📋 IB Content Statements (R3.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- R3.1.1: A state of equilibrium is reached in a closed system when the rates of forward and reverse reactions are equal (dynamic equilibrium). Concentrations remain constant but are not necessarily equal.
- R3.1.2: The position of equilibrium is affected by changes in concentration, pressure (for gases), and temperature.
- R3.1.3: Le Chatelier's Principle: if a system at equilibrium is subjected to a change, the position shifts to partially oppose that change.
- R3.1.4: A catalyst speeds up both forward and reverse reactions equally and does not change the position of equilibrium.
- R3.1.5: Temperature is the only factor that changes the value of the equilibrium constant ($K$).
⚖️ Dynamic Equilibrium
Definition
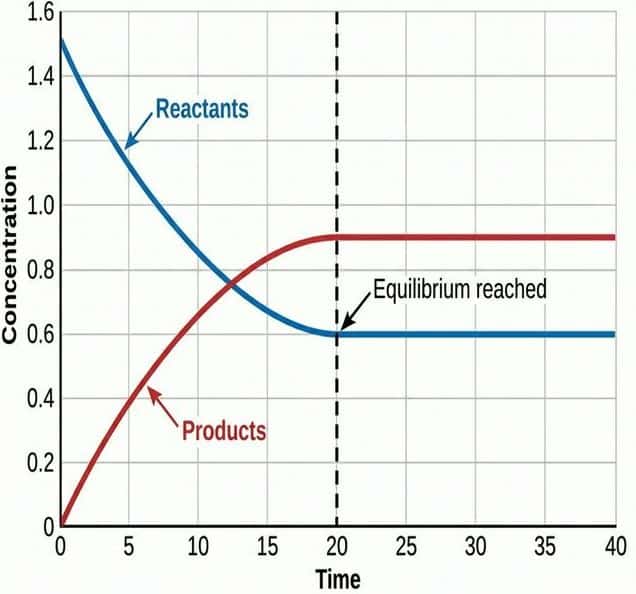
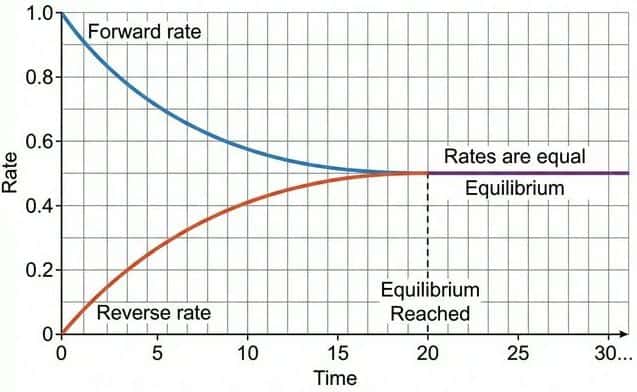
A dynamic equilibrium exists in a closed system when:
- The rate of the forward reaction equals the rate of the reverse reaction.
- The concentrations of reactants and products remain constant (but not necessarily equal).
$$Reactants \rightleftharpoons Products$$
Requirements for Equilibrium
- Closed system: No matter can enter or leave.
- Reversible reaction: The reaction must be able to proceed in both directions.
- "Dynamic": Both forward and reverse reactions are still occurring — the system is not static.
🔄 Le Chatelier's Principle
Statement
"If a system at equilibrium is subjected to a change in conditions, the position of equilibrium will shift to partially oppose that change."
| Change Applied | System Response | Shift Direction | Effect on $K$ |
|---|---|---|---|
| Increase [reactant] | Use up excess reactant | → Right (toward products) | No change |
| Increase [product] | Use up excess product | ← Left (toward reactants) | No change |
| Increase pressure (gases) | Reduce pressure | Towards side with fewer gas moles | No change |
| Increase temperature | Absorb excess heat | Towards endothermic direction | Changes $K$ |
| Decrease temperature | Release heat | Towards exothermic direction | Changes $K$ |
| Add catalyst | Both rates increase equally | NO SHIFT (equilibrium reached faster) | No change |
| Add inert gas (at constant V) | Total pressure ↑ but partial pressures unchanged | NO SHIFT | No change |
⚠️ Temperature is the ONLY factor that changes $K$. Changes in concentration, pressure, or adding a catalyst change the position of equilibrium (or not, in the case of catalysts) but never change the value of $K$.
Pressure — When Does It Apply?
Pressure changes only affect equilibria involving gases where the number of moles of gas differs on each side. If the moles are equal (e.g. $H_2 + I_2 \rightleftharpoons 2HI$, 2 moles each side), a change in pressure has no effect on position.
🏭 Industrial Applications
The Haber Process
$N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)$ $\Delta H = -92$ kJ/mol
| Condition | Value Used | Le Chatelier Reasoning |
|---|---|---|
| Pressure | 200 atm (high) | 4 mol gas → 2 mol gas. High pressure favors fewer moles (right). |
| Temperature | 450°C (moderate) | Compromise: Low T favors yield (exothermic → right), but rate is too slow. 450°C balances yield vs rate. |
| Catalyst | Iron (Fe) | Does not change yield but increases rate of reaching equilibrium. Economically essential. |
The Contact Process
$2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g)$ $\Delta H = -198$ kJ/mol
| Condition | Value Used | Le Chatelier Reasoning |
|---|---|---|
| Pressure | 1–2 atm | 3 mol → 2 mol (high P would favor right), but yield is already ~99% so high P is not economical. |
| Temperature | 450°C | Compromise: low T gives high yield but slow rate. |
| Catalyst | Vanadium(V) oxide ($V_2O_5$) | Speeds up reaction without changing yield. |
🧠 Memory Aids
🔤 Dynamic Equilibrium — "CRC" (Closed, Rates equal, Concentrations constant)
Three conditions to state: Closed system, Rate forward = Rate reverse, Concentrations constant. You need all three for full marks.
🔤 Temperature Direction — "Endo Absorbs"
Increase temperature → system shifts toward the endothermic direction (it absorbs the extra heat). Decrease temperature → exothermic direction (it releases heat to compensate).
🔤 Pressure Direction — "Fewer Molecules Win"
Increase pressure → equilibrium shifts to the side with fewer moles of gas. Count ONLY gas moles — solids and liquids don't count.
🔤 Catalyst Effect — "Faster, Not Further"
A catalyst makes the system reach equilibrium faster but doesn't shift it further. It increases both forward and reverse rates equally. $K$ doesn't change.
🌍 Real-World Applications
🩸 Hemoglobin and Oxygen — Le Chatelier in Your Blood
Context: $Hb + 4O_2 \rightleftharpoons Hb(O_2)_4$
Science: In the lungs, oxygen concentration is high → equilibrium shifts right (hemoglobin picks up $O_2$). In body tissues, oxygen concentration is low → equilibrium shifts left (hemoglobin releases $O_2$). Carbon monoxide poisoning occurs because CO binds more tightly than $O_2$, shifting the equilibrium irreversibly.
Impact: This is why oxygen masks are used at high altitudes — to increase $[O_2]$ and shift the equilibrium right.
🥤 Carbonated Drinks — Pressure and Equilibrium
Context: $CO_2(g) \rightleftharpoons CO_2(aq)$
Science: Carbonated drinks are bottled under high pressure (3–4 atm). When you open the bottle, pressure decreases → equilibrium shifts to the left (gaseous side) → $CO_2$ comes out of solution as bubbles. This is why fizzy drinks go flat when left open.
Impact: Keeping the lid on maintains high pressure and preserves carbonation — a direct application of Le Chatelier's principle to pressure changes.
🌊 Ocean Acidification — Temperature + Concentration
Context: $CO_2(g) + H_2O(l) \rightleftharpoons H_2CO_3(aq) \rightleftharpoons H^+(aq) + HCO_3^-(aq)$
Science: As atmospheric $CO_2$ increases (more reactant), the equilibrium shifts right → more $H^+$ → lower pH (ocean becomes more acidic). This dissolves calcium carbonate shells of marine organisms: $CaCO_3(s) + 2H^+(aq) → Ca^{2+}(aq) + H_2O(l) + CO_2(g)$.
Impact: Ocean pH has dropped from 8.2 to 8.1 since pre-industrial times — affecting coral reefs, shellfish, and marine ecosystems globally.
⚠️ Common Mistakes
- ❌ "Equal concentrations at equilibrium" → ✅ Concentrations are constant, not necessarily equal. The ratio depends on the value of $K$.
- ❌ "The reaction stops at equilibrium" → ✅ Equilibrium is dynamic — both reactions are still occurring, just at equal rates.
- ❌ "A catalyst shifts equilibrium to the right" → ✅ A catalyst does not shift the position. It increases both forward and reverse rates equally. It only makes equilibrium reached faster.
- ❌ Counting solid moles when predicting pressure effects → ✅ Only count gas moles. Solids and liquids are not affected by pressure changes.
- ❌ "Adding inert gas shifts equilibrium" → ✅ At constant volume, adding an inert gas increases total pressure but not the partial pressures of reactants/products. No shift occurs.
🧪 Interactive Virtual Labs
Calcium Carbonate Decomposition
AvailableObserve a dynamic equilibrium system in a closed vessel. Heating shifts equilibrium towards decomposition.
→ Launch Simulation📝 Exam-Style Questions
Question 1: Define "dynamic equilibrium". [2 marks]
Mark Scheme:
- [1 mark] Rate of forward reaction equals rate of reverse reaction.
- [1 mark] Concentrations of reactants and products remain constant (in a closed system).
Question 2: Predict the effect of increasing pressure on: $2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g)$. [2 marks]
Mark Scheme:
- [1 mark] Reactant side has 3 mol gas, product side has 2 mol gas.
- [1 mark] Equilibrium shifts to the right (fewer gas moles) to reduce pressure.
Question 3: State and explain the effect of increasing temperature on the position of equilibrium for an exothermic reaction. [2 marks]
Mark Scheme:
- [1 mark] Equilibrium shifts to the left (toward reactants).
- [1 mark] The system opposes the temperature increase by favoring the endothermic (reverse) direction to absorb heat.
Question 4: Explain why a catalyst is used in the Contact Process even though it has no effect on yield. [1 mark]
Mark Scheme:
- [1 mark] A catalyst increases the rate of reaction, allowing equilibrium to be reached faster (economic viability).
Question 5: For $N_2O_4(g) \rightleftharpoons 2NO_2(g)$ ($\Delta H = +57$ kJ), predict the color change when the tube is placed in hot water. [2 marks]
Mark Scheme:
- [1 mark] Forward reaction is endothermic. Higher temperature favors the forward (endothermic) direction.
- [1 mark] Mixture turns darker brown (more brown $NO_2$ formed).
Question 6: Explain, with reference to the Haber Process, why 450°C is described as a "compromise temperature". [2 marks]
Mark Scheme:
- [1 mark] Low temperature favors yield (exothermic reaction shifts right), but the rate would be too slow.
- [1 mark] High temperature increases rate but decreases yield. 450°C is a compromise between acceptable yield and sufficient rate.
Question 7: State the effect of adding a catalyst on the value of the equilibrium constant $K$. [1 mark]
Mark Scheme:
- [1 mark] No effect. $K$ is only affected by temperature.
Question 8: Explain why adding an inert gas at constant volume has no effect on the position of equilibrium. [2 marks]
Mark Scheme:
- [1 mark] Adding an inert gas increases total pressure, but the individual partial pressures of the reactants and products are unchanged.
- [1 mark] Since the concentrations (or partial pressures) of the reacting species are the same, the rate ratio is unchanged and no shift occurs.