📋 IB Content Statements (S1.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S1.1.1: Elements are the primary constituents of matter, which cannot be chemically broken down into simpler substances. Compounds consist of atoms of different elements chemically bonded together in a fixed ratio. Mixtures contain more than one element or compound in no fixed ratio, which are not chemically bonded and can be separated by physical methods.
- S1.1.2: The kinetic molecular theory is a model to explain the physical properties of matter (solids, liquids, and gases) and changes of state.
- S1.1.3: Temperature ($T$ in Kelvin, K) is a measure of the average kinetic energy ($E_k$) of particles.
Additional Syllabus Notes
- Separation and purification techniques: solvation, filtration, recrystallization, evaporation, distillation, and chromatography
- Distinction between homogeneous and heterogeneous mixtures
- Use of state symbols: (s), (l), (g), and (aq) in chemical equations
- Names of changes of state: melting, freezing, vaporization (evaporation and boiling), condensation, sublimation, and deposition
🔹 States of Matter
Key Definition: The Kinetic Particle Theory
All matter is made of particles in constant motion. The state of matter depends on the balance between the kinetic energy of the particles (tending to separate them) and the intermolecular forces (tending to pull them together).
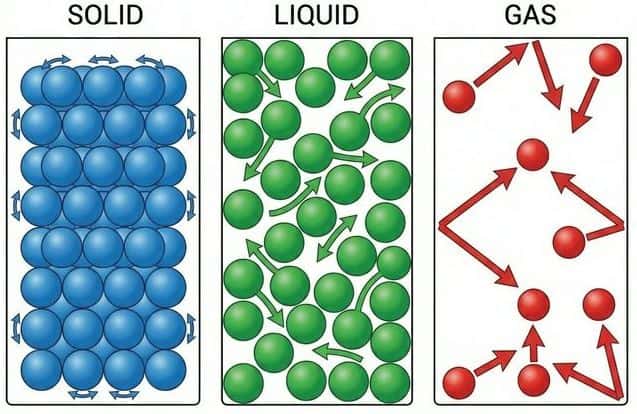
Properties of the Three States
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Particle arrangement | Regular lattice, closely packed | Close but disordered, no fixed positions | Very far apart, no pattern |
| Particle motion | Vibrate in fixed positions | Slide past each other (translational) | Rapid, random motion in all directions |
| Intermolecular forces | Strong (overcome by vibration only) | Moderate (particles can move but stay close) | Negligible (particles are independent) |
| Shape | Fixed | Takes shape of container | Fills container completely |
| Volume | Fixed | Fixed | Variable (compressible) |
| Density | High | High (slightly less than solid) | Very low (~1/1000 of liquid) |
| Compressibility | Almost incompressible | Almost incompressible | Highly compressible |
State Changes
Changes of state occur at constant temperature (melting point / boiling point) while energy is used to overcome intermolecular forces. During a phase change, temperature does not change — all energy goes into breaking or forming intermolecular bonds.
| Change of State | Transition | Energy | What Happens to IMFs |
|---|---|---|---|
| Melting | Solid → Liquid | Endothermic (absorbs) | Some IMFs broken; particles gain freedom to move |
| Boiling / Vaporization | Liquid → Gas | Endothermic (absorbs) | All remaining IMFs broken; particles escape |
| Evaporation | Liquid → Gas (surface only) | Endothermic | Highest-energy particles escape from surface |
| Freezing | Liquid → Solid | Exothermic (releases) | IMFs form; particles lock into lattice |
| Condensation | Gas → Liquid | Exothermic (releases) | IMFs re-form; particles slow and attract |
| Sublimation | Solid → Gas | Endothermic | All IMFs broken at once (e.g., dry ice, iodine) |
| Deposition | Gas → Solid | Exothermic | IMFs form directly from gas (e.g., frost) |
Evaporation vs Boiling: Evaporation occurs at the surface at any temperature (only the fastest particles escape). Boiling occurs throughout the liquid at a specific temperature (boiling point) when vapor pressure equals atmospheric pressure.
🧪 Kinetic Molecular Theory
📺 S1.1.2 — The Kinetic Molecular Theory
Core Assumptions of the Kinetic Molecular Theory
- Matter is made of tiny particles (atoms, molecules, or ions) in constant random motion.
- The kinetic energy of particles increases with temperature.
- Collisions between particles are perfectly elastic (no energy lost).
- There are no forces between gas particles except during collisions (ideal gas assumption).
- The volume of gas particles is negligible compared to the volume of the container.
Maxwell-Boltzmann Distribution
The Maxwell-Boltzmann distribution shows the spread of kinetic energies (or speeds) in a gas sample at a given temperature.
- Area under curve = Total number of particles (constant)
- Peak = Most probable speed/energy
- No particles at zero energy — the curve starts at the origin (y = 0 when x = 0)
- Long tail to the right — a few particles have very high energy
- Higher temperature → peak shifts right, becomes flatter and wider (same total area)
Interactive Simulation
AvailableExplore the Maxwell-Boltzmann distribution interactively — change temperature and see how the distribution shifts.
→ Launch Simulation🌡️ Temperature and Kinetic Energy
📺 S1.1.3 — Temperature (K) and Kinetic Energy
Key Definition: Temperature
Temperature ($T$ in Kelvin) is a measure of the average kinetic energy of particles. It is not the same as heat.
Conversion: $T(K) = T(°C) + 273$
Temperature vs Heat
| Concept | Temperature | Heat |
|---|---|---|
| What is it? | Average kinetic energy of particles | Total energy transferred between objects |
| Depends on quantity? | No (intensive property) | Yes (extensive property) |
| Units | Kelvin (K) or degrees Celsius (°C) | Joules (J) |
| Example | A spark at 3000°C has high T but low heat | A bathtub at 40°C has low T but high total heat |
Why Kelvin? The Kelvin scale starts at absolute zero (0 K = −273°C), where particles have minimum kinetic energy. This makes Kelvin directly proportional to average kinetic energy: $E_k \propto T$.
Heating Curves
A heating curve plots temperature against time as a substance is heated at constant rate. Key features:
- Sloped sections: Temperature rises — kinetic energy of particles increases
- Flat plateaus: Temperature stays constant — energy breaks intermolecular forces (phase change occurring)
- Longer plateau at boiling than melting — boiling requires more energy (all IMFs must be overcome)
⚗️ Elements, Compounds and Mixtures
Classifying Matter
All matter can be classified as either a pure substance (element or compound) or a mixture. The key distinction is whether the components are chemically bonded in a fixed ratio or simply physically combined.
| Classification | Definition | Examples |
|---|---|---|
| Element | Pure substance made of one type of atom; cannot be broken down chemically | Fe, O₂, Au, C |
| Compound | Two or more elements chemically bonded in a fixed ratio | H₂O, NaCl, CO₂ |
| Homogeneous Mixture | Uniform composition throughout; cannot see different parts | Salt water, air, alloys |
| Heterogeneous Mixture | Non-uniform; can distinguish different components | Sand + water, salad, granite |
Separation Techniques
| Technique | Separates Based On | Example Use |
|---|---|---|
| Filtration | Particle size (solid from liquid) | Sand from water |
| Distillation | Boiling point differences | Ethanol from water; desalination |
| Fractional Distillation | Boiling points (multiple liquids) | Crude oil separation; liquid air |
| Evaporation | Volatility (dissolved solid from solution) | Salt from seawater |
| Recrystallization | Solubility differences at different temperatures | Purifying impure crystals |
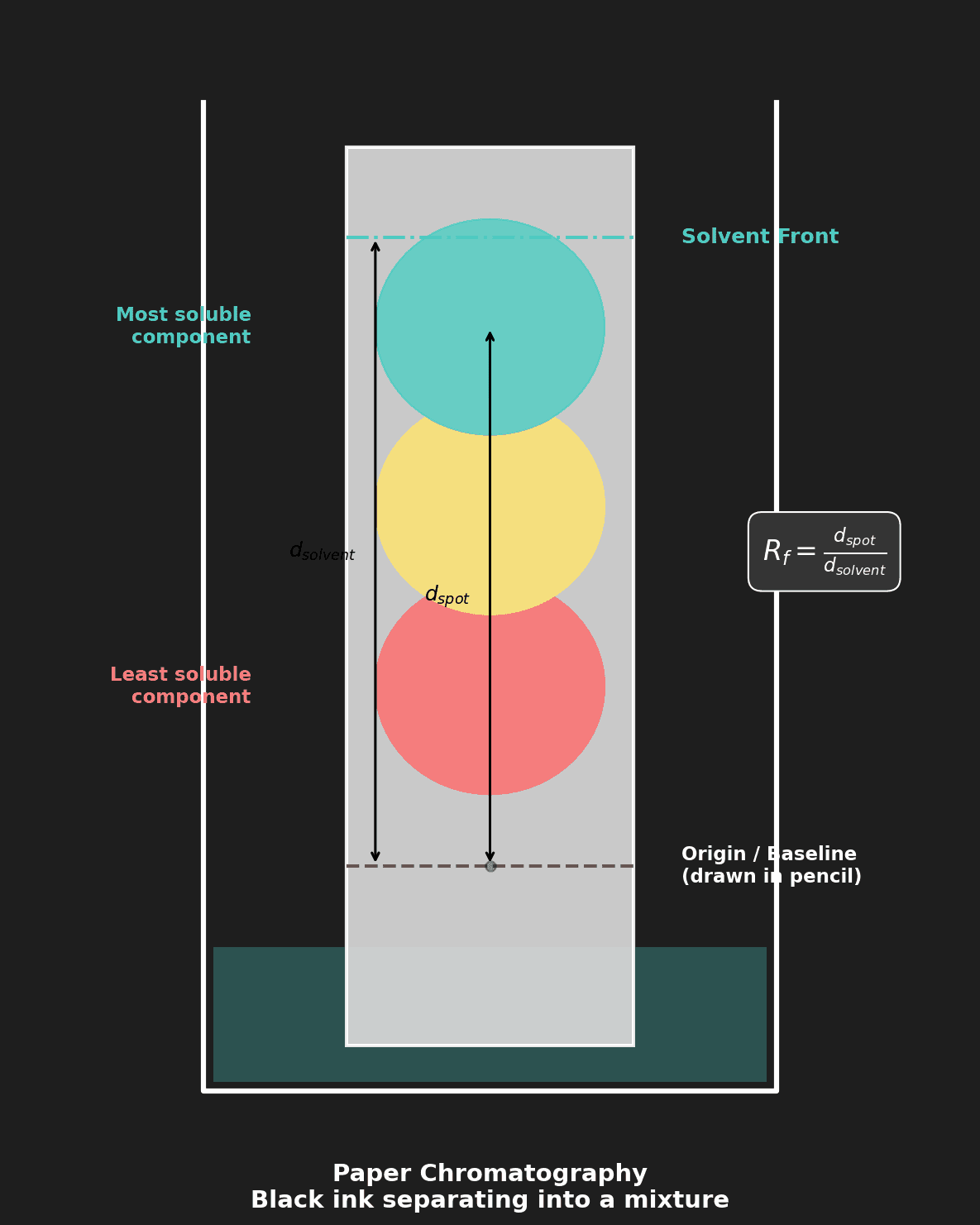
| Paper Chromatography | Solubility / adsorption (Rf values) | Separating dyes, plant pigments |
| Magnetism | Magnetic vs non-magnetic materials | Iron filings from sand |
Chromatography: The $R_f$ value = distance traveled by substance ÷ distance traveled by solvent front. Each substance has a characteristic $R_f$ value in a given solvent, which can be used for identification.
🧠 Memory Aids & Mnemonics
🔤 State Changes — Energy Direction
"MBS = Must Be Supplied" (Melting, Boiling, Sublimation = Endothermic)
All forward transitions (solid → liquid → gas) are endothermic — energy must be supplied. The reverse (condensation, freezing, deposition) are exothermic — energy is released.
🔤 State Symbols
"SLAG" = (s), (l), (aq), (g)
(s) solid, (l) liquid, (aq) aqueous (dissolved in water), (g) gas. Always include these in chemical equations.
🔤 Kelvin Conversion
K = °C + 273 → "Add 273 to climb to K"
0°C = 273 K. Absolute zero (0 K) = −273°C. Kelvin is always positive — there is no negative Kelvin.
🔤 Separation Technique Selection
"Is it dissolved? → Distill. Is it floating? → Filter."
Quick decision tree: insoluble solid → filtration. Dissolved solid → evaporation/distillation. Liquids with different BPs → fractional distillation. Colors → chromatography.
🌍 Real-World Applications
🧊 Why Does Ice Float?
Context: In almost all substances, the solid is denser than the liquid. Water is a remarkable exception — ice floats on water. Without this property, lakes would freeze from the bottom up, killing aquatic life.
Science: In ice, hydrogen bonds hold water molecules in an open hexagonal lattice. This structure has more empty space than liquid water, giving ice a lower density (~0.92 g/cm³ vs ~1.00 g/cm³ for liquid water). When ice melts, some hydrogen bonds break, and molecules pack more closely.
Impact: Floating ice insulates the water below, allowing fish and other organisms to survive winter. This anomaly is essential for the existence of freshwater ecosystems at high latitudes.
🥤 Freeze-Drying (Lyophilization)
Context: Freeze-dried food (instant coffee, astronaut meals, camping food) relies on sublimation to preserve food without destroying nutrients or flavor.
Science: Food is first frozen to very low temperatures, then placed in a vacuum chamber. Under low pressure, ice sublimes directly from solid to gas (bypassing the liquid phase). This removes water without heating, preserving the structure and nutrients of the food. The resulting product is lightweight and has a shelf life of 25+ years.
Impact: Freeze-drying is used in the pharmaceutical industry (vaccine preservation), space food programs (NASA has used it since the 1960s), and even in preserving biological specimens for research museums.
☕ Supercritical CO₂ Decaffeination
Context: How do you remove caffeine from coffee beans without using harsh chemical solvents?
Science: Carbon dioxide is heated above 31°C and compressed above 73 atm. At these conditions, it becomes a supercritical fluid. A supercritical fluid has properties of both a liquid (it can dissolve substances) and a gas (it can flow through solids). Supercritical CO₂ is pumped through coffee beans. It dissolves the caffeine but leaves the flavor compounds behind. Then the CO₂ pressure is lowered, the caffeine separates out, and the CO₂ is recycled.
Impact: This is the greenest decaffeination method — no toxic solvents, CO₂ is recycled, and the flavor of the coffee is preserved. Supercritical fluids are a distinct phase region on a phase diagram, not a traditional state of matter.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Confusing boiling with evaporation: Boiling occurs at a specific temperature throughout the liquid. Evaporation happens at any temperature, only at the surface. Examiners ask: "Explain why evaporation causes cooling" — the fastest particles leave, lowering the average KE.
- Saying "particles expand" when heated: Particles do NOT expand. They move faster and spread further apart. The substance expands, not the particles themselves.
- Forgetting state symbols in equations: IB examiners specifically penalize missing state symbols. Always write $\text{NaCl(aq)}$, not just $\text{NaCl}$.
- Mixing up homogeneous and heterogeneous: Homo = same (uniform). Hetero = different (non-uniform). A solution (e.g., salt water) looks the same everywhere → homogeneous. Sand in water → you can see both parts → heterogeneous.
- Temperature stays constant during phase changes: Students often draw heating curves without flat plateaus. During melting and boiling, all supplied energy breaks IMFs — temperature does not change.
- Using °C instead of K in gas calculations: For any gas law formula involving temperature, always convert to Kelvin first. Using °C gives wrong answers.
📝 IB-Style Exam Questions
Question 1: State what the area under a Maxwell-Boltzmann distribution curve represents. [1 mark]
Mark Scheme:
- [1 mark] The total number of particles (molecules/atoms) in the sample.
Question 2: Explain why the temperature of boiling water remains constant even though heat is being supplied. [2 marks]
Mark Scheme:
- [1 mark] The heat energy supplied is used to break intermolecular forces (hydrogen bonds).
- [1 mark] Instead of increasing the kinetic energy (temperature) of the molecules.
Question 3: Distinguish between the properties of a solid and a gas in terms of particle arrangement and motion. [3 marks]
Mark Scheme:
- [1 mark] Arrangement: Solids have particles in a fixed, regular lattice; Gases have particles far apart with no regular arrangement.
- [1 mark] Motion: Solid particles vibrate in fixed positions.
- [1 mark] Motion: Gas particles move rapidly and randomly, colliding with each other.
Question 4: A mixture contains sand and salt water. Outline the steps to obtain pure sand and pure water from this mixture. [3 marks]
Mark Scheme:
- [1 mark] Filter the mixture to separate the insoluble sand from the salt solution.
- [1 mark] Wash the sand with distilled water and dry it.
- [1 mark] Perform simple distillation on the filtrate (salt solution) to collect pure water.
Question 5: Deduce the state of substance X at 25°C given its melting point is −15°C and boiling point is 80°C. [1 mark]
Mark Scheme:
- [1 mark] Liquid (since 25°C is between MP and BP).
Question 6: Explain why evaporation causes the temperature of a liquid to decrease. [2 marks]
Mark Scheme:
- [1 mark] Evaporation involves the most energetic / fastest particles at the surface escaping the liquid.
- [1 mark] The remaining particles have a lower average kinetic energy, so the temperature decreases.
Question 7: Using the kinetic molecular theory, explain why gases are much more compressible than liquids. [2 marks]
Mark Scheme:
- [1 mark] In gases, particles are far apart with large spaces between them.
- [1 mark] Pressure can force particles closer together into the empty space. In liquids, particles are already close together with little empty space to compress into.
Question 8: Outline how paper chromatography can be used to determine whether a sample of black ink is a pure substance or a mixture. [3 marks]
Mark Scheme:
- [1 mark] A spot of ink is placed on chromatography paper above the solvent line, and the paper is placed in a solvent.
- [1 mark] The solvent moves up the paper by capillary action, carrying the components of the ink with it.
- [1 mark] If only one spot is observed, the ink is a pure substance. If multiple spots appear, the ink is a mixture of different pigments.