📋 IB Content Statements (R2.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- R2.1.1: Species react as a result of collisions of sufficient energy and proper orientation.
- R2.1.2: Activation energy ($E_a$) is the minimum energy that colliding molecules need for a successful collision.
- R2.1.3: Factors that influence the rate of reaction include temperature, concentration (or pressure for gases), surface area, and the use of a catalyst.
- R2.1.4: The Maxwell-Boltzmann distribution curve shows the number of particles at different kinetic energies. It can be used to explain the effect of temperature changes and catalysts on rate.
- R2.1.5: A catalyst increases the rate of reaction by providing an alternative reaction pathway with a lower activation energy. It is not consumed in the reaction.
⏱️ Measuring Rates
Definition
The rate of reaction is the change in concentration of a reactant or product per unit time.
$$Rate = \frac{-\Delta [\text{Reactants}]}{\Delta t} = \frac{\Delta [\text{Products}]}{\Delta t}$$
Units: $mol \ dm^{-3} \ s^{-1}$
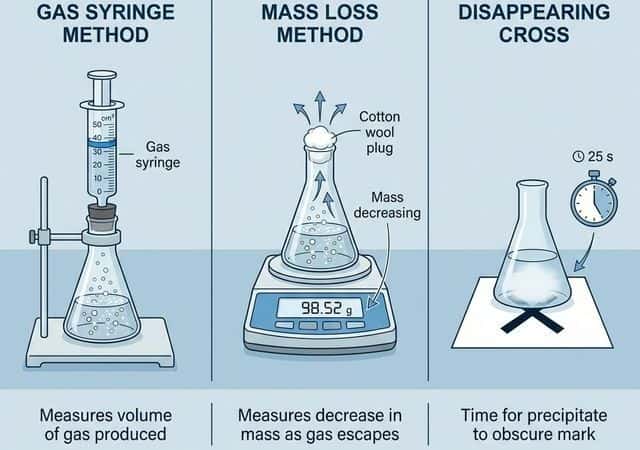
Methods of Measurement
| Method | What is Measured | Suitable When… | Example Reaction |
|---|---|---|---|
| Gas syringe | Volume of gas produced over time | A gas is a product | $Mg + 2HCl → MgCl_2 + H_2↑$ |
| Mass loss | Decrease in mass (gas escapes) | A gas is a product | $CaCO_3 + 2HCl → CaCl_2 + H_2O + CO_2↑$ |
| Disappearing cross | Time for precipitate to obscure a cross | A precipitate forms | $Na_2S_2O_3 + 2HCl → 2NaCl + H_2O + SO_2 + S↓$ |
| Colorimetry | Change in absorbance / light transmission | A coloured species is consumed or produced | $Br_2$ (brown) decolourized by alkenes |
| Conductivity | Change in electrical conductivity | Number or type of ions changes | Ester hydrolysis (produces ions) |
Reading rate from a graph: Plot [reactant] or [product] vs time. The gradient (slope) of the tangent at any point gives the instantaneous rate at that moment. The initial rate is measured from the tangent at $t = 0$.
💥 Collision Theory
Two Conditions for a Successful Collision
For a reaction to occur, particles must:
- Collide with sufficient energy ($E ≥ E_a$)
- Collide with the correct orientation (geometry)
A collision meeting both conditions is called an effective collision or successful collision.
Activation Energy ($E_a$)
The minimum energy that colliding particles must possess for a reaction to occur. It represents the energy barrier that must be overcome to break existing bonds and form new ones.
🎛️ Factors Affecting Rate
📺 R2.2.6 — Intermediates vs Transition States
| Factor | Change | Effect on Rate | Explanation (Collision Theory) |
|---|---|---|---|
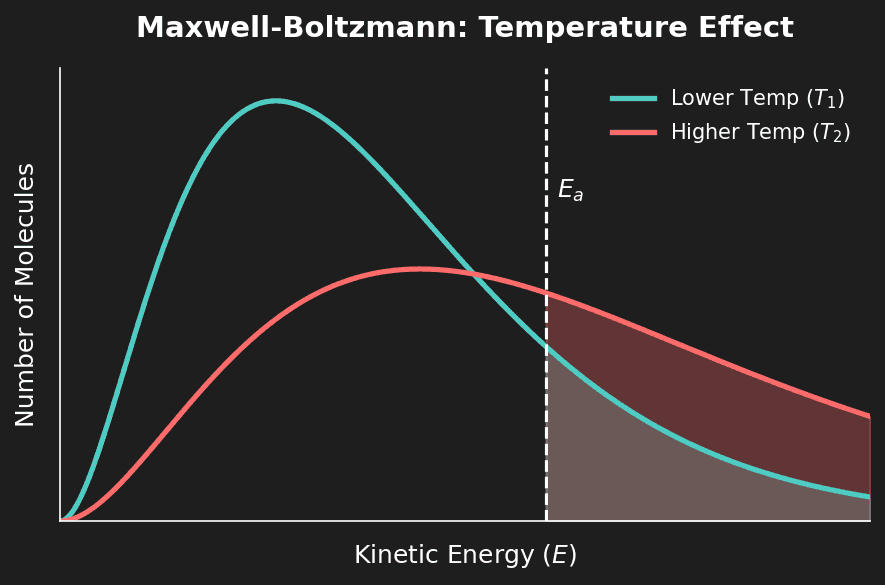
| Temperature | Increase ↑ | Rate increases ↑ | Main effect: Greater proportion of particles have $E ≥ E_a$. Minor effect: More frequent collisions. |
| Concentration | Increase ↑ | Rate increases ↑ | More particles per unit volume → more frequent collisions → more successful collisions per second. |
| Pressure (gases) | Increase ↑ | Rate increases ↑ | Same as concentration — particles are forced closer together, increasing collision frequency. |
| Surface area | Increase ↑ (smaller pieces) | Rate increases ↑ | More particles exposed at the surface → more collisions per second. |
| Catalyst | Add catalyst | Rate increases ↑ | Provides an alternative pathway with lower $E_a$. More particles now have sufficient energy. |
⚠️ Temperature is special: For temperature, the main reason rate increases is that a greater proportion of particles have energy exceeding $E_a$ (shown by the Maxwell-Boltzmann curve). The increase in collision frequency alone is a minor contribution. This distinction is heavily tested!
📈 Maxwell-Boltzmann Distribution
📺 R2.1 — The Boltzmann Distribution
What It Shows
The Maxwell-Boltzmann distribution shows the number of particles (y-axis) at each kinetic energy (x-axis) in a sample of gas. Key features:
- The curve starts at the origin (no particles have zero energy)
- It has a peak (most probable energy)
- It is asymptotic — the tail never touches the x-axis (a few particles always have very high energy)
- The area under the curve equals the total number of particles
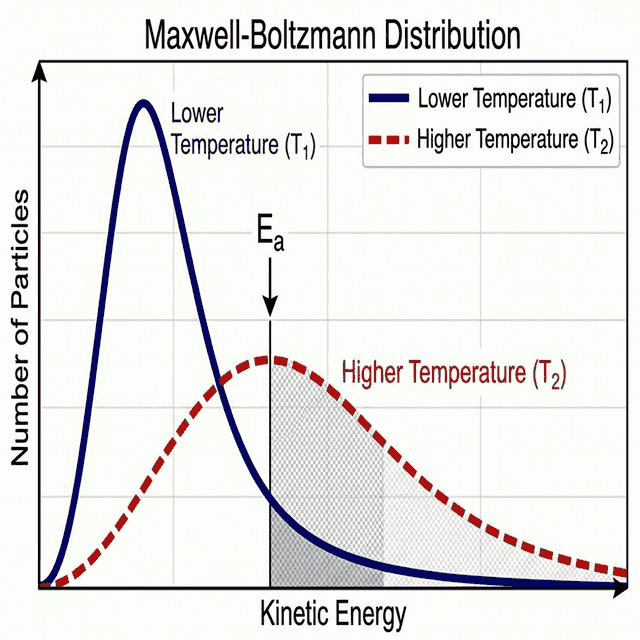
Effect of Temperature on the Distribution
| Feature | Lower Temperature | Higher Temperature |
|---|---|---|
| Peak height | Taller (more particles at peak energy) | Shorter and broader |
| Peak position | Further left (lower most probable energy) | Shifts right (higher most probable energy) |
| Area beyond $E_a$ | Smaller (fewer successful collisions) | Larger (more successful collisions) |
| Total area | Same (total number of particles unchanged) | |
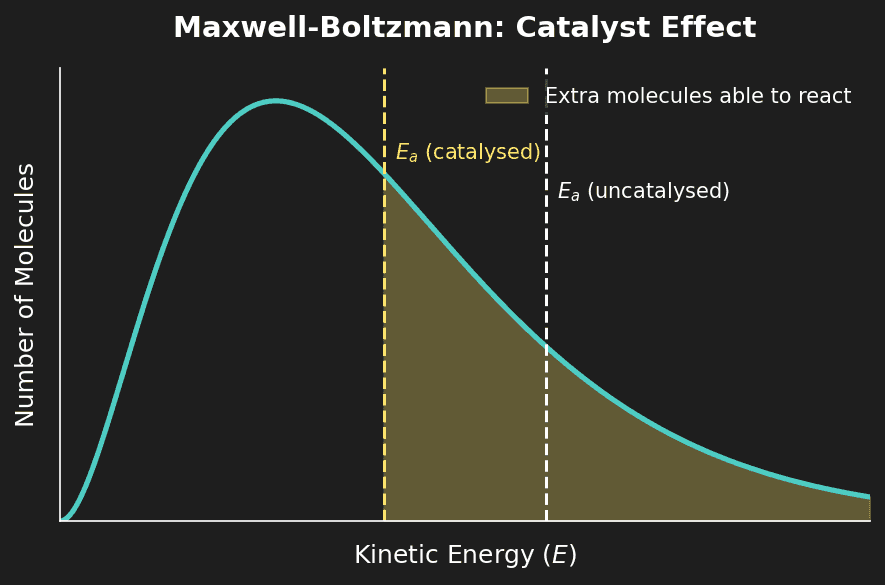
Effect of a Catalyst on the Distribution
A catalyst does not change the distribution curve. Instead, it lowers $E_a$, meaning the $E_a$ line moves to the left. This means a greater proportion of the same distribution now has enough energy to react.
🧪 Catalysts
Definition
A catalyst is a substance that increases the rate of reaction by providing an alternative reaction pathway with a lower activation energy. It is not consumed and is regenerated at the end of the reaction.
| Type | Description | Example |
|---|---|---|
| Homogeneous | Same phase as reactants | $H^+$ catalysing ester hydrolysis (all aqueous) |
| Heterogeneous | Different phase from reactants | Iron in the Haber process ($Fe(s)$ with gases) |
Important: A catalyst does not change $\Delta H$ for the reaction. It lowers $E_a$ for both the forward and reverse reactions equally, so it does not shift the position of equilibrium.
🧠 Memory Aids
🔤 Two Requirements — "Energy + Orientation = Success"
Think of a key in a lock. The key needs to be turned with enough force (energy ≥ $E_a$) AND inserted in the correct direction (orientation). Only then will it unlock (react).
🔤 Factors — "TCSC" (Temperature, Concentration, Surface area, Catalyst)
All four factors increase rate by increasing successful collisions. But remember: Temperature works mainly through energy (more particles ≥ $E_a$), while C, S, and Catalyst work through frequency or lower $E_a$.
🔤 Maxwell-Boltzmann — "SANA" (Starts At zero, Never touches Axis)
The curve starts at the origin (0,0) and the tail never touches the x-axis. Higher temperature: peak goes down and right, but the total area stays the same.
🔤 Temperature vs Catalyst — "Curve Change vs Line Shift"
Temperature changes the curve (flattens and shifts right). Catalyst doesn't change the curve — it shifts the $E_a$ line left. Both result in more particles beyond $E_a$, but by different mechanisms.
🌍 Real-World Applications
🚗 Catalytic Converters — Heterogeneous Catalysis
Context: Car exhausts contain catalytic converters with platinum, palladium, and rhodium catalysts on a honeycomb structure.
Science: The large surface area of the honeycomb maximizes contact between exhaust gases and the catalyst. The catalyst lowers $E_a$ for the conversion of toxic $CO$ and $NO_x$ into harmless $CO_2$ and $N_2$: $2CO + 2NO → 2CO_2 + N_2$.
Impact: Reduces air pollution from vehicles. This is why leaded petrol was banned — lead poisons the catalyst by blocking active sites.
🥛 Storing Milk — Temperature and Rate
Context: Milk is refrigerated at ~4°C to extend its shelf life.
Science: At lower temperatures, bacterial enzymes have less kinetic energy. Fewer enzyme-substrate collisions have $E ≥ E_a$, so the rate of decomposition decreases dramatically. This illustrates the Maxwell-Boltzmann effect of temperature on reaction rate.
Impact: Refrigeration extends the lifespan of perishable foods from hours to days/weeks — a direct application of kinetics.
💥 Dust Explosions — Surface Area and Rate
Context: Flour mills and coal mines can experience devastating dust explosions.
Science: Fine particles of combustible material (flour, coal dust) have an enormous surface area. When suspended in air and ignited, the reaction rate is so fast that it becomes an explosion. A block of coal burns slowly; the same mass as fine dust can explode.
Impact: Industrial safety regulations require dust control, ventilation, and spark prevention in flour mills and coal mines.
⚠️ Common Mistakes
- ❌ "Temperature increases rate because particles collide more often" → ✅ This is only a minor effect. The main reason is that a greater proportion of particles have $E ≥ E_a$. You must mention this for full marks.
- ❌ "A catalyst gives particles more energy" → ✅ A catalyst does not change particle energy. It provides an alternative pathway with lower $E_a$. The distribution curve stays the same.
- ❌ Drawing the MB curve touching the x-axis → ✅ The curve is asymptotic — it approaches but never touches the x-axis. There are always a few particles with very high energy.
- ❌ "Rate = speed of reaction" → ✅ Rate has specific units ($mol \ dm^{-3} \ s^{-1}$) and is defined as the change in concentration per unit time. Don't use vague language like "speed".
- ❌ Saying a catalyst "shifts equilibrium" → ✅ A catalyst increases the rate of both forward and reverse reactions equally. It does not change the position of equilibrium — only how quickly it is reached.
🧪 Interactive Virtual Labs
Experiment 1: Disappearing Cross
AvailableSimulate the reaction between Thiosulfate and Acid. Measure time for precipitate to obscure a cross.
→ Launch SimulationExperiment 2: Gas Collection
AvailableMeasure the volume of hydrogen gas produced over time from Mg + HCl.
→ Launch SimulationMaxwell-Boltzmann Distribution Simulator
AvailableVisualize how temperature affects particle energy distribution.
→ Launch Simulation📝 Exam-Style Questions
Question 1: Define the term "rate of reaction". [1 mark]
Mark Scheme:
- [1 mark] The change in concentration of a reactant (or product) per unit time.
Question 2: Explain, using collision theory, why increasing temperature increases the rate of reaction. [3 marks]
Mark Scheme:
- [1 mark] Particles gain kinetic energy / move faster.
- [1 mark] (Minor) Collision frequency increases.
- [1 mark] Crucially: A greater proportion of particles have energy ≥ $E_a$, so more collisions are successful.
Question 3: State two methods for measuring the rate of: $Mg(s) + 2HCl(aq) → MgCl_2(aq) + H_2(g)$. [2 marks]
Mark Scheme:
- [1 mark] Measure volume of $H_2$ gas over time (gas syringe).
- [1 mark] Measure decrease in mass over time (gas escapes).
Question 4: Sketch a Maxwell-Boltzmann distribution and label the axes. Show the effect of increasing temperature. [3 marks]
Mark Scheme:
- [1 mark] Y-axis: Number of particles. X-axis: Kinetic Energy. Curve starts at origin, asymptotic at high energy.
- [1 mark] Higher T curve: lower peak, shifted right.
- [1 mark] Total area under both curves is the same.
Question 5: Explain the effect of a catalyst on the rate of reaction. [2 marks]
Mark Scheme:
- [1 mark] Provides an alternative reaction pathway.
- [1 mark] With a lower activation energy ($E_a$), so a greater proportion of particles can react.
Question 6: Explain why powdered calcium carbonate reacts faster with hydrochloric acid than a lump of the same mass. [2 marks]
Mark Scheme:
- [1 mark] Powder has a larger surface area.
- [1 mark] More particles are exposed / available for collision with $HCl$ molecules → more frequent successful collisions.
Question 7: Using a Maxwell-Boltzmann diagram, explain why a catalyst increases the rate without changing the temperature. [2 marks]
Mark Scheme:
- [1 mark] The catalyst lowers $E_a$ (the $E_a$ line moves left on the diagram).
- [1 mark] The distribution curve does not change, but the area to the right of the new (lower) $E_a$ is greater → more particles can react.
Question 8: Distinguish between homogeneous and heterogeneous catalysts and give one example of each. [2 marks]
Mark Scheme:
- [1 mark] Homogeneous: same phase as reactants (e.g. $H^+$ in ester hydrolysis, both aqueous).
- [1 mark] Heterogeneous: different phase (e.g. $Fe(s)$ in the Haber process with gaseous reactants).