📋 IB Content Statements (R4.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- R4.1.1: A Brønsted-Lowry acid is a proton ($H^+$) donor, and a Brønsted-Lowry base is a proton acceptor.
- R4.1.2: A conjugate acid-base pair differs by one proton ($H^+$).
- R4.1.3: An amphiprotic substance can act as both a Brønsted-Lowry acid and base (e.g., water, $HSO_4^-$, amino acids).
- R4.1.4: $pH = -\log[H^+]$ and $[H^+] = 10^{-pH}$.
- R4.1.5: The ionic product of water: $K_w = [H^+][OH^-] = 1.00 \times 10^{-14}$ at 298 K.
- R4.1.6: Strong acids and bases dissociate completely. Weak acids and bases dissociate partially (equilibrium).
- R4.1.7: The shape and features of titration curves for combinations of strong and weak acids with strong and weak bases.
HL Extensions
- R4.1.8: A Lewis acid is an electron pair acceptor. A Lewis base is an electron pair donor.
- R4.1.9: The acid dissociation constant ($K_a$) and its relationship to acid strength: $K_a = \frac{[H^+][A^-]}{[HA]}$.
- R4.1.10: $pK_a = -\log K_a$. The smaller the $pK_a$, the stronger the acid.
- R4.1.11: Calculation of pH for strong acids/bases, and weak acids using $K_a$.
🧪 Theories of Acids & Bases
| Feature | Brønsted-Lowry | Lewis (HL) |
|---|---|---|
| Acid | Proton ($H^+$) donor | Electron pair acceptor |
| Base | Proton ($H^+$) acceptor | Electron pair donor |
| Scope | Requires proton transfer | Broader: includes reactions without protons |
| Examples | $HCl$, $NaOH$, $NH_3$ | $BF_3$ (acid), $NH_3$ (base), metal ions |
Key Insight
All Brønsted-Lowry acids are Lewis acids, but not all Lewis acids are Brønsted-Lowry acids. For example, $BF_3$ has no proton to donate — it can only act as a Lewis acid (it accepts a lone pair from $NH_3$ to form $BF_3NH_3$).
Amphiprotic Substances
An amphiprotic species can act as both an acid and a base. Water is the most important example:
- As acid: $H_2O + NH_3 \rightleftharpoons NH_4^+ + OH^-$ (donates $H^+$)
- As base: $H_2O + HCl \rightarrow H_3O^+ + Cl^-$ (accepts $H^+$)
- Other examples: $HSO_4^-$, $HCO_3^-$, amino acids
🔗 Conjugate Acid-Base Pairs
Key Definition
A conjugate acid-base pair consists of two species that differ by exactly one proton ($H^+$). When an acid donates a proton, it becomes its conjugate base. When a base accepts a proton, it becomes its conjugate acid.
Example: $NH_3 + H_2O \rightleftharpoons NH_4^+ + OH^-$
| Pair | Acid | Conjugate Base |
|---|---|---|
| Pair 1 | $H_2O$ (donates $H^+$) | $OH^-$ |
| Pair 2 | $NH_4^+$ | $NH_3$ (accepts $H^+$) |
Strength relationship: The stronger the acid, the weaker its conjugate base (and vice versa). $HCl$ is a strong acid → $Cl^-$ is a very weak base (virtually no tendency to accept $H^+$). $CH_3COOH$ is a weak acid → $CH_3COO^-$ is a relatively stronger conjugate base.
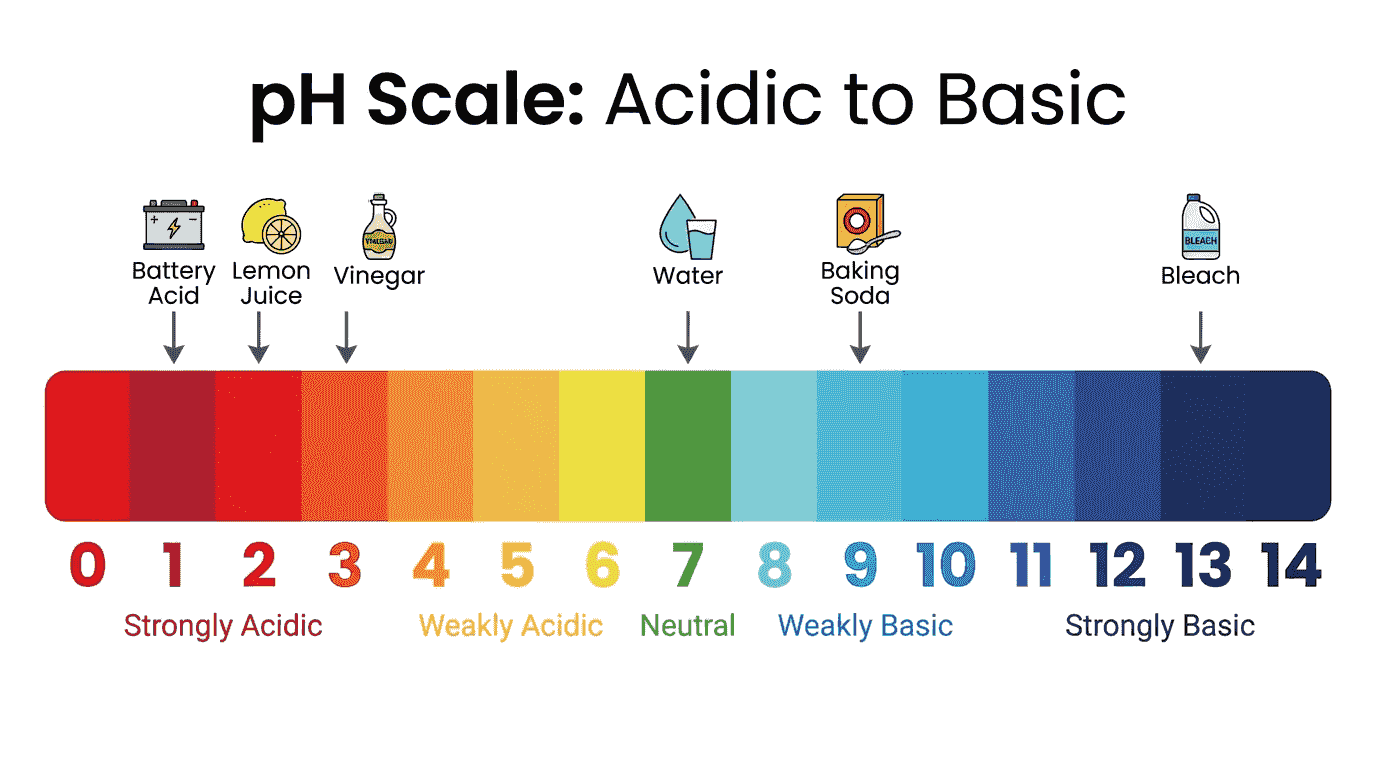
📏 The pH Scale
Key Equations
$$pH = -\log[H^+]$$
$$[H^+] = 10^{-pH}$$
$$K_w = [H^+][OH^-] = 1.00 \times 10^{-14} \text{ (at 298 K)}$$
| pH Range | Nature | $[H^+]$ vs $[OH^-]$ | Example |
|---|---|---|---|
| 0 – 6.9 | Acidic | $[H^+] > [OH^-]$ | Stomach acid (pH 1-2), vinegar (pH 3) |
| 7.0 | Neutral | $[H^+] = [OH^-]$ | Pure water at 298 K |
| 7.1 – 14 | Basic/Alkaline | $[H^+] < [OH^-]$ | Blood (pH 7.4), bleach (pH 12) |
The logarithmic scale: A change of 1 pH unit = a 10-fold change in $[H^+]$. Moving from pH 3 to pH 1 means the $[H^+]$ is 100 times larger ($10^2$).
pH Calculations
Strong acid: $[H^+] = [acid]$ (complete dissociation) → $pH = -\log[acid]$
Strong base: $[OH^-] = [base]$ → $pOH = -\log[base]$ → $pH = 14 - pOH$
Weak acid: Use $K_a$ expression: $K_a = \frac{[H^+][A^-]}{[HA]}$. Assuming $[H^+] = [A^-] = x$ and $[HA] \approx c - x \approx c$:
$$[H^+] = \sqrt{K_a \times c}$$
💪 Strong vs Weak Acids & Bases
Key Distinction
Strong ≠ Concentrated. "Strong" describes the extent of dissociation (complete vs partial), not the amount of acid. You can have a dilute solution of a strong acid (e.g., 0.001 mol dm⁻³ HCl) and a concentrated solution of a weak acid (e.g., 10 mol dm⁻³ CH₃COOH).
| Property | Strong Acid (e.g., $HCl$) | Weak Acid (e.g., $CH_3COOH$) |
|---|---|---|
| Dissociation | Complete ($\rightarrow$) | Partial ($\rightleftharpoons$) |
| $[H^+]$ (same conc.) | High | Low |
| pH (same conc.) | Lower (e.g., pH 1) | Higher (e.g., pH 3) |
| Conductivity | High (more ions) | Low (fewer ions) |
| Rate of reaction with Mg | Fast / vigorous | Slow / gentle |
| Volume of NaOH to neutralize | Same | Same (same moles of acid) |
⚠️ Must-know strong acids: $HCl$, $HNO_3$, $H_2SO_4$ (diprotic: first dissociation strong, second weak). Must-know strong bases: $NaOH$, $KOH$, $Ba(OH)_2$.
Common Weak Acids & Bases
| Weak Acids | Weak Bases |
|---|---|
| $CH_3COOH$ (ethanoic/acetic acid) | $NH_3$ (ammonia) |
| $H_2CO_3$ (carbonic acid) | $CH_3NH_2$ (methylamine) |
| $H_3PO_4$ (phosphoric acid) | $C_2H_5NH_2$ (ethylamine) |
| $HF$ (hydrofluoric acid) | $CO_3^{2-}$ (carbonate ion) |
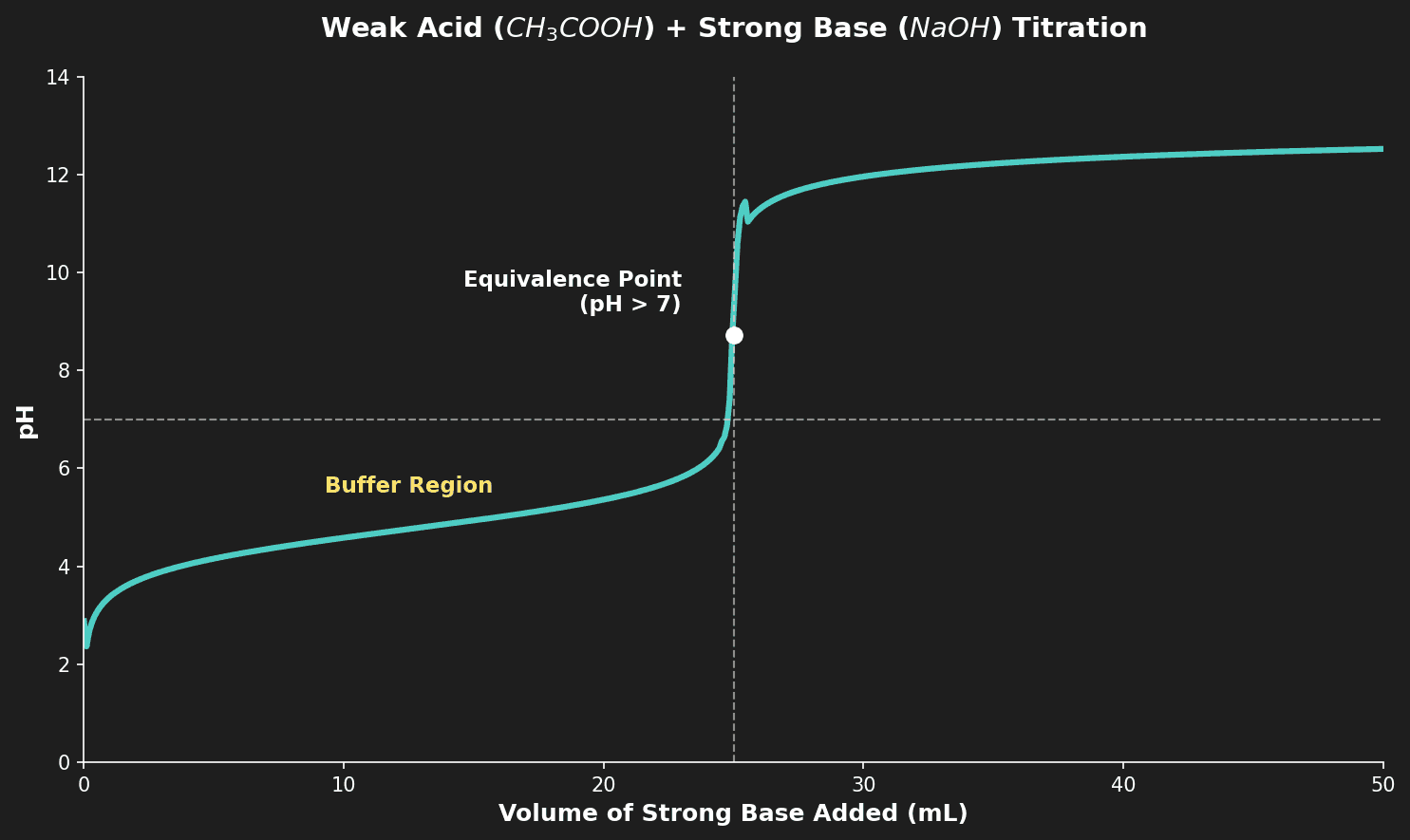
📈 Titration Curves
What is a Titration Curve?
A titration curve is a graph of pH vs volume of titrant added. The shape of the curve depends on whether the acid and base are strong or weak.
| Combination | Start pH | Equivalence pH | End pH | Buffer Region? | Suitable Indicator |
|---|---|---|---|---|---|
| Strong acid + Strong base | ~1 | 7.0 | ~13 | No | Any (phenolphthalein, methyl orange) |
| Weak acid + Strong base | ~3 | > 7 (basic salt) | ~13 | Yes (before equivalence) | Phenolphthalein |
| Strong acid + Weak base | ~1 | < 7 (acidic salt) | ~9 | Yes (before equivalence) | Methyl orange |
| Weak acid + Weak base | ~3 | Variable | ~9 | Yes | No suitable indicator (no sharp change) |
Key features to identify on a titration curve:
- Initial pH: Tells you the acid or base you start with
- Equivalence point: Where moles of acid = moles of base (steep vertical section)
- Half-equivalence point (HL): $pH = pK_a$ (half the volume needed to reach equivalence)
- Buffer region: The flat section before equivalence in weak acid or weak base titrations
🧪 Interactive Virtual Labs
Experiment 1: Acid-Base Titration
AvailablePerform a virtual titration using indicators. Generate titration curves for different acid/base combinations and observe the equivalence point.
→ Launch SimulationExperiment 2: Weak Acid pH Calculator
AvailableExplore the relationship between concentration, $K_a$, and pH for weak acids.
→ Launch Simulation🧠 Memory Aids & Mnemonics
🔤 Brønsted-Lowry Definition
"Acids are Proton Pushers, Bases are Proton Pullers"
Acid = proton donor (pushes $H^+$ away). Base = proton acceptor (pulls $H^+$ in). This is the most-tested definition in IB.
🔤 Lewis Definition
"Lewis Acids Love Electrons" (they Accept lone pairs)
Lewis acid = electron pair acceptor (empty orbital). Lewis base = electron pair donor (has lone pair to give). Remember: Lewis is the BROADER definition.
🔤 Strong vs Concentrated
"Strong describes the ACID. Concentrated describes the SOLUTION."
Strong = complete dissociation (a property of the substance itself). Concentrated = lots of moles per dm³ (a property of the solution). Never confuse the two!
🔤 pH Scale Direction
"Low pH = Lots of H⁺" → More acidic
pH 1 = $0.1$ mol dm⁻³ $H^+$. pH 7 = $10^{-7}$ mol dm⁻³ $H^+$. pH 13 = $10^{-13}$ mol dm⁻³ $H^+$. As pH goes DOWN, $[H^+]$ goes UP.
🔤 Conjugate Pair Rule
"Remove a proton → you get the conjugate BASE. Add a proton → you get the conjugate ACID."
$HCl \xrightarrow{-H^+} Cl^-$ (conjugate base). $NH_3 \xrightarrow{+H^+} NH_4^+$ (conjugate acid).
🌍 Real-World Applications
🦷 Tooth Decay and Acid Erosion
Context: Why does drinking soda dissolve your teeth? The answer is directly linked to pH and acid strength.
Science: Tooth enamel is made of hydroxyapatite ($Ca_5(PO_4)_3OH$), a basic compound that dissolves in acid: $Ca_5(PO_4)_3OH(s) + H^+(aq) \rightarrow Ca^{2+}(aq) + HPO_4^{2-}(aq) + H_2O(l)$. Bacterial metabolism of sugar produces lactic acid (a weak acid, pH ~4) on tooth surfaces. Cola drinks (pH 2.5) and citrus juices (pH 3.5) are even more erosive. Fluoride toothpaste replaces $OH^-$ with $F^-$ to form fluorapatite ($Ca_5(PO_4)_3F$), which is more resistant to acid attack because $F^-$ is a weaker base than $OH^-$.
Impact: Dentistry relies on understanding acid-base chemistry. Patients with gastric reflux (stomach acid pH 1-2) suffer severe enamel erosion, and treatment involves neutralizing the acid with basic antacids ($Mg(OH)_2$, $NaHCO_3$).
🫧 Blood pH and Buffers
Context: Human blood pH must stay between 7.35 and 7.45. Even small deviations (acidosis < 7.35 or alkalosis> 7.45) can be fatal. How does the body maintain such tight control?
Science: The blood uses a carbonic acid-bicarbonate buffer system: $CO_2(aq) + H_2O(l) \rightleftharpoons H_2CO_3(aq) \rightleftharpoons H^+(aq) + HCO_3^-(aq)$. If $[H^+]$ rises (e.g., during exercise from lactic acid production), $HCO_3^-$ (the conjugate base) reacts with excess $H^+$ to form $H_2CO_3$, which decomposes to $CO_2$ and is exhaled. If $[H^+]$ falls, $H_2CO_3$ dissociates to release more $H^+$.
Impact: Understanding acid-base equilibria is essential in medicine. Diabetic ketoacidosis (blood pH drops below 7.0) is a medical emergency where the body's buffer system is overwhelmed by ketone acids.
🌧️ Acid Rain
Context: Normal rainwater has a pH of about 5.6 (slightly acidic due to dissolved $CO_2$). Acid rain, with pH below 5.0, causes environmental damage to forests, lakes, and buildings.
Science: Sulfur dioxide ($SO_2$) and nitrogen oxides ($NO_x$) from fossil fuel combustion dissolve in rainwater: $SO_2 + H_2O \rightarrow H_2SO_3$ (sulfurous acid) and $2SO_2 + O_2 + 2H_2O \rightarrow 2H_2SO_4$ (sulfuric acid — a strong acid). Acid rain with pH 3-4 damages limestone buildings because: $CaCO_3(s) + H_2SO_4(aq) \rightarrow CaSO_4(s) + H_2O(l) + CO_2(g)$. Lakes become acidified, killing fish (most cannot survive below pH 5) and disrupting ecosystems.
Impact: Legislation requiring catalytic converters (removing $NO_x$) and desulfurization of power plant emissions has reduced acid rain significantly since the 1980s. Liming (adding $CaCO_3$) is used to neutralize acidified lakes — a direct application of acid-base chemistry.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Confusing strong/weak with concentrated/dilute: "Strong" describes the degree of dissociation (a property of the acid). "Concentrated" describes the amount dissolved (a property of the solution). A dilute strong acid still dissociates completely.
- Writing the wrong arrow for weak acids: Weak acids use the equilibrium arrow ($\rightleftharpoons$), NOT the forward arrow ($\rightarrow$). Using $\rightarrow$ for $CH_3COOH$ implies complete dissociation (wrong).
- Forgetting that neutralization volume is the SAME for strong and weak acids: At the same concentration and volume, a strong acid and a weak acid require the same volume of base to neutralize (same moles of $H^+$ available). The difference is in speed and starting pH, not total moles.
- Incorrect conjugate pair identification: The conjugate base of $H_2SO_4$ is $HSO_4^-$ (not $SO_4^{2-}$). Remove only one proton at a time.
- Equivalence point pH errors on titration curves: Strong acid + strong base → equivalence at pH 7. Weak acid + strong base → equivalence above 7 (basic salt). Strong acid + weak base → equivalence below 7 (acidic salt).
- Applying $pH = -\log[acid]$ to weak acids: This formula only works for strong acids (complete dissociation). For weak acids, you must use $K_a$ to find $[H^+]$ first.
📝 IB-Style Exam Questions
Question 1: Distinguish between a strong acid and a weak acid. [2 marks]
Mark Scheme:
- [1 mark] Strong acids dissociate completely in solution.
- [1 mark] Weak acids dissociate only partially (equilibrium is established).
Question 2: Calculate the pH of a $0.05 \ mol \ dm^{-3}$ solution of $HCl$. [1 mark]
Mark Scheme:
- [1 mark] pH = $-\log(0.05) = 1.30$. ($HCl$ is a strong acid, so $[H^+] = [HCl] = 0.05$)
Question 3: Identify the conjugate base of $H_2SO_4$. [1 mark]
Mark Scheme:
- [1 mark] $HSO_4^-$ (remove one $H^+$).
Question 4: Explain why $BF_3$ is a Lewis acid but not a Brønsted-Lowry acid. [2 marks]
Mark Scheme:
- [1 mark] $BF_3$ has an empty orbital and can accept an electron pair (Lewis acid definition).
- [1 mark] $BF_3$ has no proton ($H^+$) to donate (cannot act as Brønsted-Lowry acid).
Question 5: Explain why the pH at the equivalence point of a titration between ethanoic acid and sodium hydroxide is greater than 7. [2 marks]
Mark Scheme:
- [1 mark] At equivalence, only sodium ethanoate ($CH_3COONa$) is in solution / the salt of a weak acid and strong base.
- [1 mark] The ethanoate ion ($CH_3COO^-$) is a relatively strong conjugate base that hydrolyzes: $CH_3COO^- + H_2O \rightleftharpoons CH_3COOH + OH^-$, producing $OH^-$ and raising the pH above 7.
Question 6: Describe two differences that would be observed between equal concentrations of $HCl$ and $CH_3COOH$ reacting with magnesium ribbon. [2 marks]
Mark Scheme:
- [1 mark] $HCl$ reacts faster / more vigorously / more rapid bubbling because it has a higher $[H^+]$ (complete dissociation).
- [1 mark] Both produce the same total volume of gas ($H_2$) because they have the same number of moles of acid.
Question 7: Calculate the pH of a $0.10 \ mol \ dm^{-3}$ solution of $NaOH$. [2 marks]
Mark Scheme:
- [1 mark] $[OH^-] = 0.10$, so $pOH = -\log(0.10) = 1.00$.
- [1 mark] $pH = 14 - pOH = 14 - 1.00 = 13.00$.
Question 8: Water is described as an amphiprotic substance. Define 'amphiprotic' and write equations to illustrate this property. [3 marks]
Mark Scheme:
- [1 mark] Amphiprotic means a substance can act as both a Brønsted-Lowry acid and a Brønsted-Lowry base.
- [1 mark] As acid (proton donor): $H_2O + NH_3 \rightleftharpoons NH_4^+ + OH^-$.
- [1 mark] As base (proton acceptor): $H_2O + HCl \rightarrow H_3O^+ + Cl^-$.