📋 IB Content Statements (S1.4)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S1.4.1: The mole ($n$) is a fixed number of particles. One mole contains exactly $6.02 \times 10^{23}$ particles (Avogadro's constant, $L$).
- S1.4.2: Masses of atoms are compared on a scale relative to $^{12}C$, and expressed as relative atomic mass ($A_r$) and relative formula/molecular mass ($M_r$).
- S1.4.3: The molar mass ($M$) has units of $g \ mol^{-1}$.
- S1.4.4: The empirical formula of a compound gives the simplest ratio of atoms. The molecular formula gives the actual number.
- S1.4.5: The relationship between the empirical and molecular formulas can be determined from the molar mass.
- S1.4.6: The amount of substance in a chemical equation is shown by stoichiometric coefficients.
- S1.4.7: The limiting reagent determines the theoretical yield. The experimental yield can be expressed as a percentage of the theoretical yield.
🔹 The Mole & Avogadro's Constant
Definition: The Mole ($n$)
The amount of substance that contains exactly $6.02 \times 10^{23}$ particles (atoms, molecules, ions, electrons, etc.). This number is called Avogadro's constant ($L$).
One mole of any substance contains the same number of particles as there are atoms in exactly 12 g of Carbon-12.
How big is Avogadro's number?
- A mole of grains of sand would cover the entire United States to a depth of several meters.
- If you counted one atom per second, it would take ~19 quadrillion years to count one mole.
- One mole of water ($18 \ g$) = about 18 mL = one tablespoon of water.
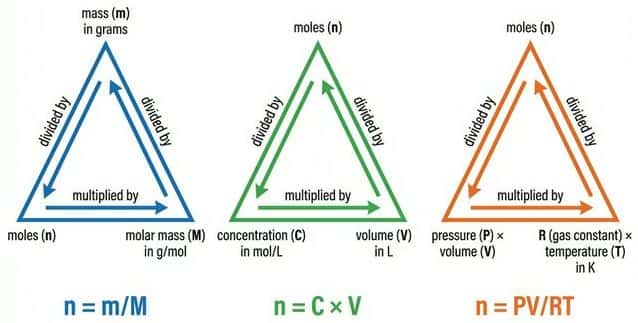
🧮 Key Formulas
| Relationship | Formula | Units | When to Use |
|---|---|---|---|
| Mass → Moles | $n = \frac{m}{M}$ | $m$ in g, $M$ in $g \ mol^{-1}$ | Most common calculation — given mass, find moles |
| Particles → Moles | $n = \frac{N}{L}$ | $N$ = number of particles | When asked about number of atoms/molecules |
| Volume of gas → Moles | $n = \frac{V}{V_m}$ | $V_m = 22.7 \ dm^3$ at STP | Gas calculations at standard conditions |
| Concentration → Moles | $n = c \times V$ | $c$ in $mol \ dm^{-3}$, $V$ in $dm^3$ | Solution calculations |
⚠️ Unit conversions: $1 \ dm^3 = 1000 \ cm^3 = 1 \ L$. Always convert $cm^3$ to $dm^3$ by dividing by 1000 before using $n = cV$.
⚗️ Empirical & Molecular Formulas
📺 S1.4.4 — Combustion Data to Calculate Empirical Formula
Definitions
- Empirical formula: The simplest whole number ratio of atoms in a compound (e.g., $CH_2O$).
- Molecular formula: The actual number of atoms in one molecule (e.g., $C_6H_{12}O_6$ — glucose).
Steps to Find Empirical Formula
| Step | Action | Example: 40.0% C, 6.7% H, 53.3% O |
|---|---|---|
| 1 | Assume 100 g → mass = % value | 40.0 g C, 6.7 g H, 53.3 g O |
| 2 | Divide by molar mass | $\frac{40.0}{12.01} = 3.33$, $\frac{6.7}{1.01} = 6.63$, $\frac{53.3}{16.00} = 3.33$ |
| 3 | Divide by smallest number | $\frac{3.33}{3.33} = 1$, $\frac{6.63}{3.33} = 2$, $\frac{3.33}{3.33} = 1$ |
| 4 | Round to nearest whole number | Empirical formula = $CH_2O$ |
Molecular Formula from Empirical Formula
$$n = \frac{M_r(\text{molecular})}{M_r(\text{empirical})}$$
If $M_r = 180$ and empirical formula is $CH_2O$ ($M_r = 30$), then $n = 180/30 = 6$, so molecular formula = $C_6H_{12}O_6$.
💧 Water of Crystallization
Hydrated Salts
Many ionic compounds crystallize from solution with water molecules trapped in the lattice. For example, $CuSO_4 \cdot 5H_2O$ (blue) → heated → $CuSO_4$ (white) + $5H_2O$.
Experimental Method
- Weigh crucible empty → weigh with hydrated salt → heat → weigh again → repeat until constant mass
- Mass of water = mass before heating − mass after heating
- Calculate moles of anhydrous salt and moles of water → find the ratio ($x$)
Interactive: Hydrate Analysis
AvailableSimulate the classic determination of water of crystallization experiment.
→ Launch Simulation⚖️ Stoichiometry & Balancing
Stoichiometric Coefficients
The numbers in front of formulas in a balanced equation represent the mole ratio of reactants and products.
Example: $2Mg + O_2 \rightarrow 2MgO$ means 2 moles of Mg react with 1 mole of $O_2$ to produce 2 moles of MgO.
Steps for Stoichiometric Calculations
- Write the balanced equation
- Convert known quantity to moles ($n = m/M$ or $n = cV$ or $n = V/V_m$)
- Use the mole ratio from the balanced equation
- Convert moles of product to required quantity (mass, volume, etc.)
Limiting Reagent
The reactant that runs out first — it determines how much product is formed. The other reactant is in excess.
To identify the limiting reagent: Calculate moles of each reactant, then divide by their stoichiometric coefficient. The smallest value is the limiting reagent.
📊 Yields & Atom Economy
Percentage Yield
$$\text{Percentage yield} = \frac{\text{Experimental yield}}{\text{Theoretical yield}} \times 100\%$$
Theoretical yield: Maximum amount of product calculated from stoichiometry. Experimental yield: Actual amount obtained in the lab (always less due to losses and side reactions).
Why is Yield Always Less Than 100%?
- Incomplete reaction (equilibrium)
- Side reactions producing unwanted products
- Practical losses during transfer, filtration, evaporation
- Impurities in the reactants
Atom Economy
$$\text{Atom economy} = \frac{M_r(\text{desired product})}{\sum M_r(\text{all products})} \times 100\%$$
A measure of how many atoms from the reactants end up in the desired product. Higher atom economy = less waste = greener chemistry.
Interactive: Atom Economy Calculator
AvailableExplore atom economy for different reaction types.
→ Launch Tool🧪 Interactive Virtual Labs
Empirical Formula: Mg → MgO Crucible Experiment
AvailableSimulate the classic IB Chemistry practical — burn magnesium to determine the formula of magnesium oxide.
→ Launch Experiment🧠 Memory Aids & Mnemonics
🔤 The Mole Triangle
$n = \frac{m}{M}$ → "moles = mass ÷ molar mass"
Draw a triangle: $m$ on top, $n$ and $M$ on the bottom. Cover the one you want — the remaining two show the formula. Also works for $n = N/L$ and $n = cV$.
🔤 Empirical Formula Steps
"Percent → Moles → Divide → Round" (PMDR)
Always the same 4 steps: assume 100 g (so % → mass), divide by $M$, divide by smallest, round to whole numbers. If you get .5, multiply all by 2. If you get .33, multiply all by 3.
🔤 Limiting Reagent
"The one that runs out first wins (controls the reaction)"
Calculate moles ÷ coefficient for each reactant. The smaller value is the limiting reagent. Think of it like making sandwiches: if you have 10 slices of bread and 3 slices of cheese, cheese is limiting — you can only make 3 sandwiches.
🔤 Unit Conversion
$cm^3 \div 1000 = dm^3$ (always convert BEFORE calculating)
$1 \ dm^3 = 1000 \ cm^3 = 1 \ L$. For $n = cV$, concentration is in $mol/dm^3$ and volume MUST be in $dm^3$. Forgetting this conversion is the #1 calculation error.
🌍 Real-World Applications
💊 Pharmaceutical Manufacturing (Percentage Yield)
Context: When a pharmaceutical company manufactures aspirin, they need to calculate the theoretical yield to determine how much raw material to purchase.
Science: Aspirin synthesis: $C_7H_6O_3 + C_4H_6O_3 \rightarrow C_9H_8O_4 + CH_3COOH$. If starting with 10.0 g of salicylic acid ($M = 138 \ g/mol$), the theoretical yield of aspirin ($M = 180 \ g/mol$) is $\frac{10.0}{138} \times 180 = 13.0 \ g$. In practice, a good student achieves ~80% yield (~10.4 g). Pharmaceutical factories aim for >95% yield through optimized conditions.
Impact: Understanding percentage yield helps companies estimate costs, reduce waste, and comply with environmental regulations. Green chemistry focuses on maximizing atom economy to reduce byproducts.
🛞 Airbag Chemistry (Stoichiometry)
Context: Car airbags deploy in ~30 milliseconds. The bag inflates with nitrogen gas produced by a chemical reaction — the exact amount must be precisely calculated using stoichiometry.
Science: $2NaN_3 \rightarrow 2Na + 3N_2$. Sodium azide decomposes to produce nitrogen gas. For a 60 L airbag at STP: $n(N_2) = 60/22.7 = 2.64 \ mol$. From the 2:3 ratio: $n(NaN_3) = \frac{2}{3} \times 2.64 = 1.76 \ mol$. Mass needed: $1.76 \times 65.0 = 114 \ g$ of $NaN_3$. Too little → bag doesn't protect. Too much → bag ruptures.
Impact: This life-saving technology relies entirely on precise stoichiometric calculations — a real-world application of $n = m/M$ and mole ratios.
🧪 Forensic Blood Alcohol Testing (Titration Stoichiometry)
Context: Police use breathalyzer tests that rely on a redox titration with potassium dichromate to determine blood alcohol concentration (BAC).
Science: Ethanol reacts with acidified potassium dichromate: $3C_2H_5OH + 2K_2Cr_2O_7 + 8H_2SO_4 \rightarrow 3CH_3COOH + 2Cr_2(SO_4)_3 + 2K_2SO_4 + 11H_2O$. The color change from orange ($Cr_2O_7^{2-}$) to green ($Cr^{3+}$) indicates the presence and amount of alcohol. The stoichiometric ratio (3:2) allows precise calculation of how much alcohol was in the breath sample.
Impact: This reaction underpins most roadside breathalyzer tests. A BAC above 0.08% (800 mg/L in many countries) is illegal. The calculation relies on $n = cV$ and mole ratios from the balanced equation — core S1.4 skills.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Forgetting to convert $cm^3$ to $dm^3$: In $n = cV$, volume must be in $dm^3$. $250 \ cm^3 = 0.250 \ dm^3$. This is the single most common calculation error in IB Chemistry.
- Not using the mole ratio: Students find moles of the given substance but forget to use the ratio from the balanced equation to find the moles of the required substance.
- Rounding empirical formula ratios incorrectly: If you get 1.5, multiply everything by 2 (not round to 2). If you get 1.33, multiply everything by 3. Only round when very close to a whole number (e.g., 1.98 → 2).
- Confusing empirical and molecular formula: The molecular formula is always a whole number multiple of the empirical formula. $CH_2O$ (empirical) can be $C_2H_4O_2$ (molecular, acetic acid) or $C_6H_{12}O_6$ (molecular, glucose).
- Writing yield > 100%: This means there's an error — usually impure product (still contains water) or wrong mole ratio used.
📝 IB-Style Exam Questions
Question 1: Calculate the number of moles in 4.00 g of Sodium Hydroxide (NaOH). [2 marks]
Mark Scheme:
- [1 mark] $M(NaOH) = 22.99 + 16.00 + 1.01 = 40.00 \ g/mol$
- [1 mark] $n = 4.00 / 40.00 = 0.100 \ mol$
Question 2: A hydrocarbon contains 85.7% carbon and 14.3% hydrogen by mass. Determine its empirical formula. [3 marks]
Mark Scheme:
- [1 mark] Moles: C = 85.7/12.01 = 7.14; H = 14.3/1.01 = 14.16
- [1 mark] Ratio: 7.14 : 14.16 → divide by 7.14 → 1 : 1.98 ≈ 1 : 2
- [1 mark] Empirical formula = $CH_2$
Question 3: Calculate the percentage by mass of water in hydrated copper(II) sulfate, $CuSO_4 \cdot 5H_2O$. [2 marks]
Mark Scheme:
- [1 mark] $M_r = 63.55 + 32.07 + 64.00 + 90.10 = 249.72$. Mass of water = $5 \times 18.02 = 90.10$
- [1 mark] $\% = (90.10 / 249.72) \times 100 = 36.1\%$
Question 4: 5.0 mol of methane ($CH_4$) burns completely in excess oxygen. Calculate the mass of $CO_2$ produced. [2 marks]
Mark Scheme:
- [1 mark] $CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O$. Ratio is 1:1, so 5.0 mol $CO_2$ produced.
- [1 mark] Mass $= n \times M = 5.0 \times 44.01 = 220 \ g$
Question 5: Define the terms "theoretical yield" and "percentage yield". [2 marks]
Mark Scheme:
- [1 mark] Theoretical yield: the maximum amount of product obtainable from a given amount of limiting reagent, assuming 100% conversion.
- [1 mark] Percentage yield: $(\text{actual yield} / \text{theoretical yield}) \times 100\%$
Question 6: 2.43 g of magnesium reacts with excess hydrochloric acid. Calculate the volume of hydrogen gas produced at STP. [3 marks]
Mark Scheme:
- [1 mark] $Mg + 2HCl \rightarrow MgCl_2 + H_2$. $n(Mg) = 2.43/24.31 = 0.100 \ mol$
- [1 mark] Ratio 1:1, so $n(H_2) = 0.100 \ mol$
- [1 mark] $V = n \times V_m = 0.100 \times 22.7 = 2.27 \ dm^3$
Question 7: 3.0 g of carbon reacts with 16.0 g of oxygen. Determine the limiting reagent. [3 marks]
Mark Scheme:
- [1 mark] $C + O_2 \rightarrow CO_2$. $n(C) = 3.0/12.01 = 0.250 \ mol$. $n(O_2) = 16.0/32.00 = 0.500 \ mol$
- [1 mark] Ratio is 1:1 → C needs 0.250 mol $O_2$, but 0.500 is available
- [1 mark] Carbon is the limiting reagent (runs out first; oxygen is in excess).
Question 8: A compound has empirical formula $CH_2O$ and a molar mass of $180 \ g/mol$. Determine its molecular formula. [2 marks]
Mark Scheme:
- [1 mark] $M_r(CH_2O) = 12.01 + 2.02 + 16.00 = 30.03$. $n = 180/30.03 = 5.99 \approx 6$
- [1 mark] Molecular formula = $C_6H_{12}O_6$ (glucose)