📋 IB Content Statements (R3.2)
This topic covers the following HL syllabus points from the IB Chemistry 2025 guide:
- R3.2.1 (HL): The equilibrium law expression: $K_c = \frac{[C]^c[D]^d}{[A]^a[B]^b}$ for the reaction $aA + bB \rightleftharpoons cC + dD$.
- R3.2.2 (HL): The magnitude of $K_c$ indicates the extent of reaction: $K_c \gg 1$ (mostly products), $K_c \ll 1$ (mostly reactants).
- R3.2.3 (HL): The reaction quotient ($Q$) uses non-equilibrium concentrations and predicts the direction in which equilibrium will be reached.
- R3.2.4 (HL): $K_c$ is only affected by changes in temperature. Changes in concentration, pressure, or catalyst do not change $K_c$.
- R3.2.5 (HL): The relationship between $\Delta G^\theta$ and $K$: $\Delta G^\theta = -RT \ln K$.
📐 Equilibrium Constant ($K_c$)
The Equilibrium Expression
For the reaction: $aA + bB \rightleftharpoons cC + dD$
$$K_c = \frac{[C]^c[D]^d}{[A]^a[B]^b}$$
where $[X]$ represents the equilibrium concentration in $mol \ dm^{-3}$.
Key Rules
- Products go on top (numerator).
- Reactants go on bottom (denominator).
- Powers are the stoichiometric coefficients.
- Pure solids and pure liquids are omitted from the expression (their concentrations are constant).
Magnitude of $K_c$
| Value of $K_c$ | Position of Equilibrium | Interpretation |
|---|---|---|
| $K_c \gg 1$ | Far to the right | Mostly products at equilibrium. Reaction goes almost to completion. |
| $K_c \approx 1$ | Roughly central | Significant amounts of both reactants and products. |
| $K_c \ll 1$ | Far to the left | Mostly reactants. Reaction barely proceeds. |
⚠️ Only temperature changes $K_c$. Adding a catalyst, changing concentration, or changing pressure shifts the position but not the value of $K_c$.
Effect of Temperature on $K_c$
| Reaction Type | Increase Temperature | Effect on $K_c$ |
|---|---|---|
| Exothermic ($\Delta H < 0$) | Shifts left (endothermic direction) | $K_c$ decreases |
| Endothermic ($\Delta H > 0$) | Shifts right (endothermic direction) | $K_c$ increases |
🧊 ICE Tables
Method: Initial → Change → Equilibrium
The ICE table organizes equilibrium calculations. For each species, track the initial concentration, the change that occurs, and the equilibrium concentration.
Worked Example
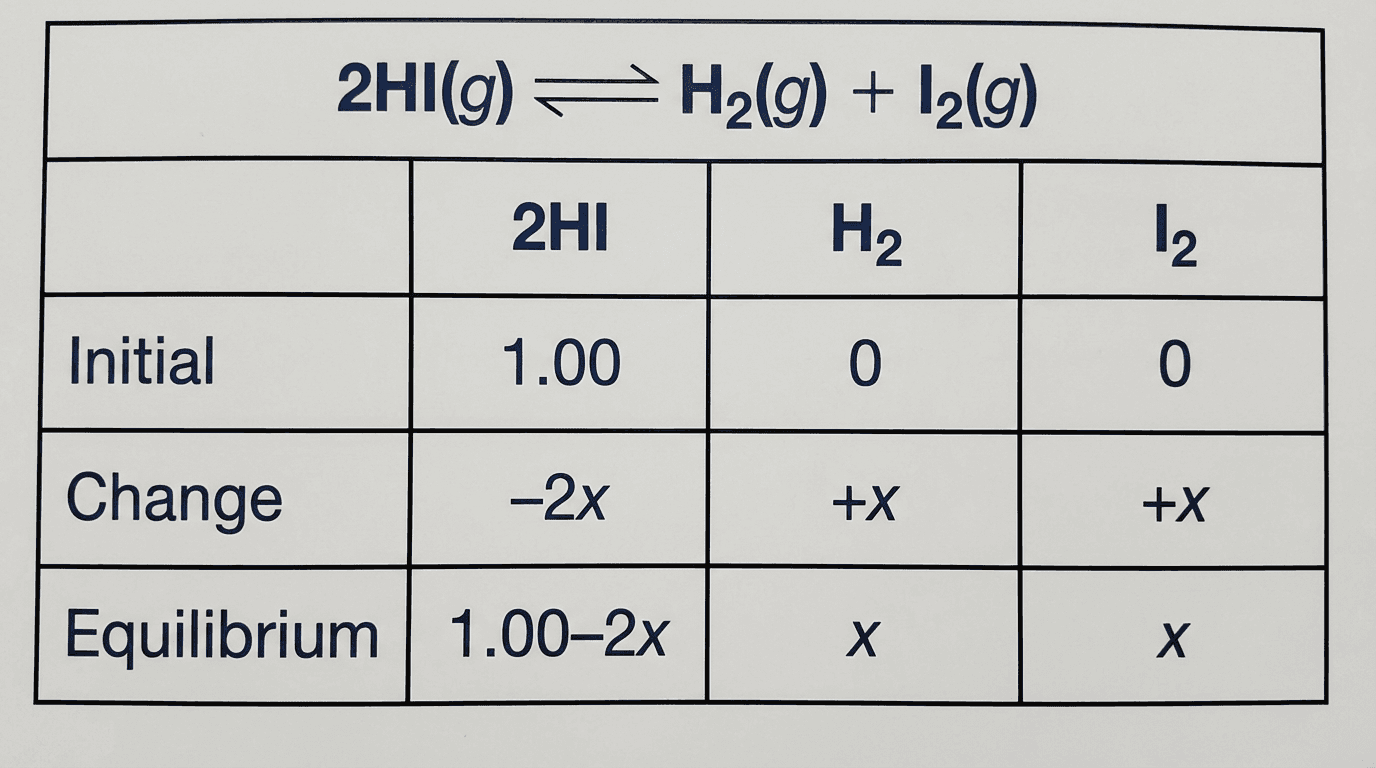
$H_2(g) + I_2(g) \rightleftharpoons 2HI(g)$. Initially, 1.0 mol $H_2$ and 1.0 mol $I_2$ are placed in a 1.0 dm³ flask. At equilibrium, $[HI] = 1.56$ mol/dm³.
| $H_2$ | $I_2$ | $2HI$ | |
|---|---|---|---|
| Initial | 1.00 | 1.00 | 0 |
| Change | $-x$ | $-x$ | $+2x$ |
| Equilibrium | $1.00 - x$ | $1.00 - x$ | $2x = 1.56$ |
Solving: $2x = 1.56 → x = 0.78$. $[H_2]_{eq} = [I_2]_{eq} = 1.00 - 0.78 = 0.22$ mol/dm³.
$$K_c = \frac{[HI]^2}{[H_2][I_2]} = \frac{(1.56)^2}{(0.22)(0.22)} = \frac{2.434}{0.0484} = 50.3$$
📊 Reaction Quotient ($Q$)
What is $Q$?
The reaction quotient ($Q$) is calculated using the same expression as $K_c$, but with non-equilibrium concentrations. It tells you which direction the reaction must shift to reach equilibrium.
| Comparison | Meaning | Shift Direction |
|---|---|---|
| $Q < K_c$ | Product/reactant ratio is too small — not enough products | → Right (toward products) |
| $Q > K_c$ | Product/reactant ratio is too large — too many products | ← Left (toward reactants) |
| $Q = K_c$ | System is at equilibrium | No shift |
⚡ Equilibrium & Gibbs Free Energy
The Relationship
$$\Delta G^\theta = -RT \ln K$$
- $\Delta G^\theta$ = standard Gibbs free energy change (J/mol)
- $R$ = 8.314 J K⁻¹ mol⁻¹
- $T$ = temperature in Kelvin
- $K$ = equilibrium constant
| If… | Then… | Interpretation |
|---|---|---|
| $K > 1$ | $\Delta G^\theta < 0$ | Reaction is spontaneous under standard conditions (products favored) |
| $K = 1$ | $\Delta G^\theta = 0$ | System is at equilibrium under standard conditions |
| $K < 1$ | $\Delta G^\theta > 0$ | Reaction is non-spontaneous under standard conditions (reactants favored) |
🧠 Memory Aids
🔤 $K_c$ Expression — "Products Over Reactants" (POR)
Products are always on top, reactants on bottom. Powers are the stoichiometric coefficients. Omit solids and liquids.
🔤 $Q$ vs $K$ — "Q Chases K"
Imagine $Q$ trying to catch $K$:
- If $Q < K$ → $Q$ needs to increase → make more products → shift right
- If $Q > K$ → $Q$ needs to decrease → make more reactants → shift left
🔤 ICE Table — "I See Equilibrium"
Initial concentrations → Change (use $x$ and stoichiometric ratios: $-x$ for consumed, $+x$ for formed) → Equilibrium concentrations. Substitute into $K_c$ expression.
🔤 $\Delta G$ and $K$ — "Negative G = Go" / "Positive G = No Go"
$\Delta G^\theta < 0$ (negative) → $K> 1$ → products favored → reaction "goes". $\Delta G^\theta > 0$ (positive) → $K < 1$ → reactants favored → "doesn't go" .
🌍 Real-World Applications
🏭 Ammonia Production — $K_c$ and Compromise Conditions
Context: The Haber Process ($K_c$ at 25°C is about $6 \times 10^5$, but at 450°C drops to about 0.5).
Science: For this exothermic reaction, $K_c$ is huge at low temperature (products strongly favored). But the rate is impractically slow. At 450°C, $K_c ≈ 0.5$ (roughly equal reactants and products), but the rate is fast enough for industrial production. This shows how $K_c$ decreases with temperature for exothermic reactions ($\Delta G^\theta$ becomes less negative).
Impact: Industrial chemists must balance thermodynamic favorability ($K$) against kinetic feasibility (rate).
🔋 Battery Design — $\Delta G$ and Cell Voltage
Context: The relationship $\Delta G^\theta = -nFE^\theta$ connects thermodynamics to electrochemistry.
Science: Since $\Delta G^\theta = -RT \ln K$ and $\Delta G^\theta = -nFE^\theta$, we can link $K$ to cell voltage: a large $K$ means a large, positive $E^\theta$ and a spontaneous cell reaction. Battery chemists use this to predict whether a cell will work before building it.
Impact: This is how lithium-ion batteries were designed — by calculating $\Delta G$ and $K$ for candidate electrode reactions.
💊 Drug Solubility — $K_{sp}$ in Pharmaceuticals
Context: The solubility product ($K_{sp}$) is a special form of $K_c$ for sparingly soluble salts.
Science: For $BaSO_4(s) \rightleftharpoons Ba^{2+}(aq) + SO_4^{2-}(aq)$, $K_{sp} = [Ba^{2+}][SO_4^{2-}] = 1.1 \times 10^{-10}$ (very small). This extremely low solubility makes barium sulfate safe to drink for X-ray contrast imaging, despite barium ions being toxic — $Q$ stays above $K_{sp}$ so it remains solid in the gut.
Impact: Millions of patients safely drink barium sulfate suspensions for medical imaging every year.
⚠️ Common Mistakes
- ❌ Including solids or pure liquids in $K_c$ → ✅ Only aqueous and gaseous species appear in the $K_c$ expression. Solids and pure liquids have constant concentrations and are omitted.
- ❌ "Increasing concentration changes $K_c$" → ✅ Only temperature changes $K_c$. Adding more reactant shifts the position of equilibrium but the value of $K_c$ stays the same.
- ❌ Confusing $Q$ and $K$ → ✅ $K$ uses equilibrium concentrations. $Q$ uses any concentrations (at the current moment) and is compared to $K$ to predict direction of shift.
- ❌ Using kJ in the $\Delta G = -RT \ln K$ calculation → ✅ $R = 8.314$ J K⁻¹ mol⁻¹, so $\Delta G^\theta$ from this equation is in J/mol. Convert to kJ by dividing by 1000.
- ❌ ICE table: wrong stoichiometric ratios → ✅ The "Change" row must follow the stoichiometric ratio. If the coefficient is 2, the change is $±2x$, not $±x$.
📝 Exam-Style Questions
Question 1: At 700 K, the reaction $2SO_2 + O_2 \rightleftharpoons 2SO_3$ has $K_c = 4.3 \times 10^6$. Comment on the position of equilibrium. [1 mark]
Mark Scheme:
- [1 mark] $K_c \gg 1$, so equilibrium lies far to the right (products strongly favored).
Question 2: Write the $K_c$ expression for: $4NH_3(g) + 5O_2(g) \rightleftharpoons 4NO(g) + 6H_2O(g)$. [1 mark]
Mark Scheme:
- [1 mark] $K_c = \frac{[NO]^4[H_2O]^6}{[NH_3]^4[O_2]^5}$
Question 3: Calculate $\Delta G^\theta$ for a reaction with $K_c = 1.5$ at 298 K. ($R = 8.314$ J K⁻¹ mol⁻¹). [2 marks]
Mark Scheme:
- [1 mark] $\Delta G^\theta = -RT \ln K = -(8.314)(298) \ln(1.5)$.
- [1 mark] $= -(2477.6)(0.405) = -1004$ J/mol = −1.0 kJ/mol.
Question 4: For $A + B \rightleftharpoons C + D$, given $[A]=0.1$, $[B]=0.1$, $[C]=0.5$, $[D]=0.5$ and $K_c=50$. Calculate $Q$ and predict shift. [2 marks]
Mark Scheme:
- [1 mark] $Q = \frac{(0.5)(0.5)}{(0.1)(0.1)} = 25$.
- [1 mark] $Q (25) < K_c (50)$ → shifts right (toward products).
Question 5: State the effect of adding a catalyst on the value of $K_c$. [1 mark]
Mark Scheme:
- [1 mark] No effect. $K_c$ is only affected by temperature.
Question 6: 0.50 mol of $PCl_5$ is placed in a 1.0 dm³ flask at 250°C. At equilibrium, 0.16 mol of $Cl_2$ is present. $PCl_5(g) \rightleftharpoons PCl_3(g) + Cl_2(g)$. Calculate $K_c$. [3 marks]
Mark Scheme:
- [1 mark] ICE: $[Cl_2]_{eq} = 0.16$, so $x = 0.16$. $[PCl_3]_{eq} = 0.16$. $[PCl_5]_{eq} = 0.50 - 0.16 = 0.34$.
- [1 mark] $K_c = \frac{[PCl_3][Cl_2]}{[PCl_5]} = \frac{(0.16)(0.16)}{0.34}$.
- [1 mark] $K_c = 0.075$.
Question 7: Explain why $K_c$ for an exothermic reaction decreases when temperature increases. [2 marks]
Mark Scheme:
- [1 mark] Increasing temperature favors the endothermic (reverse) direction → equilibrium shifts left.
- [1 mark] More reactants and fewer products at equilibrium → the ratio $\frac{[products]}{[reactants]}$ decreases → $K_c$ decreases.
Question 8: If $\Delta G^\theta$ for a reaction is $-10.5$ kJ/mol at 298 K, calculate $K$. [2 marks]
Mark Scheme:
- [1 mark] $-10500 = -(8.314)(298) \ln K$ → $\ln K = \frac{10500}{2477.6} = 4.24$.
- [1 mark] $K = e^{4.24} = \mathbf{69.4}$.