📋 IB Content Statements (S1.3)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S1.3.1: Emission spectra provide evidence for the existence of discrete energy levels in atoms.
- S1.3.2: The line emission spectrum of hydrogen provides evidence for convergence of energy levels at higher energies.
- S1.3.3: The main energy level ($n$) is split into subshells (s, p, d, f), each containing a fixed number of orbitals.
- S1.3.4: An orbital holds a maximum of two electrons with opposite spins.
- S1.3.5: The electron configuration of atoms and ions up to $Z = 36$ can be deduced from their position on the periodic table.
HL Extensions
- S1.3.6: The Aufbau principle, Pauli exclusion principle, and Hund's rule determine the order of filling of orbitals.
- S1.3.7: Successive ionization energy data provide evidence for the existence of main energy levels and subshells.
- S1.3.8: Trends in first ionization energies across periods and down groups are explained by the balance between nuclear charge, shielding, and distance.
🔹 Emission Spectra: Evidence for Shells
📺 S1.3.1 — Continuous vs Line Spectrum
The Bohr Model
Electrons orbit the nucleus in specific, quantized energy levels ($n = 1, 2, 3...$). Energy is NOT continuous — it comes in fixed amounts. Electrons can transition between levels by absorbing or emitting photons of specific energy:
$$\Delta E = h\nu = \frac{hc}{\lambda}$$
Line Spectra Explained
- Continuous spectrum: All wavelengths present (e.g., white light through a prism → rainbow).
- Line emission spectrum: Only specific wavelengths emitted → discrete colored lines on a dark background. Each line corresponds to a specific electron transition.
- Line absorption spectrum: Dark lines on a continuous background — atoms absorb specific wavelengths.
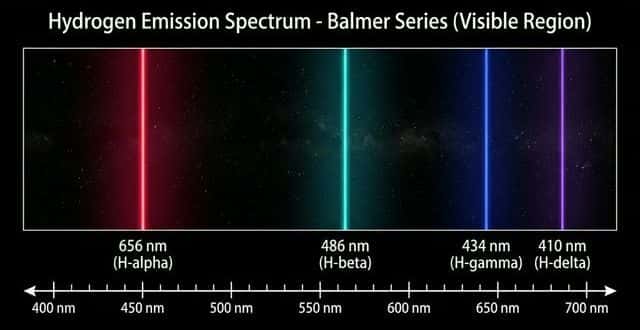
Hydrogen emission spectrum: Shows distinct series of lines:
- Lyman series (UV): transitions to $n = 1$
- Balmer series (visible): transitions to $n = 2$ — the four visible lines: red (656 nm), blue-green (486 nm), blue-violet (434 nm), violet (410 nm)
- Paschen series (IR): transitions to $n = 3$
⚠️ Convergence: Lines get closer together at higher energies because the energy levels themselves converge. At the convergence limit, the electron has been completely removed — this corresponds to ionization.
🔮 Orbitals & Subshells
📺 S1.3.3 — Energy Levels, Orbitals and Electrons
What is an Orbital?
An orbital is a region of space where there is a high probability (~95%) of finding an electron. Each orbital holds a maximum of 2 electrons with opposite spins (↑↓).
| Subshell | Shape | Number of Orbitals | Max Electrons | Available from Level |
|---|---|---|---|---|
| s | Spherical | 1 | 2 | $n = 1$ |
| p | Dumbbell (3 orientations: $p_x, p_y, p_z$) | 3 | 6 | $n = 2$ |
| d | Cloverleaf (complex shapes) | 5 | 10 | $n = 3$ |
| f | Complex multi-lobed | 7 | 14 | $n = 4$ |
Maximum electrons per level: $2n^2$. So level 1 = 2, level 2 = 8, level 3 = 18, level 4 = 32.
⚛️ Writing Electron Configurations
Three Rules for Filling Orbitals
- Aufbau Principle: Fill lowest energy orbitals first (1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p...).
- Pauli Exclusion Principle: Maximum 2 electrons per orbital, with opposite spins (↑↓).
- Hund's Rule: In degenerate orbitals (same energy, e.g., the three 2p orbitals), fill each orbital singly before pairing.
Filling Order
The order of subshell filling (by energy):
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → 5s → 4d → 5p → 6s → 4f → 5d → 6p
Note: 4s fills before 3d (4s is lower in energy). But when forming ions from d-block elements, 4s electrons are removed first (4s is now higher in energy when d orbitals are occupied).
Examples
| Element / Ion | Full Configuration | Shorthand |
|---|---|---|
| Carbon ($Z = 6$) | $1s^2 \ 2s^2 \ 2p^2$ | $[He] \ 2s^2 \ 2p^2$ |
| Iron ($Z = 26$) | $1s^2 \ 2s^2 \ 2p^6 \ 3s^2 \ 3p^6 \ 4s^2 \ 3d^6$ | $[Ar] \ 4s^2 \ 3d^6$ |
| $Fe^{2+}$ | $1s^2 \ 2s^2 \ 2p^6 \ 3s^2 \ 3p^6 \ 3d^6$ | $[Ar] \ 3d^6$ |
| $Fe^{3+}$ | $1s^2 \ 2s^2 \ 2p^6 \ 3s^2 \ 3p^6 \ 3d^5$ | $[Ar] \ 3d^5$ |
🔀 Chromium & Copper Exceptions
⚠️ You MUST memorize these two exceptions:
| Element | Expected | Actual | Reason |
|---|---|---|---|
| Chromium ($Z = 24$) | $[Ar] \ 4s^2 \ 3d^4$ | $[Ar] \ 4s^1 \ 3d^5$ | Half-filled 3d is more stable (all orbitals singly occupied) |
| Copper ($Z = 29$) | $[Ar] \ 4s^2 \ 3d^9$ | $[Ar] \ 4s^1 \ 3d^{10}$ | Fully-filled 3d is more stable |
Why? Half-filled and fully-filled d subshells have extra stability due to symmetrical electron distribution and exchange energy. A 4s electron is promoted to 3d to achieve this.
⚡ Ionization Energies (HL)
First Ionization Energy
The energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous 1+ ions:
$$X(g) \rightarrow X^+(g) + e^-$$
Factors Affecting IE
| Factor | Effect on IE |
|---|---|
| Nuclear charge ($Z$) | More protons → stronger attraction → higher IE |
| Electron shielding | More inner shells → more shielding → lower IE |
| Distance from nucleus | Further from nucleus → weaker attraction → lower IE |
| Subshell type | p electrons easier to remove than s (higher energy, more shielded) |
| Electron pairing | Paired electrons repel → easier to remove one (e.g., O vs N) |
Trends
- Across a period (→): IE generally increases (more protons, same shielding)
- Down a group (↓): IE decreases (more shells, more shielding, greater distance)
Anomalies to Explain
- Be → B drop: B's outer electron is in 2p (higher energy, shielded by 2s)
- N → O drop: O has a paired electron in 2p — electron repulsion makes it easier to remove
Successive Ionization Energies
A graph of successive IEs shows large jumps when an electron is removed from a new (inner) main energy level. This provides evidence for electron shell structure.
Interactive: Ionization Energy Grapher
AvailableAnalyze IE trends across periods and plot successive IEs for different elements.
→ Launch Simulation🧪 Interactive Virtual Labs
Hydrogen Emission Spectrum
AvailableObserve electron transitions and the resulting spectral lines for hydrogen.
→ Launch Simulation🧠 Memory Aids & Mnemonics
🔤 Filling Order
1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p
Use the diagonal rule: write subshells in a grid and read diagonally. Or just memorize the order as a chant. The key surprise: 4s fills before 3d.
🔤 Three Rules
"Aufbau Puts Hund's" → Aufbau, Pauli, Hund's
Aufbau = build up (lowest energy first). Pauli = pairs have opposite spins (max 2 per orbital). Hund's = half-fill before pairing (spread out in degenerate orbitals).
🔤 Subshell Capacities
"s=2, p=6, d=10, f=14" (increase by 4)
Each subshell has an odd number of orbitals (1, 3, 5, 7), each holding 2 electrons. So s = 2, p = 6, d = 10, f = 14. Pattern: +4 each time.
🔤 Cr and Cu Exceptions
"Crazy Copper Steals an s-electron for d-stability"
Both Cr ($[Ar]4s^13d^5$) and Cu ($[Ar]4s^13d^{10}$) move one 4s electron to 3d to achieve a half-filled or fully-filled d subshell, which is more stable.
🔤 Ion Electron Removal
"4s fills First, but Leaves First"
When forming ions of d-block elements, remove 4s electrons before 3d electrons. $Fe = [Ar]4s^23d^6$ → $Fe^{2+} = [Ar]3d^6$ (not $[Ar]4s^23d^4$).
🌍 Real-World Applications
💡 Neon Signs & Gas Discharge Tubes
Context: The colorful neon signs on streets work because of emission spectra — the exact topic of S1.3.
Science: A high voltage excites electrons in gas atoms to higher energy levels. When electrons fall back down, they emit photons at specific wavelengths determined by the energy gap ($\Delta E = h\nu$). Different gases produce different colors: neon → red/orange, argon → blue/violet, helium → yellow/pink, mercury vapor → blue/green. The line emission spectrum is unique to each element — a "fingerprint".
Impact: This principle is used in fluorescent lighting, LED screens, and analytical chemistry (flame tests and atomic emission spectroscopy for metal identification in forensics and environmental monitoring).
🔥 Flame Tests and Fireworks
Context: Fireworks produce their spectacular colors because metal ions emit specific wavelengths of light when heated.
Science: The heat energy from the explosion excites electrons in metal ions. As electrons return to ground state, they emit photons: lithium/strontium → red, sodium → yellow, barium → green, copper → blue/green, potassium → lilac. Each metal has a unique set of energy level spacings, producing characteristic colors.
Impact: Flame tests are a quick, simple analytical technique used in IB Chemistry practicals to identify unknown metal ions. Firework chemistry is a direct application of quantized energy levels and emission spectra.
⭐ Stellar Spectroscopy
Context: How do astronomers know what stars are made of, even though they can never visit one?
Science: Stars emit light that passes through their outer atmosphere. The elements in the atmosphere absorb specific wavelengths, creating dark absorption lines in the continuous spectrum. By comparing these dark lines to known emission spectra of elements on Earth, astronomers identify the chemical composition of stars. Helium was discovered this way — it was detected in the Sun's spectrum before it was found on Earth (hence "helium" from Greek "helios" = sun).
Impact: Spectroscopy reveals not only composition but also temperature, density, and motion of celestial objects. The redshift of spectral lines provided evidence for the expanding universe.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Removing 3d electrons first when forming ions: When d-block atoms form ions, the 4s electrons are removed first, not 3d. $Fe^{2+}$ is $[Ar]3d^6$, NOT $[Ar]4s^23d^4$.
- Forgetting Cr and Cu exceptions: Cr is $[Ar]4s^13d^5$ (not $4s^23d^4$) and Cu is $[Ar]4s^13d^{10}$ (not $4s^23d^9$). These are frequently tested.
- Confusing emission and absorption spectra: Emission = bright lines on dark background (atoms emit light). Absorption = dark lines on bright background (atoms absorb light). Both have lines at the same wavelengths for a given element.
- Writing subshells in wrong order for d-block: Write in order of filling (4s before 3d) for the atom, but when showing ion configurations, 3d is written before any remaining 4s. Convention: write in order of sublevel number ($3d$ before $4s$) for ions.
- Forgetting what convergence means: Lines getting closer together at higher frequencies → energy levels converge. The convergence limit = ionization energy. Students often forget to link convergence to ionization.
📝 IB-Style Exam Questions
Question 1: State the full electron configuration for the Copper(I) ion, $Cu^+$. [1 mark]
Mark Scheme:
- [1 mark] $1s^2 \ 2s^2 \ 2p^6 \ 3s^2 \ 3p^6 \ 3d^{10}$ (or $[Ar] \ 3d^{10}$). Note: Cu is $[Ar]4s^13d^{10}$, so removing one electron takes the 4s electron.
Question 2: Explain why the first ionization energy of Boron is lower than Beryllium, despite Boron having a higher nuclear charge. [2 marks]
Mark Scheme:
- [1 mark] Boron's outer electron is in the 2p subshell, which is higher in energy than the 2s subshell.
- [1 mark] The 2p electron is shielded by the 2s electrons, making it easier to remove/requiring less energy.
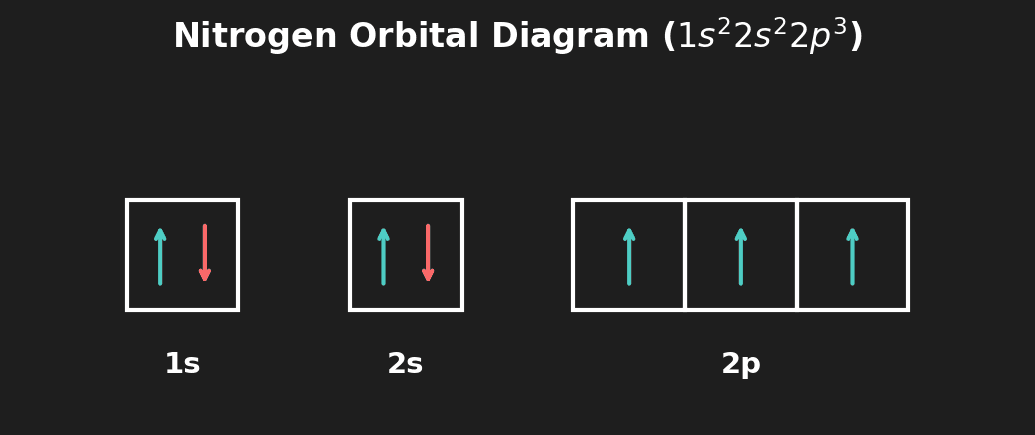
Question 3: Draw the orbital diagram (arrows in boxes) for the valence shell of Nitrogen. [1 mark]
Mark Scheme:
- [1 mark] 2s box: two opposite arrows (↑↓). Three 2p boxes: one upward arrow each (↑ ↑ ↑). This demonstrates Hund's rule — each 2p orbital is singly occupied before pairing.
Question 4: Explain the meaning of the term "convergence limit" in the hydrogen emission spectrum. [2 marks]
Mark Scheme:
- [1 mark] The point where the spectral lines merge/come together at higher frequencies.
- [1 mark] This corresponds to the electron being completely removed from the atom (ionization, $n = \infty$).

Question 5: State the maximum number of electrons that can occupy the $n = 3$ energy level. [1 mark]
Mark Scheme:
- [1 mark] 18 ($2n^2 = 2 \times 3^2 = 18$; subshells: 3s(2) + 3p(6) + 3d(10) = 18).
Question 6: Write the electron configuration of $Fe^{3+}$ and explain why 4s electrons are removed before 3d. [2 marks]
Mark Scheme:
- [1 mark] $Fe^{3+} = [Ar] \ 3d^5$
- [1 mark] When d orbitals are occupied, 4s becomes higher in energy than 3d, so 4s electrons are removed first upon ionization.
Question 7: Explain why the first ionization energy of Oxygen is lower than Nitrogen. [2 marks]
Mark Scheme:
- [1 mark] Nitrogen has a half-filled 2p subshell ($2p^3$) which has extra stability.
- [1 mark] Oxygen has a paired electron in one of its 2p orbitals ($2p^4$) — the electron-electron repulsion in the pair makes it easier to remove one electron.
Question 8: Distinguish between a continuous spectrum and a line emission spectrum. [2 marks]
Mark Scheme:
- [1 mark] A continuous spectrum contains all wavelengths/frequencies of light (e.g., white light through a prism).
- [1 mark] A line emission spectrum contains only specific/discrete wavelengths, corresponding to electron transitions between fixed energy levels in an atom.