📋 IB Content Statements (R2.2)
This topic covers the following HL syllabus points from the IB Chemistry 2025 guide:
- R2.2.1 (HL): The rate expression relates the rate of reaction to the concentrations of reactants raised to their orders: $Rate = k[A]^m[B]^n$.
- R2.2.2 (HL): Orders of reaction ($m$, $n$) can only be determined experimentally (initial rates method). They cannot be deduced from stoichiometry.
- R2.2.3 (HL): The rate constant ($k$) is temperature-dependent. Its units depend on the overall order of the reaction.
- R2.2.4 (HL): Graphs of concentration vs time and rate vs concentration can be used to determine order.
- R2.2.5 (HL): The rate-determining step (RDS) is the slowest step in a multi-step mechanism. The rate expression reflects the species involved in or before the RDS.
- R2.2.6 (HL): The Arrhenius equation: $k = Ae^{-E_a/RT}$ and its linear form $\ln k = -\frac{E_a}{R}\left(\frac{1}{T}\right) + \ln A$.
📐 The Rate Expression
The Rate Law
$$Rate = k[A]^m[B]^n$$
- $k$ = Rate constant (depends on temperature and catalyst)
- $m$, $n$ = Orders of reaction with respect to A and B
- Overall order = $m + n$
⚠️ Critical Rule: Orders of reaction are determined experimentally only (usually by the initial rates method). They cannot be deduced from the balanced equation's stoichiometric coefficients.
Units of the Rate Constant ($k$)
| Overall Order | Rate Expression | Units of $k$ |
|---|---|---|
| 0 | $Rate = k$ | $mol \ dm^{-3} \ s^{-1}$ |
| 1 | $Rate = k[A]$ | $s^{-1}$ |
| 2 | $Rate = k[A]^2$ or $k[A][B]$ | $mol^{-1} \ dm^{3} \ s^{-1}$ |
| 3 | $Rate = k[A]^2[B]$ | $mol^{-2} \ dm^{6} \ s^{-1}$ |
Deriving units: Rearrange $k = \frac{Rate}{[A]^m[B]^n}$ and substitute units: $\frac{mol \ dm^{-3} \ s^{-1}}{(mol \ dm^{-3})^{m+n}}$.
🔢 Orders of Reaction
| Order | Meaning | Effect on Rate | [A] vs Time Graph | Rate vs [A] Graph |
|---|---|---|---|---|
| Zero ($[A]^0$) | Rate independent of [A] | Changing [A] has no effect | Straight line (linear decrease) | Horizontal line |
| First ($[A]^1$) | Rate ∝ [A] | Double [A] → Double rate | Exponential decay (curve) | Straight line through origin |
| Second ($[A]^2$) | Rate ∝ $[A]^2$ | Double [A] → Quadruple rate | Steep curve (faster initial drop) | Parabolic (upward curve) |
Determining Orders from Experimental Data
Initial rates method: Compare two experiments where only one reactant concentration changes:
$$\frac{Rate_2}{Rate_1} = \left(\frac{[A]_2}{[A]_1}\right)^m$$
If [A] doubles and rate doubles → $m = 1$. If [A] doubles and rate quadruples → $m = 2$. If [A] doubles and rate stays the same → $m = 0$.
Worked Example
| Experiment | [A] (mol/dm³) | [B] (mol/dm³) | Initial Rate (mol/dm³/s) |
|---|---|---|---|
| 1 | 0.10 | 0.10 | $2.0 \times 10^{-3}$ |
| 2 | 0.20 | 0.10 | $8.0 \times 10^{-3}$ |
| 3 | 0.10 | 0.20 | $4.0 \times 10^{-3}$ |
Analysis: Exp 1→2: [A] × 2, rate × 4 → order w.r.t. A = 2. Exp 1→3: [B] × 2, rate × 2 → order w.r.t. B = 1. Rate = $k[A]^2[B]$. Overall order = 3.
📈 Graphical Analysis
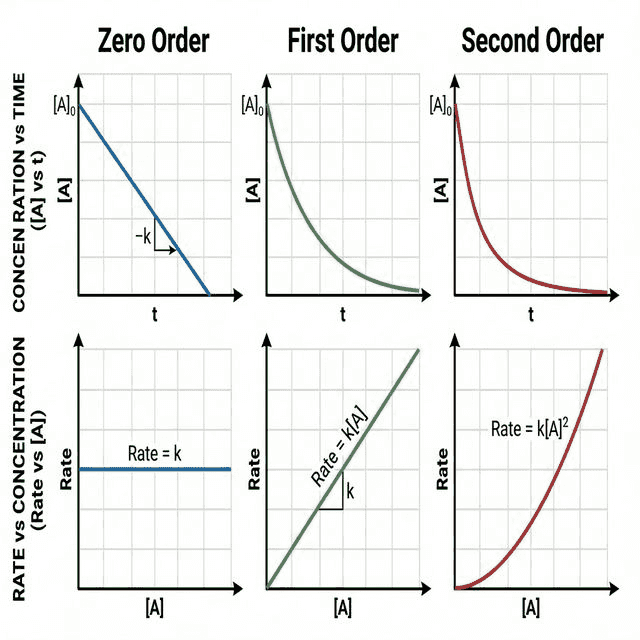
Half-Life and Order
The half-life ($t_{1/2}$) is the time for the concentration of a reactant to halve.
- First order: Half-life is constant (independent of concentration). $t_{1/2} = \frac{\ln 2}{k} = \frac{0.693}{k}$
- Second order: Half-life increases as concentration decreases.
- Zero order: Half-life decreases as concentration decreases.
⚙️ Reaction Mechanisms
Rate-Determining Step (RDS)
Most reactions occur through a series of elementary steps, not in a single step. The slowest step is the rate-determining step — it acts like a bottleneck and controls the overall rate.
Key rule: The rate expression only includes species that appear in or before the rate-determining step. This is how we use the rate expression to deduce the mechanism.
Molecularity
| Term | Number of Species in RDS | Example |
|---|---|---|
| Unimolecular | 1 molecule | $A → products$ (decomposition) |
| Bimolecular | 2 molecules collide | $A + B → products$ |
⚠️ Termolecular steps (3 molecules colliding simultaneously) are extremely rare because the probability of three particles colliding at the same instant with the correct orientation and energy is negligibly small.
Iodine Clock Reaction
AvailableSimulation of the Iodine Clock Reaction (Initial Rates Method).
→ Launch Simulation🔥 Arrhenius Equation
The Arrhenius Equation
$$k = Ae^{-E_a/RT}$$
- $k$ = rate constant
- $A$ = Arrhenius constant / frequency factor (related to collision frequency and orientation)
- $E_a$ = activation energy (J/mol)
- $R$ = gas constant (8.314 J K⁻¹ mol⁻¹)
- $T$ = temperature in Kelvin
Linear Form (for Graphical Determination)
$$\ln k = -\frac{E_a}{R}\left(\frac{1}{T}\right) + \ln A$$
| $y = mx + c$ Component | Corresponds to |
|---|---|
| $y$ | $\ln k$ |
| $x$ | $\frac{1}{T}$ (in K⁻¹) |
| Gradient ($m$) | $-\frac{E_a}{R}$ → so $E_a = -\text{gradient} \times R$ |
| y-intercept ($c$) | $\ln A$ |
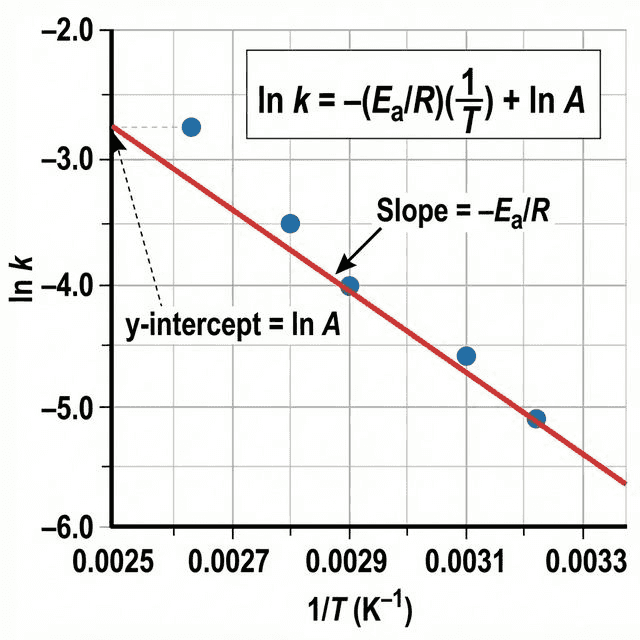
Arrhenius Plot Simulator
AvailablePlot experimental data to determine Activation Energy ($E_a$).
→ Launch Simulation🧠 Memory Aids
🔤 Orders — "Same, Double, Quadruple" (0, 1, 2)
When you double the concentration:
- Order 0: Rate stays the same ($2^0 = 1$)
- Order 1: Rate doubles ($2^1 = 2$)
- Order 2: Rate quadruples ($2^2 = 4$)
🔤 Rate vs [A] Graphs — "Flat, Line, Parabola" (0, 1, 2)
Zero: Horizontal line (rate doesn't depend on [A]). First: Straight line through origin. Second: Parabolic curve (rate ∝ $[A]^2$).
🔤 RDS — "Slowest Step Sets the Speed"
Think of a motorway traffic jam. The overall journey time is determined by the slowest section (bottleneck), not the fastest. Only the species involved in the bottleneck (RDS) appear in the rate expression.
🔤 Arrhenius Linear Form — "y = mx + c"
$\ln k$ vs $\frac{1}{T}$: Gradient = $-\frac{E_a}{R}$ (steep negative = high $E_a$). y-intercept = $\ln A$. Remember: $E_a = -\text{gradient} \times 8.314$.
🌍 Real-World Applications
⏰ Iodine Clock Reaction — Determining Order
Context: The iodine clock is a classic experiment used to determine the order of reaction by the initial rates method.
Science: Two solutions are mixed. After a fixed time delay, the solution suddenly turns dark blue (starch-iodine complex). By varying the concentration of one reactant and measuring the time delay, students can determine the order using $Rate \propto \frac{1}{t}$.
Impact: This experiment is the most common practical method for teaching the initial rates method at HL level.
🍞 Food Expiry Dates — Arrhenius in Action
Context: Food scientists use the Arrhenius equation to predict shelf life at different storage temperatures.
Science: By measuring spoilage rate ($k$) at several elevated temperatures and plotting $\ln k$ vs $\frac{1}{T}$, they extrapolate the rate at refrigerator temperature (4°C). The $E_a$ of the degradation reaction determines how sensitive the food is to temperature changes.
Impact: This accelerated testing saves months of real-time shelf-life studies with an accuracy of ±10%.
💊 Pharmaceutical Stability — Drug Degradation Kinetics
Context: Drug manufacturers must determine how long a medicine remains effective before it degrades.
Science: Most drug degradation follows first-order kinetics (constant half-life). If a drug's half-life is 2 years, after 2 years 50% remains active, after 4 years 25%, etc. The Arrhenius equation predicts how storage temperature affects this rate.
Impact: Expiry dates on medicines are calculated using these kinetic models — ensuring patient safety globally.
⚠️ Common Mistakes
- ❌ Deducing orders from the balanced equation → ✅ Orders are determined experimentally only. The coefficients in the balanced equation are not the orders (unless the reaction truly occurs in one step).
- ❌ Forgetting to derive units for $k$ → ✅ Always derive units by rearranging $k = \frac{Rate}{[A]^m[B]^n}$. Different overall orders give different units.
- ❌ "The RDS is always the first step" → ✅ The RDS is the slowest step — it can be any step in the mechanism. The rate expression tells us which step is rate-determining.
- ❌ Using °C in the Arrhenius equation → ✅ Temperature must be in Kelvin. Also, $E_a$ must be in J/mol (not kJ/mol) when $R = 8.314$ J K⁻¹ mol⁻¹.
- ❌ "Constant half-life means zero order" → ✅ Constant half-life is characteristic of first order, not zero order. Zero order has a half-life that decreases over time.
📝 Exam-Style Questions
Question 1: The rate expression is $Rate = k[A][B]^2$. State the effect on rate if [B] is tripled. [1 mark]
Mark Scheme:
- [1 mark] Second order w.r.t. B: $3^2 = 9$. Rate increases by a factor of 9.
Question 2: Define "rate-determining step". [1 mark]
Mark Scheme:
- [1 mark] The slowest step in a multi-step reaction mechanism.
Question 3: A Rate vs [A] graph gives a straight line through the origin. What is the order? [1 mark]
Mark Scheme:
- [1 mark] First order ($Rate \propto [A]$).
Question 4: Determine the units of $k$ for a reaction that is second order overall. [1 mark]
Mark Scheme:
- [1 mark] $k = Rate / [A]^2 = (mol \ dm^{-3} \ s^{-1}) / (mol \ dm^{-3})^2$ = $mol^{-1} \ dm^{3} \ s^{-1}$.
Question 5: Write the Arrhenius equation in linear form and state what the gradient represents. [2 marks]
Mark Scheme:
- [1 mark] $\ln k = -\frac{E_a}{R}\left(\frac{1}{T}\right) + \ln A$.
- [1 mark] Gradient = $-\frac{E_a}{R}$.
Question 6: Using the data table below, determine the orders with respect to X and Y, and write the rate expression. [3 marks]
| Exp | [X] | [Y] | Rate |
|---|---|---|---|
| 1 | 0.1 | 0.1 | $3.0 \times 10^{-4}$ |
| 2 | 0.2 | 0.1 | $3.0 \times 10^{-4}$ |
| 3 | 0.1 | 0.3 | $2.7 \times 10^{-3}$ |
Mark Scheme:
- [1 mark] Exp 1→2: [X] doubles, rate unchanged → order = 0 w.r.t. X.
- [1 mark] Exp 1→3: [Y] triples, rate × 9 ($3^2$) → order = 2 w.r.t. Y.
- [1 mark] Rate = $k[Y]^2$.
Question 7: Explain how a constant half-life can be used to confirm a first-order reaction. [2 marks]
Mark Scheme:
- [1 mark] Plot [A] vs time and measure successive half-lives.
- [1 mark] If $t_{1/2}$ is constant regardless of [A], the reaction is first order. (For other orders, $t_{1/2}$ varies with [A].)
Question 8: The Arrhenius plot for a reaction has a gradient of $-5200$ K. Calculate $E_a$. [2 marks]
Mark Scheme:
- [1 mark] $E_a = -\text{gradient} \times R = -(-5200) \times 8.314$.
- [1 mark] $E_a = 43233$ J/mol = 43.2 kJ/mol.