📋 IB Content Statements (S2.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S2.1.1: When metal atoms lose electrons, they form positive ions (cations). When non-metal atoms gain electrons, they form negative ions (anions).
- S2.1.2: The ionic bond is due to electrostatic attraction between oppositely charged ions.
- S2.1.3: Ionic compounds exist as three-dimensional lattice structures. The ratio of ions in the lattice is determined by the charges on the ions.
- S2.1.4: Ionic compounds have high melting points, and conduct electricity when molten or dissolved, but not when solid.
HL Extensions
- S2.1.5: Lattice enthalpy is a measure of the strength of the ionic bond in an ionic compound.
- S2.1.6: Born-Haber cycles can be used to calculate lattice enthalpy and other enthalpy changes that cannot be measured directly.
- S2.1.7: The difference between theoretical and experimental lattice enthalpies provides evidence for covalent character in some ionic compounds.
🔹 Formation of Ions
Ionic Bonding
The electrostatic attraction between oppositely charged ions. Typically formed between a metal (Group 1, 2, 3 — loses electrons → cation) and a non-metal (Group 15, 16, 17 — gains electrons → anion).
Common Ion Charges
| Group | Examples | Ion Charge | Electron Configuration |
|---|---|---|---|
| Group 1 | $Na$, $K$, $Li$ | $1+$ | Lose 1 electron → noble gas config |
| Group 2 | $Mg$, $Ca$, $Ba$ | $2+$ | Lose 2 electrons → noble gas config |
| Group 13 | $Al$ | $3+$ | Lose 3 electrons → noble gas config |
| Group 16 | $O$, $S$ | $2-$ | Gain 2 electrons → noble gas config |
| Group 17 | $F$, $Cl$, $Br$ | $1-$ | Gain 1 electron → noble gas config |
| Transition metals | $Fe$, $Cu$ | Variable ($2+$, $3+$) | Lose different numbers of d/s electrons |
Writing ionic formulas: The charges must balance to zero. For example, $Al^{3+}$ and $O^{2-}$: need 2 of $Al$ and 3 of $O$ → $Al_2O_3$. Cross-over method: the charge of one ion becomes the subscript of the other.
🔗 Polyatomic Ions
You must memorize these polyatomic ions for IB exams:
| Name | Formula | Charge | Found In |
|---|---|---|---|
| Ammonium | $NH_4^+$ | $+1$ | Fertilizers ($NH_4NO_3$) |
| Hydroxide | $OH^-$ | $-1$ | Bases ($NaOH$) |
| Nitrate | $NO_3^-$ | $-1$ | Fertilizers, explosives |
| Hydrogencarbonate | $HCO_3^-$ | $-1$ | Baking soda ($NaHCO_3$) |
| Carbonate | $CO_3^{2-}$ | $-2$ | Limestone ($CaCO_3$) |
| Sulfate | $SO_4^{2-}$ | $-2$ | Plaster of Paris ($CaSO_4$) |
| Phosphate | $PO_4^{3-}$ | $-3$ | Fertilizers, bones |
🧱 Ionic Lattice & Physical Properties
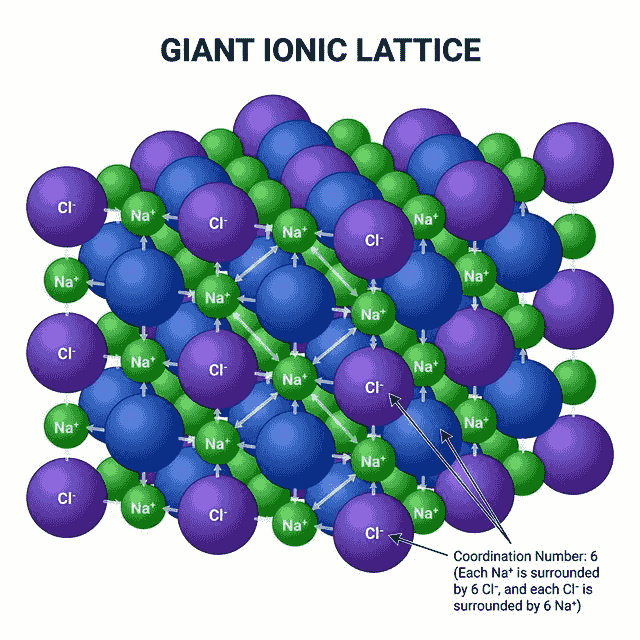
Giant Ionic Lattice Structure
Ionic compounds form 3D lattice structures — a regular arrangement of alternating positive and negative ions extending in all directions. There are no individual molecules — this is why we use formula units (e.g., NaCl) rather than molecular formulas.
Properties Explained
| Property | Observation | Explanation |
|---|---|---|
| Melting point | Very high (often >500°C) | Strong electrostatic forces between many ions — large amount of energy needed to break the lattice |
| Hardness | Hard but brittle | Hard: strong bonds. Brittle: if layers shift, like-charges align → repulsion → crystal shatters |
| Solubility | Generally soluble in water | Polar water molecules surround ions (ion-dipole interactions) → hydration energy overcomes lattice energy |
| Conductivity (solid) | Does NOT conduct | Ions in fixed positions — cannot move to carry charge |
| Conductivity (molten) | Conducts | Ions free to move — act as mobile charge carriers |
| Conductivity (aqueous) | Conducts | Ions dissociate in water — free to move and carry charge |
🔥 Factors Affecting Melting Point
The strength of the ionic bond (and therefore the melting point) depends on:
| Factor | Effect | Example |
|---|---|---|
| Ionic charge | Higher charge → stronger attraction → higher MP | $MgO$ (2+/2−) MP 2852°C > $NaCl$ (1+/1−) MP 801°C |
| Ionic radius | Smaller ions → closer together → stronger attraction → higher MP | $LiF$ MP 845°C > $CsI$ MP 621°C |
Coulomb's Law (qualitative): The electrostatic force between ions is proportional to the product of the charges and inversely proportional to the square of the distance: $F \propto \frac{q^+ \times q^-}{r^2}$. When comparing melting points, always mention both charge and size.
⚡ Born-Haber Cycles (HL)
Lattice Enthalpy ($\Delta H_{lat}$)
The enthalpy change when one mole of an ionic compound is formed from its gaseous ions under standard conditions. It is always exothermic (negative) and is a measure of ionic bond strength.
$$M^+(g) + X^-(g) \rightarrow MX(s) \quad \Delta H_{lat} < 0$$
Born-Haber Cycle Steps (for NaCl)
| Step | Process | Enthalpy Term | Sign |
|---|---|---|---|
| 1 | $Na(s) \rightarrow Na(g)$ | Atomization of Na | Endothermic (+) |
| 2 | $Na(g) \rightarrow Na^+(g) + e^-$ | 1st Ionization Energy | Endothermic (+) |
| 3 | $\frac{1}{2}Cl_2(g) \rightarrow Cl(g)$ | Atomization of Cl | Endothermic (+) |
| 4 | $Cl(g) + e^- \rightarrow Cl^-(g)$ | 1st Electron Affinity | Exothermic (−) |
| 5 | $Na^+(g) + Cl^-(g) \rightarrow NaCl(s)$ | Lattice Enthalpy | Exothermic (−) |
Hess's Law application: $\Delta H_f = \Delta H_{at}(Na) + IE_1(Na) + \Delta H_{at}(Cl) + EA_1(Cl) + \Delta H_{lat}$. Rearrange to find any unknown step.
Theoretical vs Experimental Lattice Enthalpy
If the experimental lattice enthalpy is more negative than the theoretical value, this suggests some covalent character in the ionic bond (the bonding is stronger than a purely ionic model predicts). This occurs when cations are small and highly charged (high charge density → polarizing power), and anions are large and easily polarized.
Interactive: Born-Haber Cycle Builder
AvailableBuild a Born-Haber cycle step-by-step and calculate lattice enthalpy.
→ Launch Simulation🧪 Interactive Virtual Labs
3D Ionic Lattice Explorer
AvailableVisualize the NaCl crystal lattice in 3D — rotate, zoom, and examine the structure.
→ Launch Visualization🧠 Memory Aids & Mnemonics
🔤 Cation vs Anion
"Cats are Positive" (cation = positive)
Cations are positive (like a cat with a + sign). Anions are negative (a-n-ion: "a negative ion"). Metals form cations. Non-metals form anions.
🔤 Cross-Over Method
$Al^{3+}$ and $O^{2-}$ → $Al_2O_3$
The charge of one ion becomes the subscript of the other (ignoring the sign). This automatically balances the formula. Always simplify: $Ca^{2+}$ and $O^{2-}$ → $Ca_1O_1$ → $CaO$ (not $Ca_2O_2$).
🔤 Polyatomic Ion Charges
"Nick the Camel had a Clam for Supper in Phoenix"
Nitrate ($NO_3^-$, 1−), Carbonate ($CO_3^{2-}$, 2−), Clam = Chlorate (not needed for IB), Sulfate ($SO_4^{2-}$, 2−), Phosphate ($PO_4^{3-}$, 3−). The number of syllables = the charge (minus).
🔤 Properties of Ionic Compounds
"High MP, Hard-Brittle, Solid=no, Liquid=yes, Aqueous=yes"
Melting point: HIGH ($> 500°C$). Hardness: hard but BRITTLE. Conductivity: NO as solid (ions fixed), YES as molten/aqueous (ions free to move). Solubility: generally YES in water (polar solvent).
🔤 Born-Haber Steps
"All Iron And Each Lock" = Atomize, Ionize, Atomize, EA, Lattice
Steps for building a Born-Haber cycle: Atomize the metal → Ionize the metal → Atomize the non-metal → Electron affinity (non-metal gains e⁻) → Lattice enthalpy (ions combine).
🌍 Real-World Applications
⚡ Electrolytes in Sports Drinks
Context: Sports drinks like Gatorade/Pocari Sweat advertise "electrolytes" for hydration. What exactly are these?
Science: "Electrolytes" are dissolved ionic compounds — $Na^+$, $K^+$, $Ca^{2+}$, $Mg^{2+}$, and $Cl^-$ ions in water. When you sweat, you lose these ions. They are essential for nerve impulses (electrical signals travel as ion gradients, $Na^+/K^+$ pump), muscle contraction ($Ca^{2+}$ triggers muscle fibers), and maintaining osmotic balance (water follows ions). Severe electrolyte imbalance can cause muscle cramps, irregular heartbeat, or even death.
Impact: This is a direct application of S2.1 — ionic compounds dissolve in water to produce free ions. The ability of dissolved ionic compounds to conduct electricity is used in medical diagnostics (ECG/EKG measures heart muscle ion flow).
🧂 Road Salt & Freezing Point Depression
Context: In winter, cities spread $NaCl$ (rock salt) or $CaCl_2$ on icy roads. Why does this melt the ice?
Science: When $NaCl$ dissolves in the thin water layer on ice, it dissociates into $Na^+$ and $Cl^-$ ions. These ions disrupt the water's crystal lattice (ice structure), lowering the freezing point — this is colligative property freezing point depression. $NaCl$ produces 2 ions per formula unit, lowering the freezing point to about $-21°C$. $CaCl_2$ produces 3 ions ($Ca^{2+}$ + $2Cl^-$) and can work down to $-29°C$, making it more effective in extreme cold.
Impact: Over 20 million tonnes of salt are used on roads annually in the US alone. This application connects to S2.1 (ionic dissociation) and colligative properties.
💎 Ceramics & Refractory Materials
Context: Why are ionic compounds like $MgO$ and $Al_2O_3$ used to line furnaces and make heat-resistant tiles?
Science: $MgO$ has ions with charges $2+$ and $2-$, and both are small → extremely strong electrostatic attraction → melting point of 2852°C. $Al_2O_3$ (alumina) melts at 2072°C. These materials can withstand the intense heat inside steel furnaces ($\sim$1600°C) without melting. Their hardness (strong lattice) combined with brittleness (ionic materials fracture along crystal planes) makes them ideal for applications where thermal and chemical resistance matters more than flexibility.
Impact: Space shuttle heat tiles used alumina-based ceramics. This directly illustrates how ionic charge and size (Coulomb's Law) determine real-world thermal properties.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Saying "ionic molecules": Ionic compounds do NOT form molecules. They form giant lattice structures. The formula ($NaCl$) represents the simplest ratio of ions, not a molecule. Never use "molecule" for ionic compounds.
- Confusing melting and conducting: Melting requires breaking the lattice (overcoming electrostatic forces). Conducting requires ions to be free to move. These are related but different concepts — always specify which you're explaining.
- Forgetting to mention "ions" in conductivity: When explaining conductivity, you must say "ions are free to move" (not just "particles" or "it can move"). The examiner needs the word "ions" for the mark.
- Not balancing charges correctly: $Al^{3+}$ and $SO_4^{2-}$: need to balance 3+ and 2−. LCM = 6, so need 2 Al and 3 sulfate → $Al_2(SO_4)_3$ (brackets around polyatomic ion).
- Confusing lattice enthalpy direction: Formation of the lattice from gaseous ions is exothermic ($\Delta H < 0$). Breaking the lattice (dissociation) is endothermic. Check which definition the question uses.
📝 IB-Style Exam Questions
Question 1: Explain why $CaO$ has a higher melting point than $KCl$. [3 marks]
Mark Scheme:
- [1 mark] $Ca^{2+}$ and $O^{2-}$ have higher charges than $K^+$ and $Cl^-$
- [1 mark] $Ca^{2+}$ is smaller than $K^+$ (and $O^{2-}$ smaller than $Cl^-$), so ions are closer together
- [1 mark] Therefore stronger electrostatic attraction / more energy required to break the lattice
Question 2: State the formula of the compound formed between Magnesium and Nitrogen. [1 mark]
Mark Scheme:
- [1 mark] $Mg_3N_2$ ($Mg^{2+}$ and $N^{3-}$: cross-over gives $Mg_3N_2$)
Question 3: Describe the structure and bonding in solid sodium chloride. [3 marks]
Mark Scheme:
- [1 mark] Giant/3D ionic lattice structure
- [1 mark] Regular arrangement of alternating $Na^+$ and $Cl^-$ ions
- [1 mark] Held together by strong electrostatic forces of attraction between oppositely charged ions

Question 4: Explain why ionic compounds conduct electricity when molten but not when solid. [2 marks]
Mark Scheme:
- [1 mark] In the solid, ions are in fixed positions in the lattice and cannot move to carry charge
- [1 mark] When molten, ions are free to move and act as charge carriers
Question 5: Define the term "lattice enthalpy". [2 marks]
Mark Scheme:
- [1 mark] The enthalpy change when one mole of an ionic compound is formed (from gaseous ions)
- [1 mark] Under standard conditions / from infinitely separated gaseous ions
Question 6: Write the formula for: (a) aluminium sulfate, (b) calcium phosphate. [2 marks]
Mark Scheme:
- [1 mark] (a) $Al_2(SO_4)_3$ ($Al^{3+}$ and $SO_4^{2-}$: LCM 6 → 2 Al, 3 sulfate)
- [1 mark] (b) $Ca_3(PO_4)_2$ ($Ca^{2+}$ and $PO_4^{3-}$: LCM 6 → 3 Ca, 2 phosphate)
Question 7: Explain why ionic compounds are generally soluble in water but not in non-polar solvents. [2 marks]
Mark Scheme:
- [1 mark] Water is a polar molecule — it can form ion-dipole interactions with the ions, surrounding and separating them (hydration)
- [1 mark] Non-polar solvents cannot form these interactions — the hydration energy is not sufficient to overcome the lattice enthalpy
Question 8: The experimental lattice enthalpy of $AgI$ is significantly more negative than its theoretical value. Explain what this suggests. [2 marks]
Mark Scheme:
- [1 mark] The bonding in $AgI$ has significant covalent character / is not purely ionic
- [1 mark] $Ag^+$ has high charge density / polarizing power, and $I^-$ is large and easily polarized → electron cloud distorted → shared electron density