📋 IB Content Statements (R4.2)
This topic covers the following HL syllabus points from the IB Chemistry 2025 guide:
- R4.2.1 (HL): A buffer solution resists changes in pH on the addition of small amounts of acid, base, or on dilution.
- R4.2.2 (HL): Acidic buffers consist of a weak acid and its conjugate base (salt). Basic buffers consist of a weak base and its conjugate acid (salt).
- R4.2.3 (HL): The Henderson-Hasselbalch equation: $pH = pK_a + \log\frac{[A^-]}{[HA]}$.
- R4.2.4 (HL): Buffer capacity is greatest when $[HA] = [A^-]$ (i.e., $pH = pK_a$).
- R4.2.5 (HL): The pH of a salt solution can be predicted by considering the relative strengths of the parent acid and base (hydrolysis).
🛡️ Buffer Solutions
Definition
A buffer is a solution that resists changes in pH when small amounts of acid or base are added, or when the solution is diluted.
Types of Buffers
| Type | Composition | Example | pH Range |
|---|---|---|---|
| Acidic Buffer | Weak acid + conjugate base (salt) | $CH_3COOH$ + $CH_3COONa$ | < 7 |
| Basic Buffer | Weak base + conjugate acid (salt) | $NH_3$ + $NH_4Cl$ | > 7 |
How Buffers Work (Acidic Buffer Example)
Consider a buffer made from $CH_3COOH$ (weak acid) and $CH_3COO^-$ (conjugate base):
- If $H^+$ is added: The conjugate base ($CH_3COO^-$) reacts with the added acid: $CH_3COO^- + H^+ → CH_3COOH$. The extra $H^+$ is consumed, so pH barely changes.
- If $OH^-$ is added: The weak acid ($CH_3COOH$) reacts with the added base: $CH_3COOH + OH^- → CH_3COO^- + H_2O$. The extra $OH^-$ is consumed, so pH barely changes.
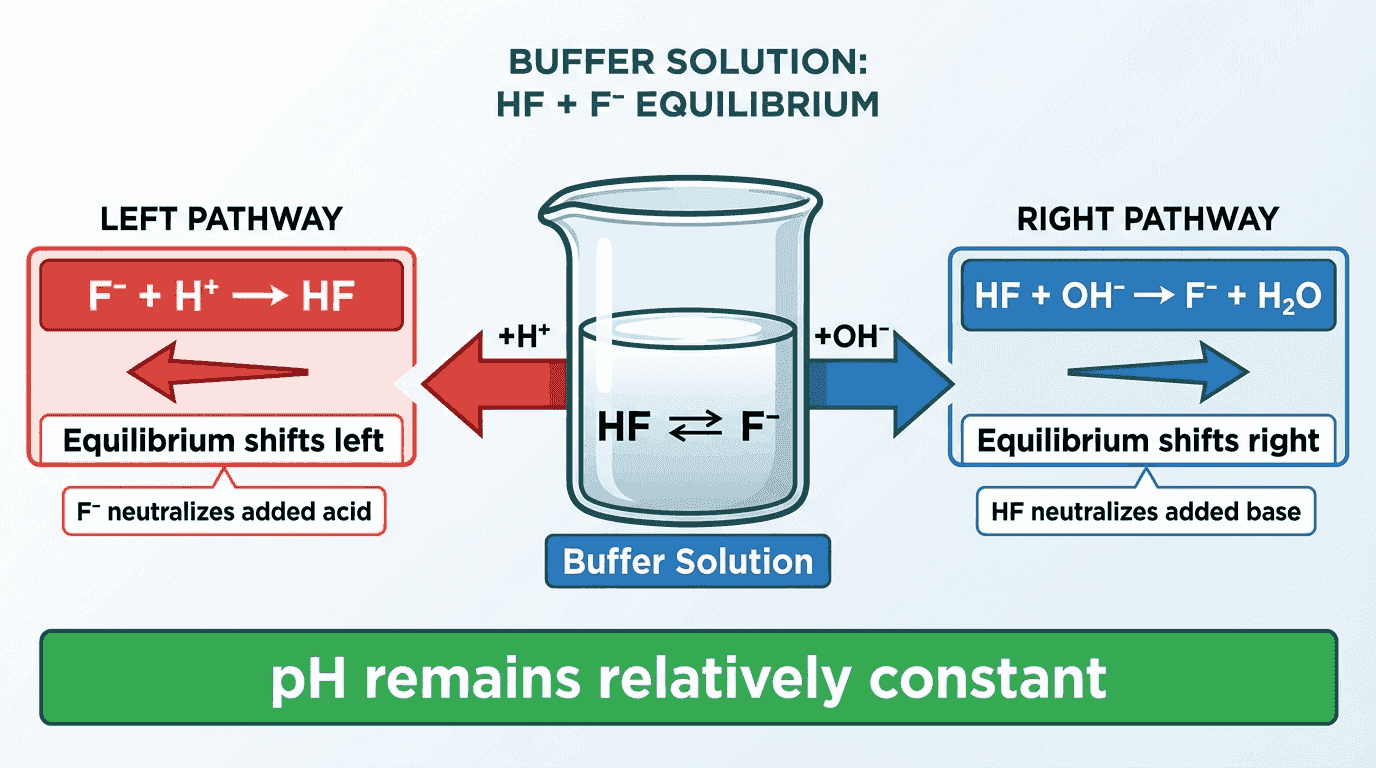
🧮 Buffer Calculations
Henderson-Hasselbalch Equation
$$pH = pK_a + \log\left(\frac{[A^-]}{[HA]}\right)$$
For basic buffers:
$$pOH = pK_b + \log\left(\frac{[BH^+]}{[B]}\right)$$
Worked Example
Calculate the pH of a buffer containing 0.30 M ethanoic acid ($pK_a = 4.76$) and 0.15 M sodium ethanoate.
$$pH = 4.76 + \log\left(\frac{0.15}{0.30}\right) = 4.76 + \log(0.50) = 4.76 + (-0.30) = 4.46$$
Buffer Capacity
Buffer capacity is the amount of acid or base a buffer can neutralize before its pH changes significantly.
- Greatest when $[HA] = [A^-]$ (equal amounts of acid and salt).
- At this point, $pH = pK_a$ (the log term = 0).
- Increasing total buffer concentration (while keeping the ratio constant) increases capacity.
💧 Salt Hydrolysis
Key Principle
When a salt dissolves in water, ions from the weak parent acid or base will hydrolyze (react with water), changing the pH. Ions from strong parents do not hydrolyze.
| Salt Origin | Example | Solution pH | Hydrolyzing Ion | Equation |
|---|---|---|---|---|
| Strong Acid + Strong Base | $NaCl$ | 7 (Neutral) | None | — |
| Strong Acid + Weak Base | $NH_4Cl$ | < 7 (Acidic) | $NH_4^+$ | $NH_4^+ + H_2O \rightleftharpoons NH_3 + H_3O^+$ |
| Weak Acid + Strong Base | $CH_3COONa$ | > 7 (Basic) | $CH_3COO^-$ | $CH_3COO^- + H_2O \rightleftharpoons CH_3COOH + OH^-$ |
| Weak Acid + Weak Base | $CH_3COONH_4$ | Depends on $K_a$ vs $K_b$ | Both ions | Compare $K_a$ and $K_b$ values |
Highly Charged Cation Hydrolysis
Small, highly charged cations like $Al^{3+}$ and $Fe^{3+}$ have high charge density. They polarize water molecules in their hydration shell, causing hydrolysis:
$$[Al(H_2O)_6]^{3+} \rightleftharpoons [Al(H_2O)_5(OH)]^{2+} + H^+$$
This is why solutions of $AlCl_3$ and $FeCl_3$ are acidic even though they come from a strong acid.
🧠 Memory Aids
🔤 Buffer Composition — "WAC" (Weak Acid + Conjugate base)
Acidic buffer = Weak Acid + Conjugate base salt. Basic buffer = Weak Base + Conjugate acid salt. The weak species is the reservoir; its conjugate partner neutralizes the threat.
🔤 How It Works — "The Sponge"
Think of the buffer as a pH sponge: $A^-$ soaks up added $H^+$, and $HA$ soaks up added $OH^-$. The buffer "absorbs" the disturbance until one reservoir runs out.
🔤 Salt Hydrolysis — "Weak Side Wins"
The ion from the weak parent hydrolyzes. If the weak parent was an acid → conjugate base is in solution → accepts $H^+$ from water → solution is basic. If the weak parent was a base → conjugate acid donates $H^+$ → solution is acidic.
🔤 Henderson-Hasselbalch — "pKa is the Anchor"
The $pK_a$ sets the center of the buffer range. When $[A^-] = [HA]$, the log term = 0 and $pH = pK_a$ exactly. Buffers work best within ±1 pH unit of $pK_a$.
🌍 Real-World Applications
🩸 Blood Buffer System
Context: Blood pH must stay between 7.35–7.45 for survival.
Science: The carbonic acid/bicarbonate buffer maintains blood pH: $H_2CO_3 / HCO_3^-$. When exercise produces lactic acid ($H^+$), the bicarbonate ion neutralizes it: $HCO_3^- + H^+ → H_2CO_3 → CO_2 + H_2O$. You then breathe out the $CO_2$ to remove the acid. Similarly, if blood becomes too basic, $H_2CO_3$ donates $H^+$.
Impact: A blood pH below 7.0 or above 7.8 is fatal. This is the most critical buffer system in the human body.
🧴 Cosmetic & Pharmaceutical Buffers
Context: Skin has a natural pH of about 5.5 (slightly acidic).
Science: Skincare products use citrate or phosphate buffers to maintain a pH compatible with skin. Anti-acne creams (salicylic acid, $pK_a ≈ 3.0$) are buffered to pH ~3.5 so the acid is partially ionized — strong enough to exfoliate but buffered enough not to cause chemical burns.
Impact: Without buffering, many pharmaceutical formulations would be either ineffective or dangerous.
🌊 Ocean Buffering and CO₂
Context: Seawater acts as a natural buffer via the $CO_2/HCO_3^-/CO_3^{2-}$ system.
Science: Dissolved $CO_2$ forms $H_2CO_3$ which releases $H^+$. The carbonate ion ($CO_3^{2-}$) acts as the conjugate base, neutralizing excess $H^+$: $CO_3^{2-} + H^+ → HCO_3^-$. However, as atmospheric $CO_2$ increases, the ocean's buffer capacity is being overwhelmed.
Impact: Ocean pH has dropped by ~0.1 units since 1800. While this sounds small, pH is logarithmic — this represents a 26% increase in $[H^+]$.
⚠️ Common Mistakes
- ❌ "A buffer prevents pH from changing" → ✅ A buffer resists pH change but does not prevent it. It works only for small additions. Too much acid/base will overwhelm the buffer.
- ❌ "A strong acid + strong base salt can be a buffer" → ✅ Buffers require a weak acid or base and its conjugate. $NaCl$ is NOT a buffer — it has no reservoir to react with added $H^+$ or $OH^-$.
- ❌ Confusing $[HA]$ and $[A^-]$ in Henderson-Hasselbalch → ✅ $[A^-]$ (conjugate base/salt) goes on top. $[HA]$ (weak acid) goes on the bottom.
- ❌ "Salt hydrolysis: the strong parent determines pH" → ✅ The weak parent's ion is the one that hydrolyzes. Ions from strong acids/bases are spectator ions and don't affect pH.
- ❌ "Adding water to a buffer dramatically changes pH" → ✅ Dilution changes both $[HA]$ and $[A^-]$ equally, so their ratio stays the same. The Henderson-Hasselbalch equation shows pH barely changes (though capacity decreases).
🧪 Interactive Virtual Labs
Buffer Capacity Lab
AvailableCreate buffers of different ratios and test their ability to resist pH change upon addition of HCl/NaOH.
→ Launch Simulation📝 Exam-Style Questions
Question 1: Calculate the pH of a buffer containing 0.1 M ethanoic acid ($pK_a = 4.76$) and 0.2 M sodium ethanoate. [2 marks]
Mark Scheme:
- [1 mark] $pH = 4.76 + \log(0.2 / 0.1)$.
- [1 mark] $pH = 4.76 + 0.30 = \mathbf{5.06}$.
Question 2: Explain how an acidic buffer resists pH change when a small amount of $NaOH$ is added. [2 marks]
Mark Scheme:
- [1 mark] The added $OH^-$ reacts with the weak acid ($HA$): $HA + OH^- → A^- + H_2O$.
- [1 mark] The $[H^+]$ (or pH) remains approximately constant because the weak acid reservoir is consumed rather than free $OH^-$ remaining.
Question 3: Explain why a solution of aluminium chloride ($AlCl_3$) is acidic. [2 marks]
Mark Scheme:
- [1 mark] $Al^{3+}$ has high charge density (small, highly charged ion).
- [1 mark] It polarizes water molecules in its hydration shell, releasing $H^+$: $[Al(H_2O)_6]^{3+} \rightleftharpoons [Al(H_2O)_5(OH)]^{2+} + H^+$.
Question 4: Predict whether a solution of $NH_4CH_3COO$ is acidic, basic, or neutral. ($pK_a(CH_3COOH) = 4.76$, $pK_b(NH_3) = 4.75$). [1 mark]
Mark Scheme:
- [1 mark] Approximately neutral (~pH 7) because $pK_a ≈ pK_b$ (or $K_a ≈ K_b$) for the parent acid and base.
Question 5: State the components required to make a basic buffer, with an example. [1 mark]
Mark Scheme:
- [1 mark] A weak base and its conjugate acid salt (e.g., $NH_3$ and $NH_4Cl$).
Question 6: A buffer is prepared by mixing 0.40 mol $CH_3COOH$ and 0.40 mol $CH_3COONa$ in 1.0 dm³ water ($pK_a = 4.76$). Calculate the pH. [1 mark]
Mark Scheme:
- [1 mark] $pH = 4.76 + \log(0.40/0.40) = 4.76 + 0 = \mathbf{4.76}$. (When $[A^-] = [HA]$, $pH = pK_a$.)
Question 7: Explain why a solution of $Na_2CO_3$ (sodium carbonate) is basic. [2 marks]
Mark Scheme:
- [1 mark] $CO_3^{2-}$ is the conjugate base of the weak acid $HCO_3^-$ (or $H_2CO_3$).
- [1 mark] It hydrolyzes water: $CO_3^{2-} + H_2O \rightleftharpoons HCO_3^- + OH^-$, producing $OH^-$ ions.
Question 8: Explain why diluting a buffer solution with water has minimal effect on its pH. [2 marks]
Mark Scheme:
- [1 mark] Dilution decreases both $[HA]$ and $[A^-]$ by the same factor.
- [1 mark] The ratio $\frac{[A^-]}{[HA]}$ remains constant, so the $\log$ term in Henderson-Hasselbalch doesn't change and pH is approximately unchanged.