🧮 Index of Hydrogen Deficiency (IHD)
Formula
$$ IHD = \frac{2C + 2 + N - H - X}{2} $$
Also known as "Degree of Unsaturation". Determines rings (1) or pi bonds (Double=1, Triple=2).
🌈 Infrared Spectroscopy (IR)
Identifies Bonds (Functional Groups) based on vibrational frequency.
Key Absorptions (wavenumbers $cm^{-1}$):
- O-H (Alcohol): 3200-3600 (Broad, Strong)
- C=O (Carbonyl): 1700-1750 (Sharp, Strong)
- C=C (Alkene): 1620-1680 (Variable)
⚖️ Mass Spectrometry (MS)
Identifies Molecular Mass ($M^+$) and fragments.
- Molecular Ion ($M^+$): Peak with highest m/z value (represents $M_r$).
- Fragmentation: Breaking of bonds. e.g., Loss of $CH_3$ ($M-15$).
🧲 Proton NMR ($^1H$-NMR)
Provides information on the Chemical Environment of hydrogen atoms.
Chemical Shift ($\delta$)
Position of peak. Indicates environment (e.g., near electronegative atom).
Integration (Area)
Ratio of protons in that specific environment.
HL
Splitting Patterns (Spin-Spin Coupling)
n+1 Rule: Peak splits into $n+1$ sub-peaks if there are $n$ adjacent protons.
- Singlet (0 adjacent)
- Doublet (1 adjacent)
- Triplet (2 adjacent)
- Quartet (3 adjacent)
📝 Exam-Style Questions
Question 1: An organic compound has the molecular formula $C_4H_8O$. Calculate its Index of Hydrogen Deficiency (IHD). [1 mark]
Mark Scheme:
- [1 mark] $IHD = \frac{2(4) + 2 - 8}{2} = \frac{10 - 8}{2} = 1$. (Implies 1 double bond or 1 ring).
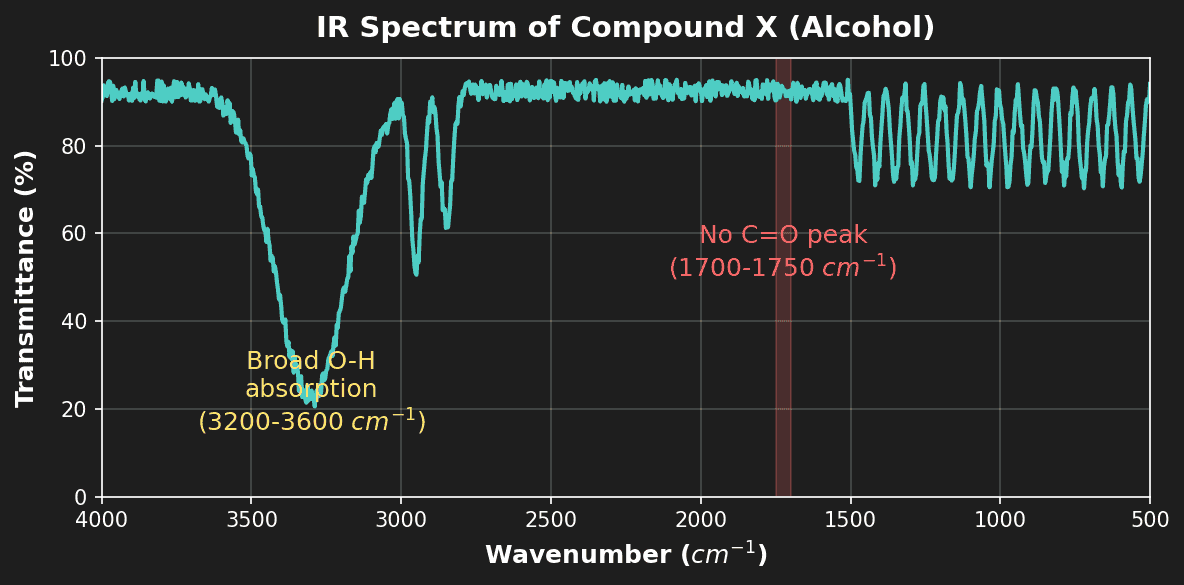
Question 2: The IR spectrum of compound X shows a broad absorption at $3200-3600 \ cm^{-1}$ and no peak at $1700-1750 \ cm^{-1}$. Deduce the functional group present. [1 mark]
Mark Scheme:
- [1 mark] Alcohol (-OH group). (Broad peak is characteristic of H-bonded OH; absence of C=O confirms it's not a carboxylic acid).
Question 3: A mass spectrum shows a molecular ion peak at $m/z = 74$. A major fragment peak appears at $m/z = 59$. Identify the fragment lost. [1 mark]
Mark Scheme:
- [1 mark] Mass lost = $74 - 59 = 15$. Loss of a methyl group ($-CH_3$).
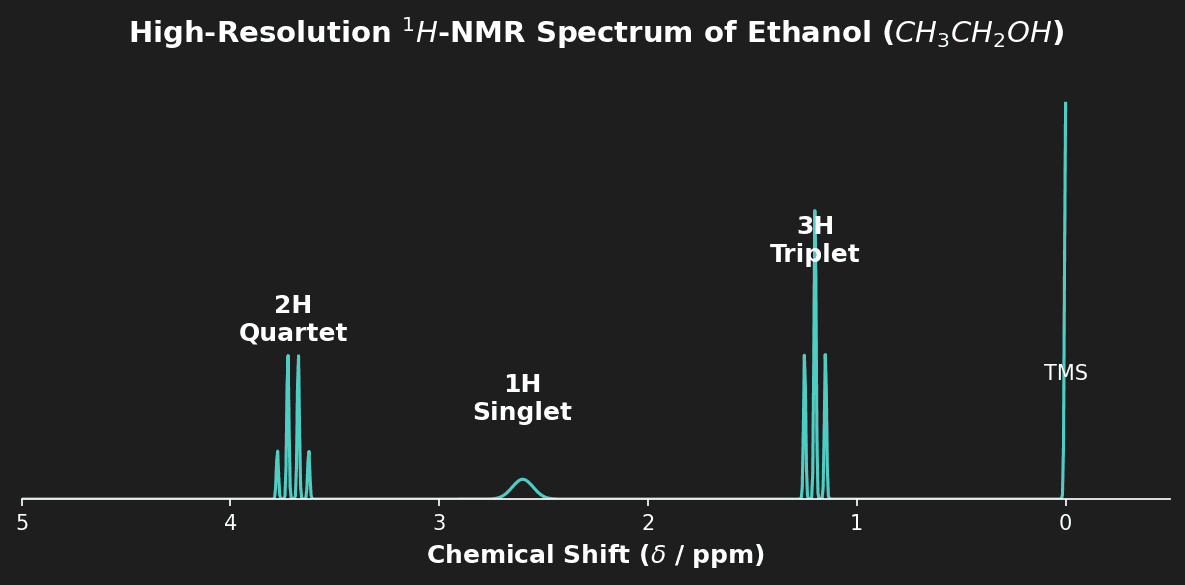
Question 4: Predict the number of signals and the splitting pattern for the $CH_3$ group in Ethanol ($CH_3CH_2OH$) in a high-resolution $^1H$-NMR spectrum. [2 marks]
Mark Scheme:
- [1 mark] One signal (3 equivalent protons).
- [1 mark] Triplet (adjacent to $CH_2$ group, so $n=2$, $n+1=3$).
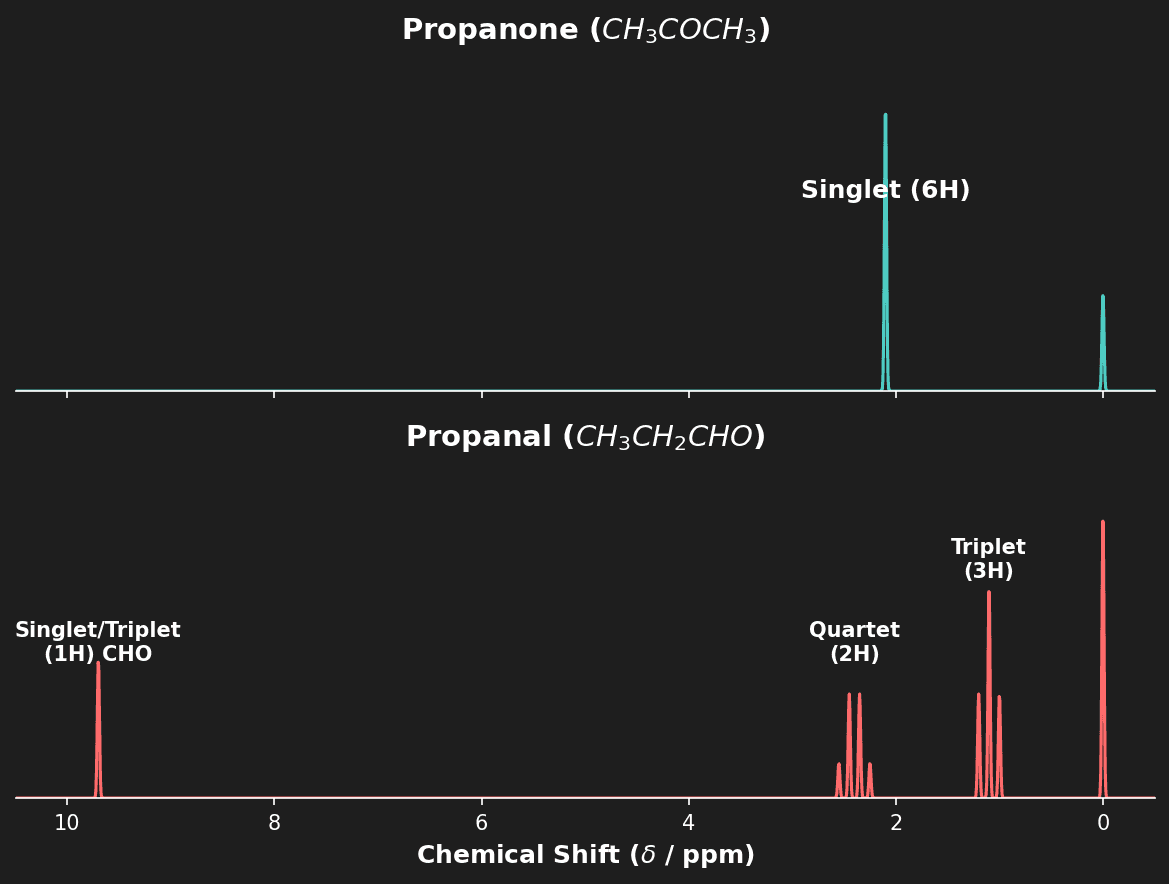
Question 5: Distinguish between the $^1H$-NMR spectra of Propanone ($CH_3COCH_3$) and Propanal ($CH_3CH_2CHO$). [2 marks]
Mark Scheme:
- [1 mark] Propanone: 1 signal (singlet) - all 6 protons are equivalent.
- [1 mark] Propanal: 3 signals (triplet $CH_3$, multiplet $CH_2$, triplet/singlet $CHO$). Distinctive aldehyde peak at $\delta = 9-10 \ ppm$.