📋 IB Content Statements (R5.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- R5.1.1: Oxidation and reduction can be defined in terms of electron transfer, change in oxidation state, loss/gain of oxygen, or loss/gain of hydrogen.
- R5.1.2: Oxidation states can be determined from the formula of a compound or ion using a set of rules.
- R5.1.3: In a redox reaction, the oxidizing agent is reduced and the reducing agent is oxidized.
- R5.1.4: Half-equations show the separate oxidation and reduction reactions occurring in a redox process.
- R5.1.5: Voltaic (galvanic) cells convert chemical energy to electrical energy through spontaneous redox reactions.
- R5.1.6: Electrolytic cells convert electrical energy to chemical energy through non-spontaneous redox reactions.
- R5.1.7: The products of electrolysis of aqueous solutions depend on the nature of the ions present and their relative ease of discharge.
🔄 Oxidation & Reduction
OIL RIG
- Oxidation Is Loss (of electrons). Increase in oxidation state.
- Reduction Is Gain (of electrons). Decrease in oxidation state.
Oxidation and reduction ALWAYS occur together — you cannot have one without the other. That is why they are called redox reactions.
Multiple Definitions of Oxidation
| Definition | Oxidation | Reduction |
|---|---|---|
| Electron transfer | Loss of electrons | Gain of electrons |
| Oxidation state | Increase in oxidation state | Decrease in oxidation state |
| Oxygen | Gain of oxygen | Loss of oxygen |
| Hydrogen | Loss of hydrogen | Gain of hydrogen |
Oxidizing and Reducing Agents
Oxidizing Agent
Accepts electrons from another species.
Is itself reduced (gains electrons).
Examples: $KMnO_4$, $K_2Cr_2O_7$, $Cl_2$, $O_2$, $HNO_3$ (conc.)
Reducing Agent
Donates electrons to another species.
Is itself oxidized (loses electrons).
Examples: Metals (Zn, Mg, Fe), $C$, $CO$, $H_2$, $SO_2$
Key exam phrasing: "The oxidizing agent is reduced" sounds confusing, but it's logical: the oxidizing agent causes oxidation in something else, while it itself undergoes reduction (gains electrons). Think: it takes electrons away from the other species.
📊 Oxidation State Rules
Rules for Assigning Oxidation States
Oxidation states (numbers) are assigned using these rules, in order of priority:
| Rule | Oxidation State | Example |
|---|---|---|
| Free/uncombined elements | 0 | $Na(s)$, $O_2(g)$, $Fe(s)$ |
| Monatomic ions | = charge | $Na^+ = +1$, $Cl^- = -1$, $Fe^{3+} = +3$ |
| Fluorine (in compounds) | −1 always | $NaF$, $OF_2$ |
| Oxygen (in compounds) | −2 (except peroxides: −1) | $H_2O$, $Na_2O_2$ (peroxide) |
| Hydrogen (in compounds) | +1 (except metal hydrides: −1) | $H_2O$, $NaH$ (hydride) |
| Sum in neutral compound | = 0 | $H_2SO_4$: $2(+1) + x + 4(-2) = 0$ → $x = +6$ |
| Sum in polyatomic ion | = charge of ion | $Cr_2O_7^{2-}$: $2x + 7(-2) = -2$ → $x = +6$ |
⚠️ Roman numerals: When a metal has a Roman numeral in its name, that IS the oxidation state. Iron(III) oxide = $Fe_2O_3$ where Fe = +3. Copper(II) sulfate = $CuSO_4$ where Cu = +2.
⚗️ Half-Equations
Writing Half-Equations
A half-equation shows either the oxidation or the reduction part of a redox reaction. The two half-equations can be combined to give the overall equation.
Balancing Steps
- Write the unbalanced half-equation (atoms that change oxidation state)
- Balance atoms other than O and H
- Balance O by adding $H_2O$
- Balance H by adding $H^+$ (in acidic solution)
- Balance charge by adding electrons ($e^-$)
Common Half-Equations You Must Know
| Half-Equation | Type |
|---|---|
| $Zn(s) \rightarrow Zn^{2+}(aq) + 2e^-$ | Oxidation |
| $Cu^{2+}(aq) + 2e^- \rightarrow Cu(s)$ | Reduction |
| $2Cl^-(aq) \rightarrow Cl_2(g) + 2e^-$ | Oxidation |
| $2H^+(aq) + 2e^- \rightarrow H_2(g)$ | Reduction |
| $2H_2O(l) \rightarrow O_2(g) + 4H^+(aq) + 4e^-$ | Oxidation |
| $MnO_4^-(aq) + 8H^+(aq) + 5e^- \rightarrow Mn^{2+}(aq) + 4H_2O(l)$ | Reduction |
Combining half-equations: The number of electrons lost in oxidation must equal the number gained in reduction. Multiply half-equations as needed to equalize electrons, then add and cancel $e^-$ from both sides.
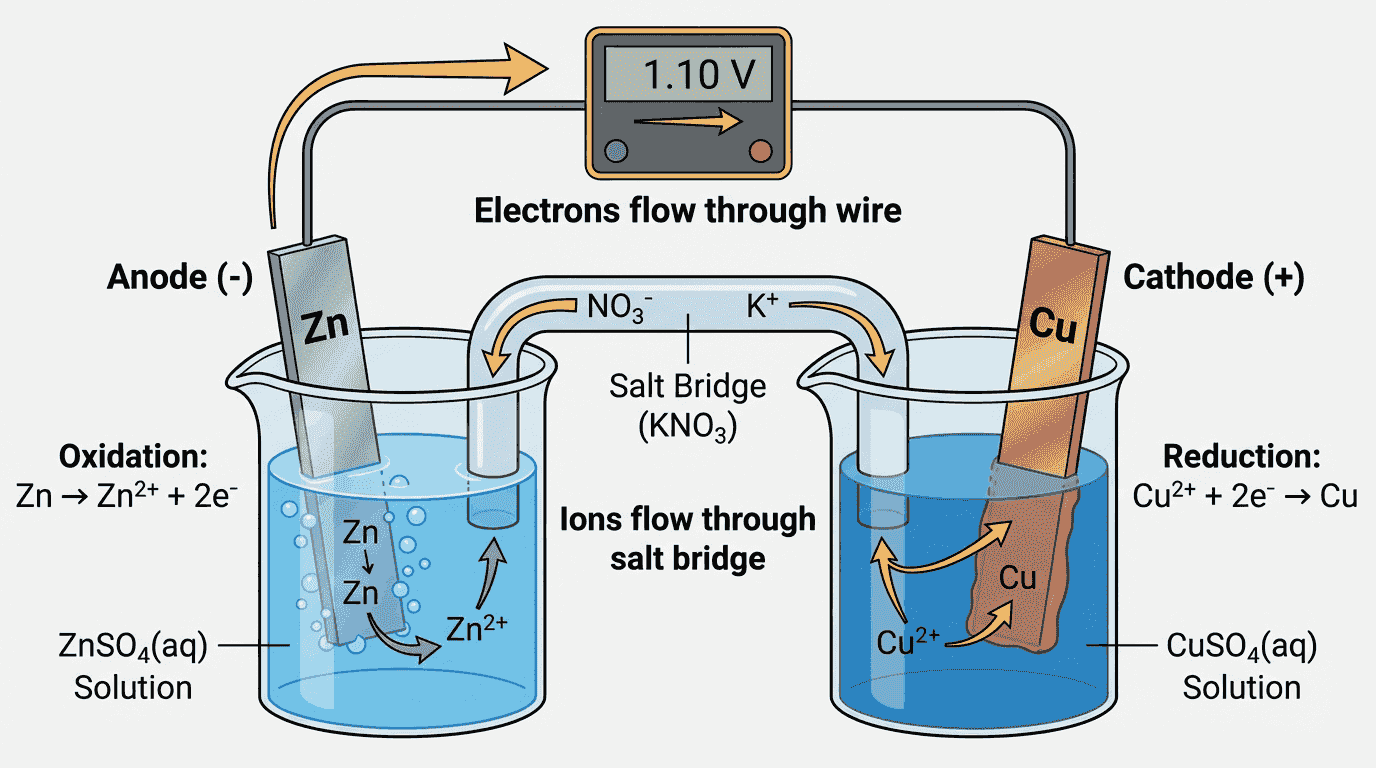
🔋 Voltaic (Galvanic) Cells
Key Definition
A voltaic cell converts chemical energy into electrical energy through a spontaneous redox reaction ($\Delta G < 0$). The two half-cells are connected by a wire (external circuit) and a salt bridge (internal circuit).
The Daniell Cell (Zn–Cu)
- Anode (−): Oxidation: $Zn(s) \rightarrow Zn^{2+}(aq) + 2e^-$. Zinc electrode dissolves (mass decreases).
- Cathode (+): Reduction: $Cu^{2+}(aq) + 2e^- \rightarrow Cu(s)$. Copper deposits on electrode (mass increases).
- Electron flow: From anode (Zn) → through wire → to cathode (Cu)
- Salt bridge: Contains $KNO_3$ or $NaCl$ in gel. Allows ions to flow between half-cells to maintain electrical neutrality. Without it, charge would build up and the reaction would stop.
- Cell voltage: ~1.10 V for the Zn–Cu cell at standard conditions
Experiment 1: The Daniell Cell
AvailableBuild a Zn-Cu voltaic cell. Observe electron flow and voltage generation.
→ Launch Simulation⚡ Electrolytic Cells
Key Definition
An electrolytic cell converts electrical energy into chemical energy. It uses an external power source (battery) to force a non-spontaneous redox reaction ($\Delta G > 0$).
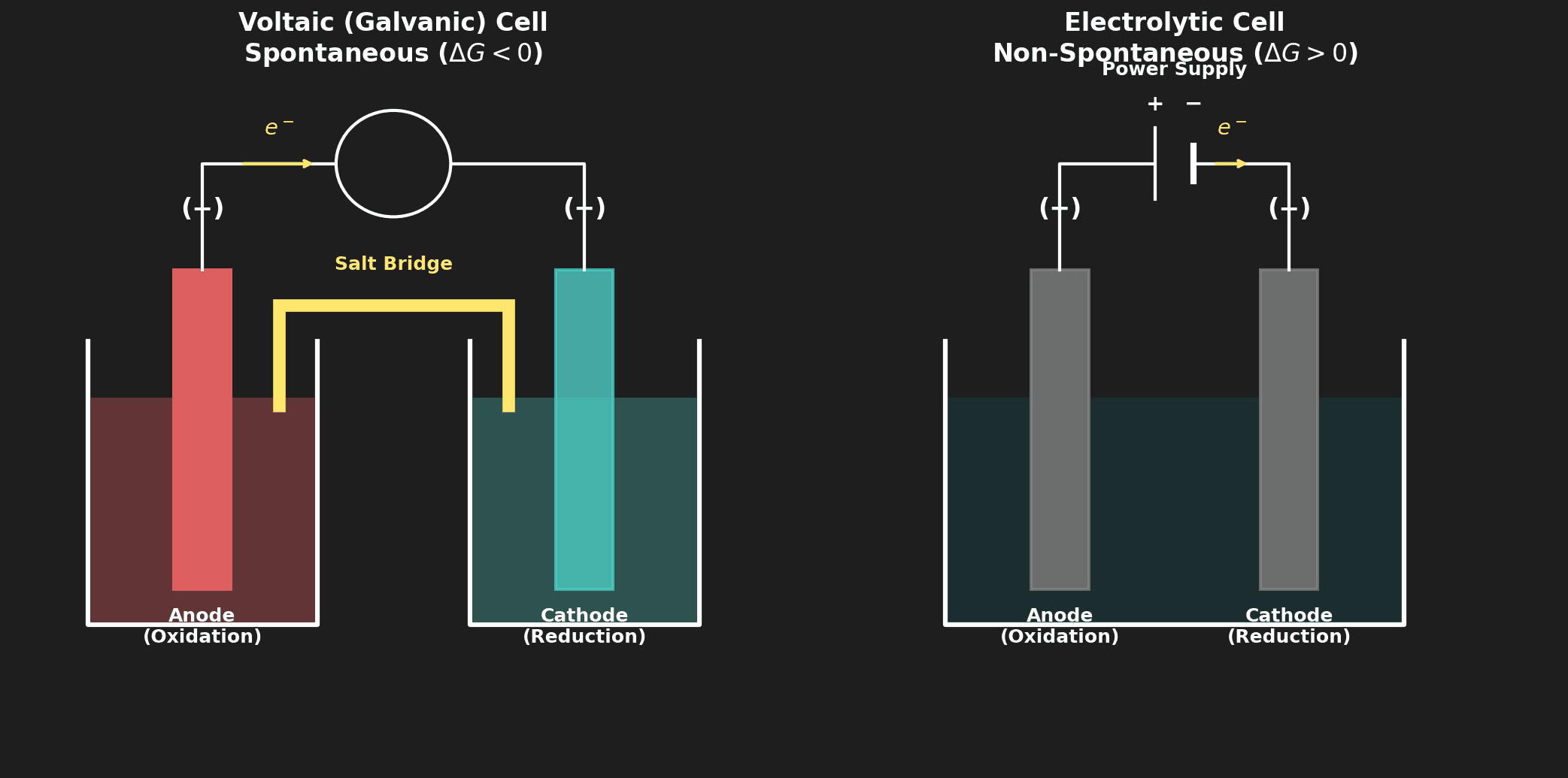
Voltaic vs Electrolytic — Complete Comparison
| Feature | Voltaic Cell | Electrolytic Cell |
|---|---|---|
| Energy conversion | Chemical → Electrical | Electrical → Chemical |
| Spontaneity | Spontaneous ($\Delta G < 0$) | Non-spontaneous ($\Delta G > 0$) |
| External energy | Not required (generates electricity) | Required (battery/power supply) |
| Anode (oxidation) | Negative (−) | Positive (+) |
| Cathode (reduction) | Positive (+) | Negative (−) |
| Salt bridge | Yes (two separate half-cells) | No (one container with electrolyte) |
| Electron flow | Anode → Cathode (through wire) | Anode → Cathode (through wire) |
| Example | Batteries, fuel cells | Electroplating, aluminium extraction |
⚠️ Critical fact: In BOTH cell types, oxidation always occurs at the anode and reduction always occurs at the cathode. What changes is the sign (+/−) of each electrode.
Electrolysis of Aqueous Solutions
When electrolyzing aqueous solutions (not molten), water competes with the dissolved ions for discharge. The product depends on which species is more easily discharged:
| At Cathode (−) | At Anode (+) |
|---|---|
| Less reactive metals are preferentially discharged: $Cu^{2+} + 2e^- \rightarrow Cu$ | Halide ions are preferentially discharged (if concentrated): $2Cl^- \rightarrow Cl_2 + 2e^-$ |
| If metal is very reactive (Na⁺, K⁺, Al³⁺): water is reduced instead: $2H_2O + 2e^- \rightarrow H_2 + 2OH^-$ | If no halide (or dilute halide): water is oxidized: $2H_2O \rightarrow O_2 + 4H^+ + 4e^-$ |
🧪 Interactive Virtual Labs
Experiment 2: Aqueous Electrolysis
AvailablePredict products at electrodes for aqueous solutions ($NaCl$, $CuSO_4$). Consider selective discharge rules.
→ Launch SimulationExperiment 3: Electroplating
AvailableUse electrolysis to coat a metal object with copper or silver. Observe mass changes at electrodes.
→ Launch Simulation🧠 Memory Aids & Mnemonics
🔤 OIL RIG
Oxidation Is Loss, Reduction Is Gain (of electrons)
The most important mnemonic in all of chemistry. If a species loses electrons, it is oxidized. If it gains electrons, it is reduced.
🔤 AN OX and RED CAT
Anode = Oxidation, Reduction at Cathode
This works in BOTH voltaic and electrolytic cells. Oxidation always at the anode, reduction always at the cathode. What changes between cell types is only the charge (+/−) of each electrode.
🔤 Voltaic Charge Signs
"Voltaic = VAN" → Voltaic Anode Negative
In a voltaic cell, the anode is negative. Opposite in electrolytic: anode is positive. Remember: VAN drives electricity OUT (spontaneous).
🔤 Agent Confusion Buster
"The agent does the OPPOSITE of what it's called"
Oxidizing agent → is itself reduced. Reducing agent → is itself oxidized. The agent causes the named process in the OTHER species.
🔤 Oxidation State of Oxygen
"Oxygen is −2 unless it's F₂O (then −1 in peroxides, +2 in OF₂)"
Fluorine beats oxygen. In peroxides ($H_2O_2$, $Na_2O_2$), oxygen is −1. In $OF_2$, oxygen is +2 because F is always −1.
🌍 Real-World Applications
🔋 Lithium-Ion Batteries
Context: Your phone, laptop, and electric car all run on lithium-ion batteries — rechargeable voltaic cells that power modern life.
Science: During discharge (use), the lithium-ion battery works as a voltaic cell: lithium is oxidized at the anode ($Li \rightarrow Li^+ + e^-$) and cobalt oxide is reduced at the cathode ($CoO_2 + Li^+ + e^- \rightarrow LiCoO_2$). The electrons flow through the external circuit (your device). During charging, an external power source reverses the process — the cell acts as an electrolytic cell, forcing lithium ions back to the anode.
Impact: Lithium's small atomic radius and low density give Li-ion batteries the highest energy density of commercial rechargeable batteries. This technology enables electric vehicles, grid-scale energy storage, and portable electronics. The 2019 Nobel Prize in Chemistry was awarded for developing lithium-ion batteries.
🏗️ Corrosion and Rusting
Context: Rusting costs the global economy over $2.5 trillion per year. It is an electrochemical process — a mini voltaic cell on the surface of iron.
Science: Iron is oxidized at anodic sites: $Fe(s) \rightarrow Fe^{2+}(aq) + 2e^-$. At cathodic sites (where water and oxygen are present): $O_2(g) + 2H_2O(l) + 4e^- \rightarrow 4OH^-(aq)$. The $Fe^{2+}$ and $OH^-$ combine and further oxidize to form hydrated iron(III) oxide — rust ($Fe_2O_3 \cdot xH_2O$). Prevention methods all work by disrupting this electrochemical process: painting (barrier), galvanizing (sacrificial protection — Zn is more reactive, so it oxidizes instead of Fe), stainless steel (Cr forms a protective oxide layer).
Impact: Engineers must consider redox chemistry when designing bridges, ships, pipelines, and reinforced concrete. Cathodic protection (attaching a more reactive "sacrificial" metal) is used on oil platforms and ship hulls.
🧹 Aluminium Extraction (Hall-Héroult Process)
Context: Aluminium is the most abundant metal in Earth's crust, but extracting it from its ore ($Al_2O_3$, bauxite) requires electrolysis because Al³⁺ is too reactive to reduce with carbon.
Science: The ore is dissolved in molten cryolite ($Na_3AlF_6$) to lower the melting point from 2072°C to ~960°C. At the cathode (carbon-lined steel tank): $Al^{3+} + 3e^- \rightarrow Al(l)$. At the anode (carbon electrodes): $2O^{2-} \rightarrow O_2(g) + 4e^-$. The $O_2$ produced reacts with the carbon anode, consuming it: $C + O_2 \rightarrow CO_2$. This means the anodes must be replaced regularly.
Impact: Aluminium production consumes about 3% of global electricity. Recycling aluminium uses only 5% of the energy needed for primary extraction — making it one of the most energy-efficient materials to recycle. Every recycled can saves enough electricity to power a TV for 3 hours.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Getting the anode/cathode charge wrong: In voltaic cells, the anode is negative (−). In electrolytic cells, the anode is positive (+). The REACTION doesn't change (oxidation is always at the anode), but the SIGN flips between cell types.
- Confusing oxidizing and reducing agents: The oxidizing agent is the species that gets reduced (it takes electrons from something else, oxidizing it). Students often reverse this.
- Unbalanced half-equations: Check that: (1) atoms are balanced, (2) charge is balanced, (3) electrons appear on the correct side (products for oxidation, reactants for reduction).
- Forgetting the salt bridge function: It doesn't carry electrons — it allows ions to flow between half-cells. Without it, charge builds up and the cell stops. Electrons flow through the WIRE, ions flow through the SALT BRIDGE.
- Ignoring selective discharge in aqueous electrolysis: When electrolyzing $NaCl(aq)$, Na⁺ is NOT discharged at the cathode — water is reduced instead (producing $H_2$), because Na⁺ is too reactive. Similarly, dilute sulfate solutions produce $O_2$ at the anode (water oxidized), not $SO_4^{2-}$.
- Wrong sign for oxidation state changes: If an element goes from 0 to +2, it has been oxidized (increase). If it goes from +7 to +2, it has been reduced (decrease). Students confuse the direction.
📝 IB-Style Exam Questions
Question 1: State what happens at the negative electrode during the electrolysis of molten sodium chloride. [1 mark]
Mark Scheme:
- [1 mark] Reduction of sodium ions: $Na^+ + e^- \rightarrow Na(l)$. (In electrolysis, the negative electrode is the cathode.)
Question 2: Determine the oxidation state of Chromium in the dichromate ion, $Cr_2O_7^{2-}$. [1 mark]
Mark Scheme:
- [1 mark] +6. ($2x + 7(-2) = -2 \rightarrow 2x - 14 = -2 \rightarrow 2x = 12 \rightarrow x = +6$).
Question 3: State the function of the salt bridge in a voltaic cell. [1 mark]
Mark Scheme:
- [1 mark] To complete the electrical circuit by allowing the flow of ions between the two half-cells, maintaining electrical neutrality.
Question 4: Deduce the half-equation for the oxidation of iodide ions to iodine. [1 mark]
Mark Scheme:
- [1 mark] $2I^-(aq) \rightarrow I_2(s) + 2e^-$
Question 5: Compare the anode in a voltaic cell vs an electrolytic cell in terms of charge and reaction type. [2 marks]
Mark Scheme:
- [1 mark] Voltaic anode: negative (−), oxidation occurs.
- [1 mark] Electrolytic anode: positive (+), oxidation occurs. (Both undergo oxidation — only the sign differs.)
Question 6: Predict the products at each electrode during the electrolysis of concentrated aqueous sodium chloride ($NaCl$). [2 marks]
Mark Scheme:
- [1 mark] Cathode (−): Hydrogen gas ($H_2$). Water is reduced because Na⁺ is too reactive: $2H_2O + 2e^- \rightarrow H_2 + 2OH^-$.
- [1 mark] Anode (+): Chlorine gas ($Cl_2$). Concentrated $Cl^-$ is preferentially discharged: $2Cl^- \rightarrow Cl_2 + 2e^-$.
Question 7: Identify the oxidizing and reducing agents in the reaction: $Zn(s) + Cu^{2+}(aq) \rightarrow Zn^{2+}(aq) + Cu(s)$. [2 marks]
Mark Scheme:
- [1 mark] $Cu^{2+}$ is the oxidizing agent (it is reduced: gains electrons, going from +2 to 0).
- [1 mark] $Zn$ is the reducing agent (it is oxidized: loses electrons, going from 0 to +2).
Question 8: Explain why the carbon anodes need to be replaced regularly during the electrolysis of aluminium oxide. [2 marks]
Mark Scheme:
- [1 mark] At the anode, oxide ions are oxidized to produce oxygen gas: $2O^{2-} \rightarrow O_2 + 4e^-$.
- [1 mark] The oxygen produced at high temperature reacts with the carbon anode: $C + O_2 \rightarrow CO_2$, causing the anode to gradually burn away/erode.