oxygen → $CO_2 + H_2O$ (good burn). Low oxygen → $CO + H_2O$ or $C + H_2O$ (bad burn, toxic/sooty).
🔤 CO Toxicity — "CO steals the SEAT"
Carbon monoxide binds to haemoglobin more strongly than oxygen does, "stealing" the binding site. The victim's blood cannot carry oxygen → suffocation even while breathing.
🔤 Fuel Cell — "Direct ConVersion = Higher effiCiency"
Fuel cells convert chemical → electrical directly, while combustion engines go chemical → heat → mechanical → electrical. Each conversion step loses energy, so fuel cells win on efficiency.
🔤 Carbon Neutral Myth — "Plant Absorbs = Burn Releases, BUT..."
The theory: $CO_2$ in (photosynthesis) = $CO_2$ out (combustion). The reality: farming, transport, and processing all use fossil fuels → not truly zero net $CO_2$.
🌍 Real-World Applications
🚗 Toyota Mirai — Hydrogen Fuel Cell Car
Context: The Toyota Mirai is one of the first mass-produced hydrogen fuel cell vehicles, available since 2014.
Science: It uses a PEM (proton exchange membrane) fuel cell stack to convert $H_2$ and $O_2$ into electricity. The only exhaust is water vapour. It stores hydrogen at 700 atm pressure and has a range of ~650 km.
Impact: Demonstrates that hydrogen vehicles are viable, but adoption is limited by high cost, lack of refueling infrastructure, and the fact that most hydrogen is currently produced from natural gas (steam reforming).
🌽 Brazil's Ethanol Programme
Context: Brazil produces bioethanol from sugarcane and mandates blending with petrol (up to E27 — 27% ethanol).
Science: Sugarcane is fermented to produce ethanol: $C_6H_{12}O_6 \rightarrow 2C_2H_5OH + 2CO_2$. The $CO_2$ released was recently absorbed by the sugarcane, making it approximately carbon neutral.
Impact: Brazil has reduced its dependence on oil imports, but concerns include deforestation for sugarcane plantations and competition with food production.
⚠️ Carbon Monoxide Poisoning — The Silent Killer
Context: CO detectors are required in most homes with gas appliances because incomplete combustion in boilers can produce lethal CO.
Science: CO is colourless and odourless. It binds to haemoglobin with a 200× higher affinity than $O_2$, forming carboxyhemoglobin ($HbCO$). Even low concentrations (100 ppm) cause headaches; high concentrations are fatal.
Impact: Regular servicing of gas appliances and CO detectors save thousands of lives each year. This is a direct consequence of incomplete combustion chemistry.
⚠️ Common Mistakes
- ❌ "Hydrogen fuel cells have zero emissions" → ✅ Zero emissions at point of use (only water produced). But hydrogen production (steam reforming of methane) currently produces $CO_2$. Must specify "at point of use" in exams.
- ❌ "Biofuels are carbon neutral" → ✅ Only approximately carbon neutral. Must acknowledge that energy is used in farming, processing, and transport of the biofuel.
- ❌ Confusing specific energy with energy density → ✅ Specific energy = kJ per gram (mass). Energy density = kJ per litre (volume). Hydrogen has the highest specific energy but very low energy density (gas).
- ❌ "CO₂ is toxic" → ✅ $CO_2$ is not toxic at normal levels — it's a greenhouse gas. $CO$ (carbon monoxide) is the toxic one — it binds to haemoglobin.
- ❌ "Fuel cells burn hydrogen" → ✅ Fuel cells involve electrochemical reactions (oxidation at anode, reduction at cathode), not combustion. There is no flame.
📝 Exam-Style Questions
Question 1: Write the equation for complete combustion of ethanol ($C_2H_5OH$). [1 mark]
Mark Scheme:
- [1 mark] $C_2H_5OH + 3O_2 \rightarrow 2CO_2 + 3H_2O$.
Question 2: State two differences between the products of complete and incomplete combustion. [2 marks]
Mark Scheme:
- [1 mark] Complete: produces $CO_2$. Incomplete: produces $CO$ or $C$ (soot).
- [1 mark] Complete combustion releases more energy per mole of fuel / $CO$ is toxic while $CO_2$ is not (at normal levels).
Question 3: Calculate the specific energy of methane ($\Delta H_c = -890$ kJ/mol, $M_r = 16$). [1 mark]
Mark Scheme:
- [1 mark] Specific energy = 890 / 16 = 55.6 kJ/g.
Question 4: Explain why biofuels are described as approximately carbon neutral. [2 marks]
Mark Scheme:
- [1 mark] The $CO_2$ released during combustion was absorbed by the plant during photosynthesis.
- [1 mark] However, energy from fossil fuels is used in farming, processing, and transporting the biofuel, so net $CO_2$ is not zero.
Question 5: Describe the half-reactions at each electrode in a hydrogen fuel cell. [2 marks]
Mark Scheme:
- [1 mark] Anode: $H_2 \rightarrow 2H^+ + 2e^-$ (oxidation).
- [1 mark] Cathode: $O_2 + 4H^+ + 4e^- \rightarrow 2H_2O$ (reduction).
Question 6: State two advantages and two disadvantages of hydrogen as a fuel. [2 marks]
Mark Scheme:
- [1 mark] Advantages: any two from — clean (only water produced), high specific energy, high efficiency in fuel cells.
- [1 mark] Disadvantages: any two from — difficult to store (low density), most H₂ made from fossil fuels, expensive infrastructure, platinum catalyst cost.
Question 7: Explain why fuel cells are more efficient than combustion engines. [2 marks]
Mark Scheme:
- [1 mark] Fuel cells convert chemical energy directly to electrical energy.
- [1 mark] Combustion engines convert chemical → heat → mechanical → electrical, with energy lost at each conversion step (limited by Carnot efficiency).
Question 8: Explain why carbon monoxide is dangerous despite being produced in small quantities. [2 marks]
Mark Scheme:
- [1 mark] CO is colourless and odourless, so it cannot be detected without a sensor.
- [1 mark] It binds irreversibly / very strongly to haemoglobin, preventing oxygen transport → can be fatal even at low concentrations.