📋 IB Content Statements (S1.2)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S1.2.1: Atoms contain a positively charged, dense nucleus composed of protons and neutrons (nucleons). Negatively charged electrons occupy the space outside the nucleus.
- S1.2.2: Isotopes are atoms of the same element with different numbers of neutrons.
- S1.2.3: The relative atomic mass ($A_r$) of an element is the weighted mean of the masses of its isotopes relative to ¹²C.
- S1.2.4: The number of protons (atomic number, $Z$) defines the element. The mass number ($A$) is the sum of protons and neutrons.
HL Extensions
- S1.2.5: Mass spectrometry is used to determine the relative atomic mass and the abundance of isotopes of an element.
- S1.2.6: Ions are created in a mass spectrometer by bombardment with high-energy electrons.
🔹 Subatomic Particles
The atom consists of a small, dense nucleus (containing protons and neutrons) surrounded by electrons in energy levels (shells).
| Particle | Relative Mass | Relative Charge | Location | Symbol |
|---|---|---|---|---|
| Proton | 1 | +1 | Nucleus | $p^+$ |
| Neutron | 1 | 0 | Nucleus | $n^0$ |
| Electron | $\frac{1}{1840}$ (≈ 0) | −1 | Energy levels (shells) | $e^-$ |
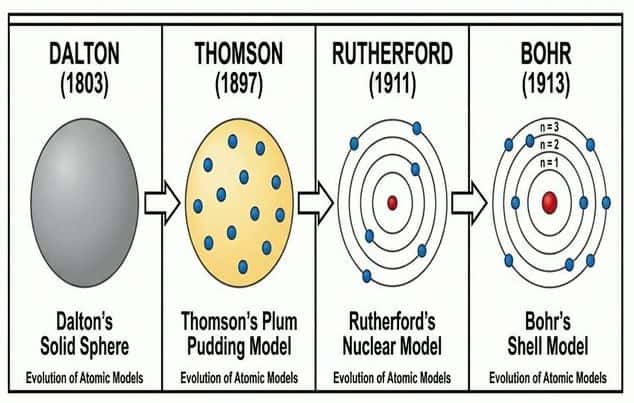
Historical models:
- Dalton (1803): Atoms are solid, indivisible spheres.
- Thomson (1897): "Plum pudding" — electrons embedded in positive dough. Discovered the electron.
- Rutherford (1911): Gold foil experiment → most of the atom is empty space with a small, dense, positive nucleus.
- Bohr (1913): Electrons orbit in fixed energy levels (shells).
🔢 Nuclear Notation
Notation: ${}_{Z}^{A}X$
- $A$ = Mass number = protons + neutrons (top)
- $Z$ = Atomic number = number of protons (bottom) — this defines the element
Calculating Subatomic Particles
| Quantity | Formula | Example: ${}_{17}^{35}Cl$ | Example: ${}_{17}^{37}Cl^-$ |
|---|---|---|---|
| Protons | $Z$ | 17 | 17 |
| Neutrons | $A - Z$ | $35 - 17 = 18$ | $37 - 17 = 20$ |
| Electrons | $Z - \text{charge}$ | 17 (neutral atom) | $17 + 1 = 18$ (anion) |
⚠️ Ions: For cations (+), subtract the charge from $Z$ to get electrons. For anions (−), add the magnitude of the charge. $Na^+$ has 10 electrons (11 − 1). $O^{2-}$ has 10 electrons (8 + 2).
⚛️ Isotopes & Relative Atomic Mass
📺 S1.2.2 — RAM Calculations with Isotope Abundance
📺 S1.2.2 — Physical Properties of Isotopes
Definition: Isotopes
Atoms of the same element (same number of protons / same $Z$) with different numbers of neutrons (different $A$).
Properties of Isotopes
| Property Type | Same or Different? | Reason |
|---|---|---|
| Chemical properties | Same | Same number of electrons → same bonding and reactivity |
| Physical properties | Different | Different mass → different density, melting/boiling point, rate of diffusion |
| Radioactivity | May differ | Some isotopes are stable, others are radioactive (unstable nucleus) |
Calculating Relative Atomic Mass ($A_r$)
$$A_r = \frac{(\text{mass}_1 \times \text{abundance}_1) + (\text{mass}_2 \times \text{abundance}_2)}{100}$$
Worked example: Chlorine has two isotopes: $^{35}Cl$ (75.8%) and $^{37}Cl$ (24.2%).
$A_r = \frac{(35 \times 75.8) + (37 \times 24.2)}{100} = \frac{2653 + 895.4}{100} = 35.48$
Why $A_r$ values on the periodic table aren't whole numbers: They are weighted averages of all naturally occurring isotopes. Chlorine's $A_r$ of 35.5 reflects that it has roughly 3 parts $^{35}Cl$ for every 1 part $^{37}Cl$.
🧪 Mass Spectrometry (HL)
📺 S1.2.3 — Mass Spectra of Isotopes (HL)
The Mass Spectrometer
A mass spectrometer separates atoms or molecules based on their mass-to-charge ratio ($m/z$).
Five Stages of Mass Spectrometry
| Stage | Process | Detail |
|---|---|---|
| 1. Vaporization | Sample → gas phase | Heated to produce gaseous atoms/molecules |
| 2. Ionization | Atoms → positive ions | High-energy electron bombardment removes an electron: $X(g) \rightarrow X^+(g) + e^-$ |
| 3. Acceleration | Ions accelerated | Electric field gives all ions the same kinetic energy |
| 4. Deflection | Ions separated | Magnetic field deflects lighter ions more; heavier ions deflect less |
| 5. Detection | Ions detected | Produces a mass spectrum: peaks at each $m/z$ value |
Reading a Mass Spectrum
- Number of peaks = number of isotopes
- Position (x-axis) = mass-to-charge ratio ($m/z$)
- Height (y-axis) = relative abundance (%)
- Tallest peak = most abundant isotope
Interactive: Mass Spectrometry
AvailableExplore mass spectra of different elements and calculate $A_r$ from spectral data.
→ Launch Simulation🧪 Interactive Virtual Labs
Mass Spectrometry Explorer
AvailableExplore mass spectra of different elements. Calculate relative atomic mass from abundance data.
→ Launch Simulation🧠 Memory Aids & Mnemonics
🔤 Subatomic Particle Table
"PEN" → Proton, Electron, Neutron (+1, −1, 0)
The charges go +1, −1, 0 in alphabetical order of the first letter. P is positive, E is negative (think: e for electron, e for "elementary negative charge"), N is neutral.
🔤 Mass Number vs Atomic Number
"A is Above, Z is Below" (in nuclear notation ${}_{Z}^{A}X$)
$A$ (mass number) is the superscript (above). $Z$ (atomic number) is the subscript (below). $Z$ = protons = defines the element. $A$ = protons + neutrons.
🔤 Isotopes Definition
"Same Protons, Different Neutrons"
Isotopes have the same atomic number ($Z$) but different mass numbers ($A$). They are the same element because they have the same number of protons. Chemical properties are the same because they have the same number of electrons.
🔤 Mass Spectrometer Steps (HL)
"Very Intelligent Athletes Dodge Defenders"
Vaporization → Ionization → Acceleration → Deflection → Detection. Five stages in order.
🌍 Real-World Applications
🦴 Radiocarbon Dating (Carbon-14)
Context: How do archaeologists know a bone is 5,000 years old? They use the isotope Carbon-14.
Science: Living organisms constantly absorb $^{14}C$ from the atmosphere (via $CO_2$). When the organism dies, $^{14}C$ is no longer absorbed and begins to decay: $^{14}C \rightarrow ^{14}N + \beta^-$ with a half-life of 5,730 years. By measuring the ratio of $^{14}C$ to stable $^{12}C$ in a sample, scientists calculate the time since death. This works for samples up to ~50,000 years old.
Impact: Radiocarbon dating has revolutionized archaeology, geology, and climate science. It confirmed the age of the Dead Sea Scrolls and helped date the last Ice Age.
🏥 Medical Isotopes (Technetium-99m)
Context: Hospitals use radioactive isotopes to diagnose diseases without surgery. Over 40 million nuclear medicine procedures are performed worldwide each year.
Science: Technetium-99m ($^{99m}Tc$) is the most widely used medical isotope. It emits gamma rays (detected by cameras outside the body) and has a short half-life of just 6 hours — long enough to perform scans but short enough to minimize radiation exposure. It can be attached to different molecules to target specific organs (bones, heart, kidneys, brain).
Impact: Medical isotopes save millions of lives annually. Iodine-131 treats thyroid cancer by selectively destroying thyroid cells. PET scans use Fluorine-18 to detect cancerous tumors by tracking glucose metabolism.
🔬 Forensic Mass Spectrometry
Context: Mass spectrometry is used in drug testing, forensic investigations, and doping control at the Olympics.
Science: In a mass spectrometer, unknown substances are vaporized, ionized, and separated by mass. The resulting mass spectrum acts as a chemical fingerprint — each substance produces a unique pattern of peaks. By comparing the spectrum to a database, forensic scientists can identify drugs, poisons, explosives, or environmental pollutants in tiny quantities (parts per billion).
Impact: GC-MS (gas chromatography–mass spectrometry) is the "gold standard" for forensic drug testing. It was used to identify nerve agents in the Novichok poisoning cases and is routinely used in anti-doping tests at the Olympics.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Confusing mass number with relative atomic mass: Mass number ($A$) is always a whole number (protons + neutrons for one specific isotope). $A_r$ is a weighted average and usually has decimal places (e.g., Cl = 35.5).
- Forgetting to adjust electrons for ions: $Fe^{3+}$ has 23 electrons (26 − 3), not 26. $S^{2-}$ has 18 electrons (16 + 2), not 16. Always check the charge!
- Saying isotopes have different chemical properties: Isotopes have the same chemical properties because they have the same electron configuration. Only physical properties differ (mass, density, rate of diffusion).
- Wrong formula for $A_r$ calculation: Don't just average the masses. You must use a weighted average: $A_r = \frac{\sum(\text{mass} \times \text{abundance})}{100}$. If one isotope is much more abundant, $A_r$ will be closer to that isotope's mass.
- Confusing atomic number with mass number positions: $A$ (mass) is the superscript (top), $Z$ (atomic number) is the subscript (bottom). Many students reverse these.
📝 IB-Style Exam Questions
Question 1: Calculate the relative atomic mass of Boron given: Boron-10 (19.9%) and Boron-11 (80.1%). [2 marks]
Mark Scheme:
- [1 mark] Calculation: $\frac{(10 \times 19.9) + (11 \times 80.1)}{100}$
- [1 mark] Answer: 10.8
Question 2: State two physical properties that differ for isotopes of the same element. [2 marks]
Mark Scheme:
- [1 mark] Mass / density
- [1 mark] Boiling point / melting point / rate of diffusion
Question 3: Describe the purpose of the magnetic field in a mass spectrometer. [1 mark]
Mark Scheme:
- [1 mark] To deflect ions based on their mass-to-charge ratio ($m/z$). Lighter ions are deflected more than heavier ions.
Question 4: Determine the number of protons, neutrons, and electrons in $^{37}Cl^-$. [2 marks]
Mark Scheme:
- [1 mark] Protons = 17, Neutrons = 20 ($37 - 17$)
- [1 mark] Electrons = 18 ($17 + 1$ for the negative charge)
Question 5: Explain why isotopes of the same element have the same chemical properties. [1 mark]
Mark Scheme:
- [1 mark] They have the same number of electrons / same electron configuration, so they react in the same way.
Question 6: Define the term "relative atomic mass". [2 marks]
Mark Scheme:
- [1 mark] The weighted mean/average of the masses of all naturally occurring isotopes of an element.
- [1 mark] Relative to $\frac{1}{12}$ the mass of a carbon-12 atom.
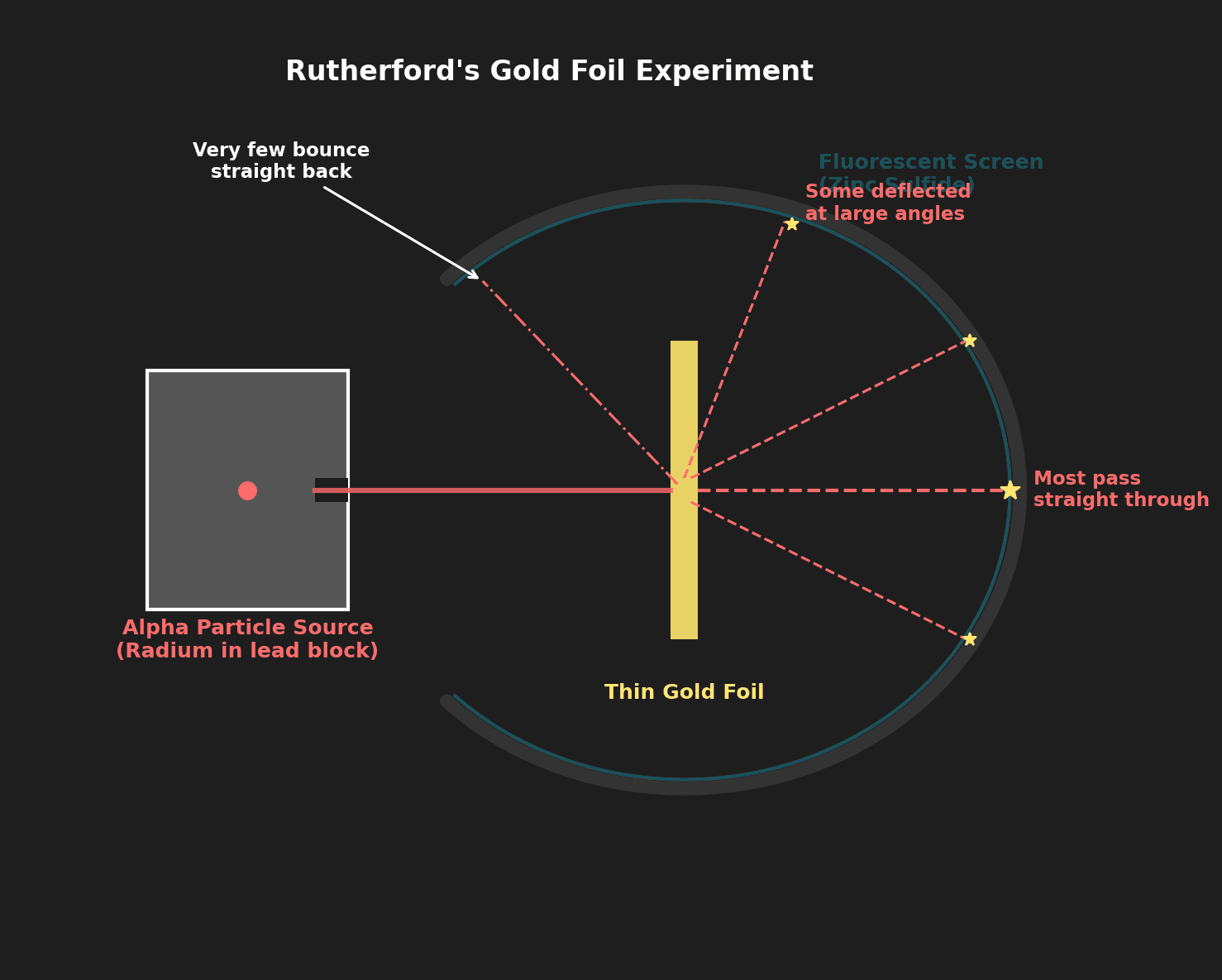
Question 7: Outline how Rutherford's gold foil experiment provided evidence for the nuclear model of the atom. [3 marks]
Mark Scheme:
- [1 mark] Most alpha particles passed straight through → most of the atom is empty space.
- [1 mark] A few were deflected at large angles → the nucleus is positively charged (repels positive alpha particles).
- [1 mark] Very few bounced back → the nucleus is very small and dense (concentrated positive charge).
Question 8: Silicon has three isotopes: $^{28}Si$ (92.2%), $^{29}Si$ (4.7%), $^{30}Si$ (3.1%). Calculate the relative atomic mass of silicon. [2 marks]
Mark Scheme:
- [1 mark] $A_r = \frac{(28 \times 92.2) + (29 \times 4.7) + (30 \times 3.1)}{100}$
- [1 mark] $= \frac{2581.6 + 136.3 + 93.0}{100} = \frac{2810.9}{100} = 28.1$