📋 IB Content Statements (S1.5)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S1.5.1: An ideal gas consists of moving particles with negligible volume and no intermolecular forces. All collisions are perfectly elastic.
- S1.5.2: Real gases deviate from the ideal gas model at low temperature and high pressure.
- S1.5.3: The molar volume of an ideal gas at STP (273.15 K, 100 kPa) is $22.7 \ dm^3 \ mol^{-1}$.
- S1.5.4: The relationship between the pressure, volume, temperature, and amount of gas is shown in the ideal gas equation: $PV = nRT$.
- S1.5.5: The combined gas law is derived from the ideal gas equation and can be used to relate changing conditions.
🔹 The Ideal Gas Model
Assumptions of Kinetic Molecular Theory
An ideal gas is a theoretical model. It assumes:
- Gas particles have negligible volume compared to the container
- There are no intermolecular forces (no attraction or repulsion) between particles
- All collisions are perfectly elastic (no kinetic energy lost)
- Particles move in random, straight-line motion
- The average kinetic energy is proportional to absolute temperature ($E_k \propto T$)
Which gases behave most ideally? Noble gases (He, Ne) and small non-polar molecules ($H_2$) at high temperature and low pressure — because they have the weakest intermolecular forces and smallest particle size.
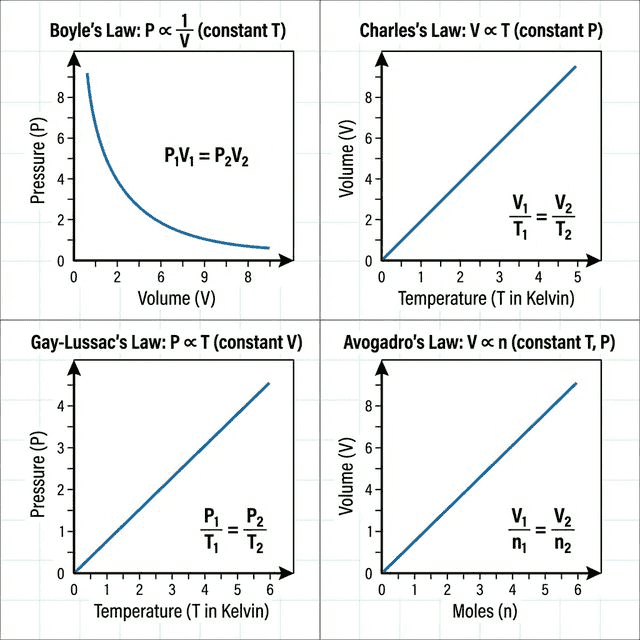
📈 The Gas Laws
| Law | Relationship | Constant | Equation | Graph Shape |
|---|---|---|---|---|
| Boyle's | $P \propto \frac{1}{V}$ | $T$, $n$ | $P_1V_1 = P_2V_2$ | Inverse curve (hyperbola) |
| Charles's | $V \propto T$ | $P$, $n$ | $\frac{V_1}{T_1} = \frac{V_2}{T_2}$ | Straight line through origin (in K) |
| Gay-Lussac's | $P \propto T$ | $V$, $n$ | $\frac{P_1}{T_1} = \frac{P_2}{T_2}$ | Straight line through origin (in K) |
| Avogadro's | $V \propto n$ | $P$, $T$ | $\frac{V_1}{n_1} = \frac{V_2}{n_2}$ | Straight line through origin |
⚠️ Critical: Temperature must always be in Kelvin ($K$) for all gas law calculations. $T(K) = T(°C) + 273$. Using °C will give wrong answers because the gas laws require an absolute scale.
⚗️ The Ideal Gas Equation
$$PV = nRT$$
| Variable | Symbol | SI Unit | Common Conversions |
|---|---|---|---|
| Pressure | $P$ | Pa (Pascals) | $1 \ kPa = 1000 \ Pa$; $1 \ atm = 101325 \ Pa$ |
| Volume | $V$ | $m^3$ | $1 \ dm^3 = 10^{-3} \ m^3$; $1 \ cm^3 = 10^{-6} \ m^3$ |
| Amount | $n$ | mol | $n = m/M$ |
| Gas constant | $R$ | $8.31 \ J \ K^{-1} \ mol^{-1}$ | Given in Data Booklet |
| Temperature | $T$ | K (Kelvin) | $T(K) = T(°C) + 273$ |
Finding Molar Mass from PV=nRT
Since $n = m/M$, substitute into $PV = nRT$:
$$M = \frac{mRT}{PV}$$
This is used to determine the molar mass of an unknown gas experimentally.
Combined Gas Law
When $n$ is constant (same amount of gas, changing conditions):
$$\frac{P_1 V_1}{T_1} = \frac{P_2 V_2}{T_2}$$
📦 Molar Volume at STP
Standard Temperature and Pressure (STP)
STP = 273.15 K (0°C) and 100 kPa
At STP, one mole of any ideal gas occupies $22.7 \ dm^3$.
$$n = \frac{V}{V_m} = \frac{V}{22.7}$$
⚠️ Old vs New STP: Some older textbooks use 1 atm (101.325 kPa) and quote $V_m = 22.4 \ dm^3$. The IB 2025 syllabus uses 100 kPa and $22.7 \ dm^3$. Always use the value in your Data Booklet.
When to use molar volume vs $PV = nRT$:
- Use $n = V/22.7$ when conditions are at STP (quick and easy)
- Use $PV = nRT$ when conditions are not at STP (general case)
💨 Real Gases vs Ideal Gases
| Feature | Ideal Gas | Real Gas |
|---|---|---|
| Particle volume | Zero (negligible) | Finite (particles occupy space) |
| IMFs | None | Present (London forces, dipole-dipole, H-bonds) |
| Collisions | Perfectly elastic | Some energy transfer during collisions |
| Obeys $PV = nRT$ | Always | Only approximately (best at high T, low P) |
When Do Real Gases Deviate Most?
- Low temperature: Particles move slowly → IMFs become significant → particles attract each other → actual pressure is lower than predicted
- High pressure: Particles forced close together → their volume becomes significant → actual volume is greater than predicted
Key exam point: At low T and high P, intermolecular forces and particle volume can no longer be ignored → the ideal gas model breaks down. Gases with stronger IMFs (like $NH_3$, $H_2O$) deviate more than those with weaker IMFs (like $He$, $Ne$).
🧪 Interactive Virtual Labs
Interactive: Ideal Gas Law Explorer
AvailableAdjust P, V, n, and T to see how they relate through $PV = nRT$.
→ Launch SimulationGas Collection & Rate of Reaction
AvailableCollect gas over water and measure reaction rates using gas volumes.
→ Launch Experiment🧠 Memory Aids & Mnemonics
🔤 PV = nRT Units
"Pa, m³, mol, 8.31, K" → all SI units
If you use Pa and $m^3$, $R = 8.31$. If using kPa, use $dm^3$ (they cancel correctly). The most common error: mixing $cm^3$ with Pa or forgetting to convert temperature to Kelvin.
🔤 Real Gas Deviations
"Low T, High P = Bad for Ideal"
Low temperature → particles slow → IMFs matter. High pressure → particles close → volume matters. The opposite conditions (high T, low P) make gases behave most ideally.
🔤 Three Gas Laws
"BCA" → Boyle (P∝1/V), Charles (V∝T), Avogadro (V∝n)
Boyle is the inverse one (squeeze → pressure up). Charles is the linear one (heat → volume up). Avogadro is the moles one (more gas → more volume).
🔤 STP Values
"0°C, 100 kPa, 22.7 dm³"
STP = Standard Temperature and Pressure. Temperature = 273 K (0°C). Pressure = 100 kPa. Molar volume = 22.7 dm³. These three numbers go together. Memorize them as a triple.
🌍 Real-World Applications
🎈 Hot Air Balloons (Charles's Law)
Context: Hot air balloons rise because heating the air inside makes it expand, reducing its density below the surrounding cooler air.
Science: Charles's Law: $V \propto T$ (at constant P). When the burner heats air inside the balloon from 20°C (293 K) to 100°C (373 K), the volume increases by a factor of $373/293 = 1.27$ — a 27% expansion. Since the same mass of air now occupies more volume, the density drops. When the density of the air inside is less than the air outside, the balloon experiences buoyancy and rises.
Impact: Hot air balloons were the first successful human flight technology (1783, Montgolfier brothers). The same principle applies to convection currents in weather systems — warm air rises, creating wind patterns.
🛞 Tire Pressure Warning (Gay-Lussac's Law)
Context: Your car's tire pressure sensor often triggers a warning on cold mornings. Why?
Science: $P \propto T$ at constant V (rigid tire). On a cold night, the temperature drops from 25°C (298 K) to 0°C (273 K). If initial pressure is 220 kPa: $P_2 = 220 \times (273/298) = 201 \ kPa$. This ~9% pressure drop can trigger monitoring systems (usually set at ~200 kPa). After driving, friction heats the tires back up, and the pressure recovers.
Impact: Correctly inflated tires improve fuel efficiency by ~3% and reduce accident risk. Understanding gas laws helps explain why tire pressure varies with season and driving conditions.
🤿 Scuba Diving (Boyle's Law)
Context: Why can't scuba divers ascend too quickly? The answer is Boyle's Law.
Science: $P \propto 1/V$. At 30 m depth, the pressure is ~4 atm (3 atm water + 1 atm air). A diver breathing at this depth has air in their lungs at 4 atm pressure. If they ascend rapidly to the surface (1 atm) without exhaling, the gas volume would quadruple ($V_2 = V_1 \times 4/1$), rupturing lung tissue. Additionally, dissolved nitrogen comes out of solution as bubbles ("the bends" / decompression sickness), which can be fatal.
Impact: Dive tables and computers calculate safe ascent rates using gas law principles. Nitrox (enriched air with lower $N_2$) reduces decompression risk — a direct application of Dalton's Law of partial pressures.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Using °C instead of K: ALL gas law calculations require Kelvin. $T(K) = T(°C) + 273$. Using Celsius gives completely wrong answers. This is the #1 error in gas calculations.
- Unit mismatch in PV = nRT: If using $R = 8.31$, then $P$ must be in Pa and $V$ in $m^3$. Alternatively, $P$ in kPa and $V$ in $dm^3$ also works with $R = 8.31$. Never mix kPa with $m^3$ or Pa with $dm^3$.
- Converting $cm^3$ to $m^3$: $1 \ cm^3 = 10^{-6} \ m^3$ (divide by $10^6$, not 1000). Students often confuse $cm^3 \to dm^3$ ($\div 1000$) with $cm^3 \to m^3$ ($\div 10^6$).
- Confusing STP molar volume values: IB 2025 uses $22.7 \ dm^3$ at $100 \ kPa$. Older resources quote $22.4 \ dm^3$ at $101.3 \ kPa$. Use 22.7.
- Forgetting which variable is constant: Each gas law holds different variables constant. Read the question carefully to identify which variables are changing and which are fixed.
📝 IB-Style Exam Questions
Question 1: Calculate the volume occupied by 0.500 mol of an ideal gas at 300 K and 100 kPa. [2 marks]
Mark Scheme:
- [1 mark] $V = nRT/P = (0.500 \times 8.31 \times 300) / 100$ (using kPa and $dm^3$)
- [1 mark] $V = 12.5 \ dm^3$ (or $0.0125 \ m^3$)
Question 2: Under what conditions do real gases deviate most from ideal behavior? Explain why. [2 marks]
Mark Scheme:
- [1 mark] Low temperature and high pressure
- [1 mark] At low T, particles move slowly so IMFs become significant. At high P, particles are close together so their volume becomes significant.
Question 3: State Avogadro's Law. [1 mark]
Mark Scheme:
- [1 mark] Equal volumes of gases, at the same temperature and pressure, contain the same number of particles/moles.
Question 4: A gas sample has a volume of $2.0 \ dm^3$ at $25°C$. Calculate its volume at $50°C$ if pressure remains constant. [2 marks]
Mark Scheme:
- [1 mark] Convert: $T_1 = 298 \ K$, $T_2 = 323 \ K$
- [1 mark] $V_2 = V_1 \times T_2/T_1 = 2.0 \times 323/298 = 2.17 \ dm^3$
Question 5: Calculate the molar mass of a gas if 0.200 g occupies $100 \ cm^3$ at $27°C$ and 100 kPa. [3 marks]
Mark Scheme:
- [1 mark] Convert: $V = 100 \ cm^3 = 0.100 \ dm^3$; $T = 300 \ K$
- [1 mark] $n = PV/RT = (100 \times 0.100)/(8.31 \times 300) = 0.00401 \ mol$
- [1 mark] $M = m/n = 0.200/0.00401 = 49.9 \ g \ mol^{-1}$
Question 6: State three assumptions of the kinetic molecular theory for an ideal gas. [3 marks]
Mark Scheme:
- [1 mark] Gas particles have negligible volume / are point particles
- [1 mark] No intermolecular forces / attractions between particles
- [1 mark] Collisions are perfectly elastic / no kinetic energy is lost during collisions
Question 7: Calculate the volume of $CO_2$ gas produced at STP when 10.0 g of $CaCO_3$ decomposes completely. [3 marks]
Mark Scheme:
- [1 mark] $CaCO_3 \rightarrow CaO + CO_2$. $n(CaCO_3) = 10.0/100.09 = 0.0999 \ mol$
- [1 mark] 1:1 ratio, so $n(CO_2) = 0.0999 \ mol$
- [1 mark] $V = n \times V_m = 0.0999 \times 22.7 = 2.27 \ dm^3$
Question 8: Explain why $NH_3$ deviates more from ideal gas behavior than $He$ under the same conditions. [2 marks]
Mark Scheme:
- [1 mark] $NH_3$ has stronger intermolecular forces (hydrogen bonding + dipole-dipole) compared to He (only weak London dispersion forces).
- [1 mark] $NH_3$ molecules are larger, so their volume is more significant relative to the container / $NH_3$ particles are more likely to attract each other, reducing the actual pressure below the ideal value.