📋 IB Content Statements (R5.2)
This topic covers the following HL syllabus points from the IB Chemistry 2025 guide:
- R5.2.1 (HL): Standard electrode potentials ($E^\theta$) are measured relative to the standard hydrogen electrode (SHE) under standard conditions (298 K, 100 kPa, 1.0 mol dm⁻³).
- R5.2.2 (HL): In the electrochemical series, species with more positive $E^\theta$ values are stronger oxidizing agents; those with more negative $E^\theta$ values are stronger reducing agents.
- R5.2.3 (HL): Standard cell potential: $E^\theta_{cell} = E^\theta_{cathode} - E^\theta_{anode}$. A positive $E^\theta_{cell}$ indicates a spontaneous reaction.
- R5.2.4 (HL): The relationship between $\Delta G^\theta$ and $E^\theta_{cell}$: $\Delta G^\theta = -nFE^\theta$.
- R5.2.5 (HL): Non-standard conditions: actual cell potential can differ from $E^\theta$ if concentrations, temperatures, or pressures are not standard.
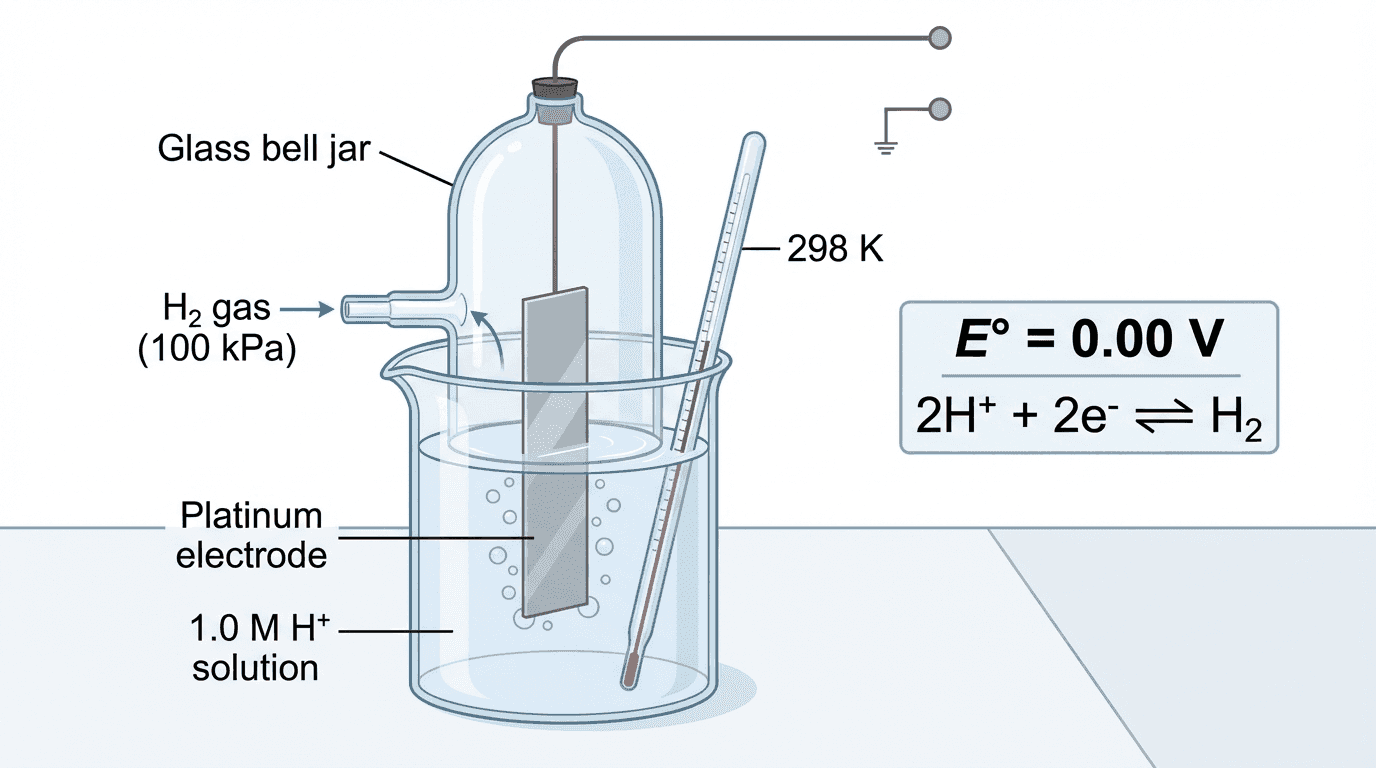
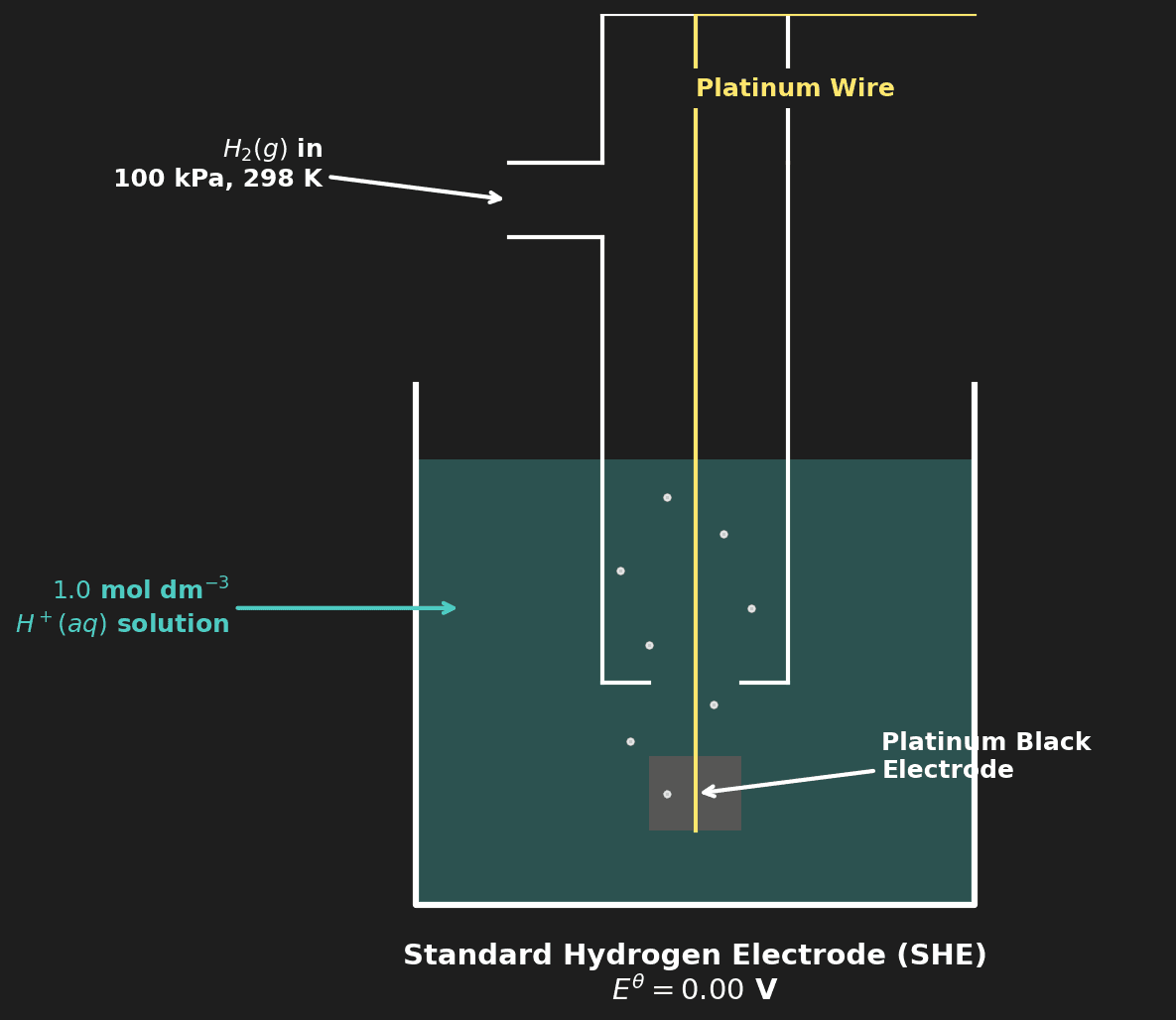
⚖️ Standard Hydrogen Electrode (SHE)
The Reference Standard
All standard electrode potentials are measured relative to the SHE, which is assigned a value of exactly 0.00 V.
Standard Conditions
| Parameter | Standard Value |
|---|---|
| Temperature | 298 K (25°C) |
| Pressure | 100 kPa |
| Concentration of $H^+(aq)$ | $1.0$ mol dm⁻³ |
| Electrode Material | Platinum (inert, provides surface for reaction) |
How It Works
The half-reaction: $2H^+(aq) + 2e^- \rightleftharpoons H_2(g)$ $E^\theta = 0.00$ V
Hydrogen gas is bubbled over a platinum electrode immersed in 1.0 mol dm⁻³ $H^+$ solution. The platinum provides a surface for the equilibrium between $H^+$ and $H_2$.
📉 Electrochemical Series
Reading the Data Booklet
The electrochemical series lists standard reduction potentials ($E^\theta$) for half-reactions written as reductions.
| Position in Series | $E^\theta$ Value | As Oxidizing Agent | As Reducing Agent |
|---|---|---|---|
| Top (e.g., $F_2$, $MnO_4^-$) | Most positive | Strongest (most easily reduced) | Weakest |
| Middle (e.g., $H^+/H_2$) | ~0.00 V | Moderate | Moderate |
| Bottom (e.g., $Li^+$, $K^+$) | Most negative | Weakest | Strongest (most easily oxidized) |
Key Examples
| Half-Reaction (Reduction) | $E^\theta$ / V |
|---|---|
| $F_2 + 2e^- \rightleftharpoons 2F^-$ | +2.87 |
| $Ag^+ + e^- \rightleftharpoons Ag$ | +0.80 |
| $Cu^{2+} + 2e^- \rightleftharpoons Cu$ | +0.34 |
| $2H^+ + 2e^- \rightleftharpoons H_2$ | 0.00 |
| $Zn^{2+} + 2e^- \rightleftharpoons Zn$ | −0.76 |
| $Li^+ + e^- \rightleftharpoons Li$ | −3.04 |
Activity Series Simulation
AvailableTest metals against solutions to determine the Activity Series experimentally.
→ Launch Simulation⚡ Calculating Cell Potential ($E^\theta_{cell}$)
The Formula
$$E^\theta_{cell} = E^\theta_{cathode} - E^\theta_{anode}$$
Where:
- Cathode = where reduction occurs (higher $E^\theta$ value)
- Anode = where oxidation occurs (lower $E^\theta$ value)
Worked Example: Daniell Cell
Calculate $E^\theta_{cell}$ for a cell made of $Zn/Zn^{2+}$ ($E^\theta = -0.76$ V) and $Cu/Cu^{2+}$ ($E^\theta = +0.34$ V).
Step 1: Identify cathode (higher $E^\theta$): $Cu^{2+}/Cu$ (+0.34 V)
Step 2: Identify anode (lower $E^\theta$): $Zn/Zn^{2+}$ (−0.76 V)
Step 3: $E^\theta_{cell} = (+0.34) - (-0.76) = +1.10$ V ✓
Spontaneity Rule: If $E^\theta_{cell} > 0$ → the reaction is spontaneous (thermodynamically feasible). If $E^\theta_{cell} < 0$ → the reaction is non-spontaneous.
Predicting Reactions
An oxidizing agent (top of the series) will react with a reducing agent (bottom of the series). The "anti-clockwise" rule: draw a line from the reduction half-reaction (top-right) to the oxidation half-reaction (bottom-left). If this line goes anti-clockwise through the table, the reaction is spontaneous.
🔗 Spontaneity & $\Delta G$
The Relationship
$$\Delta G^\theta = -nFE^\theta_{cell}$$
- $n$ = moles of electrons transferred
- $F$ = Faraday's constant = 96 485 C mol⁻¹
- $E^\theta_{cell}$ = standard cell potential (V)
| $E^\theta_{cell}$ | $\Delta G^\theta$ | Reaction |
|---|---|---|
| Positive (+) | Negative (−) | Spontaneous (voltaic/galvanic cell) |
| Negative (−) | Positive (+) | Non-spontaneous (electrolytic cell) |
Connecting the Three Thermodynamic Quantities
The three key relationships can be combined:
$$\Delta G^\theta = -nFE^\theta = -RT\ln K$$
This means you can calculate $K$ from $E^\theta$ (and vice versa):
$$E^\theta_{cell} = \frac{RT}{nF}\ln K$$
🧠 Memory Aids
🔤 Cell Potential Formula — "Cathode Minus Anode" (CMA)
$E^\theta_{cell} = E^\theta_{cathode} - E^\theta_{anode}$. Simple subtraction. The cathode always has the higher $E^\theta$ value. No need to reverse signs or multiply — just subtract directly using data booklet values.
🔤 Identifying Cathode/Anode — "OIL RIG at AN OX RED CAT"
OIL RIG: Oxidation Is Loss, Reduction Is Gain (of electrons). AN OX: Oxidation at the ANode. RED CAT: REDuction at the CAThode.
🔤 Spontaneity — "Positive E, Negative G = Go"
If $E^\theta_{cell}$ is positive, then $\Delta G^\theta$ is negative (because of the minus sign in $\Delta G = -nFE$). Negative $\Delta G$ = spontaneous.
🔤 Electrochemical Series — "Positive = Grab"
Species with more positive $E^\theta$ are better at grabbing electrons (stronger oxidizing agents). Species with more negative $E^\theta$ are better at giving away electrons (stronger reducing agents).
🌍 Real-World Applications
🔋 Lithium-Ion Batteries
Context: $Li^+/Li$ has $E^\theta = -3.04$ V, the most negative value in the electrochemical series.
Science: Lithium is the strongest reducing agent. When paired with a cathode material like $CoO_2$ ($E^\theta ≈ +0.6$ V), the resulting cell voltage is $E_{cell} ≈ 0.6 - (-3.04) = 3.64$ V — one of the highest voltages per cell. Li-ion batteries also benefit from lithium's very low molar mass (6.94 g/mol), giving the highest energy per gram.
Impact: Li-ion batteries power smartphones, laptops, electric vehicles, and grid storage — chosen specifically because of lithium's extreme position in the electrochemical series.
🏗️ Cathodic Protection of Pipelines
Context: Zinc ($E^\theta = -0.76$ V) is a stronger reducing agent than iron ($E^\theta = -0.44$ V).
Science: Blocks of zinc (sacrificial anodes) are attached to iron pipelines or ship hulls. Zinc is oxidized preferentially ($Zn → Zn^{2+} + 2e^-$) because it has a more negative $E^\theta$. The electrons flow to the iron, keeping it in the reduced (metallic) state — preventing rust.
Impact: This saves billions in corrosion damage annually. The same principle is used in galvanizing (coating iron with zinc).
🏥 Fuel Cells — Hydrogen Economy
Context: The hydrogen fuel cell uses $H_2/H^+$ ($E^\theta = 0.00$ V) and $O_2/H_2O$ ($E^\theta = +1.23$ V).
Science: $E^\theta_{cell} = 1.23 - 0.00 = +1.23$ V. Overall: $2H_2 + O_2 → 2H_2O$. $\Delta G^\theta = -nFE = -(4)(96485)(1.23) = -474.7$ kJ/mol. This large negative $\Delta G$ confirms the reaction is highly spontaneous. The only byproduct is water.
Impact: Fuel cells are used in space shuttles, buses, and backup power. Toyota Mirai and Hyundai Nexo are commercial fuel cell vehicles.
⚠️ Common Mistakes
- ❌ Reversing the $E^\theta$ sign when writing oxidation → ✅ Use the formula $E_{cell} = E_{cathode} - E_{anode}$ with both values as given in the data booklet (reduction potentials). You do NOT need to flip the sign of the anode value — the subtraction handles it.
- ❌ "More negative $E^\theta$ = stronger oxidizing agent" → ✅ It's the opposite. More positive $E^\theta$ = stronger oxidizing agent. More negative $E^\theta$ = stronger reducing agent.
- ❌ Multiplying $E^\theta$ by stoichiometric coefficients → ✅ Electrode potentials are intensive properties — they do NOT depend on the amount of substance. Never multiply $E^\theta$ by 2, 3, etc.
- ❌ Forgetting units in $\Delta G = -nFE$ calculation → ✅ $F = 96485$ C/mol, $E$ is in V, so $\Delta G$ comes out in joules. Convert to kJ by dividing by 1000.
- ❌ Confusing positive $E_{cell}$ with negative $\Delta G$ → ✅ They are opposite signs because of the minus in $\Delta G = -nFE$. Positive $E$ → negative $\Delta G$ → spontaneous.
📝 Exam-Style Questions
Question 1: Calculate $E^\theta_{cell}$ for a cell made of $Zn/Zn^{2+}$ ($-0.76$ V) and $Cu/Cu^{2+}$ ($+0.34$ V). [2 marks]
Mark Scheme:
- [1 mark] Cathode: Cu (higher $E^\theta$, +0.34 V). Anode: Zn (lower $E^\theta$, −0.76 V).
- [1 mark] $E^\theta_{cell} = (+0.34) - (-0.76) = +1.10$ V.
Question 2: Explain why $Cu + ZnSO_4$ does not react spontaneously. [2 marks]
Mark Scheme:
- [1 mark] Zn is a stronger reducing agent than Cu (more negative $E^\theta$). Cu cannot displace Zn from solution.
- [1 mark] $E^\theta_{cell}$ would be negative ($(−0.76) − (+0.34) = −1.10$ V), so $\Delta G > 0$ (non-spontaneous).
Question 3: Calculate $\Delta G^\theta$ for a cell with $E^\theta_{cell} = +1.10$ V and $n = 2$. [2 marks]
Mark Scheme:
- [1 mark] $\Delta G^\theta = -nFE^\theta = -(2)(96485)(1.10)$.
- [1 mark] $= -212\,267$ J $= \mathbf{-212.3}$ kJ mol⁻¹.
Question 4: State the conditions required for the Standard Hydrogen Electrode. [2 marks]
Mark Scheme:
- [1 mark] 100 kPa $H_2$ gas, 298 K temperature.
- [1 mark] 1.0 mol dm⁻³ $H^+(aq)$, inert platinum electrode.
Question 5: Identify the strongest oxidizing agent: $Ag^+$ (+0.80 V), $Cu^{2+}$ (+0.34 V), $Zn^{2+}$ (−0.76 V). [1 mark]
Mark Scheme:
- [1 mark] $Ag^+$ (most positive $E^\theta$ = strongest tendency to gain electrons).
Question 6: Explain why electrode potentials do not change when the half-equation is multiplied by a coefficient. [1 mark]
Mark Scheme:
- [1 mark] Electrode potential is an intensive property — it measures the tendency of the reaction to occur, not the amount. Doubling the equation doubles both $n$ and the charge transferred, but the voltage (energy per coulomb) remains the same.
Question 7: Predict whether $Fe^{3+}$ (+0.77 V) can oxidize $I^-$ (+0.54 V) under standard conditions. [2 marks]
Mark Scheme:
- [1 mark] $E^\theta_{cell} = E^\theta(Fe^{3+}) - E^\theta(I_2/I^-) = 0.77 - 0.54 = +0.23$ V.
- [1 mark] Positive $E^\theta_{cell}$, so the reaction is spontaneous. $Fe^{3+}$ can oxidize $I^-$ to $I_2$.
Question 8: Calculate the equilibrium constant $K$ for a cell with $E^\theta_{cell} = +0.46$ V at 298 K ($n = 2$). [2 marks]
Mark Scheme:
- [1 mark] $\ln K = \frac{nFE^\theta}{RT} = \frac{(2)(96485)(0.46)}{(8.314)(298)} = \frac{88766}{2477.6} = 35.83$.
- [1 mark] $K = e^{35.83} = \mathbf{3.7 \times 10^{15}}$.