📋 IB Content Statements (S2.3)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S2.3.1: A metallic bond is the electrostatic attraction between a lattice of cations and delocalized electrons.
- S2.3.2: The strength of a metallic bond depends on the charge of the ions and the radius of the metal ion.
- S2.3.3: Transition metals have properties associated with their ability to form ions with variable charges, complex ions, and act as catalysts.
- S2.3.4: Alloys are mixtures of a metal with other metals or non-metals. Alloys are generally harder than the component metals.
🔹 Metallic Bonding
Definition
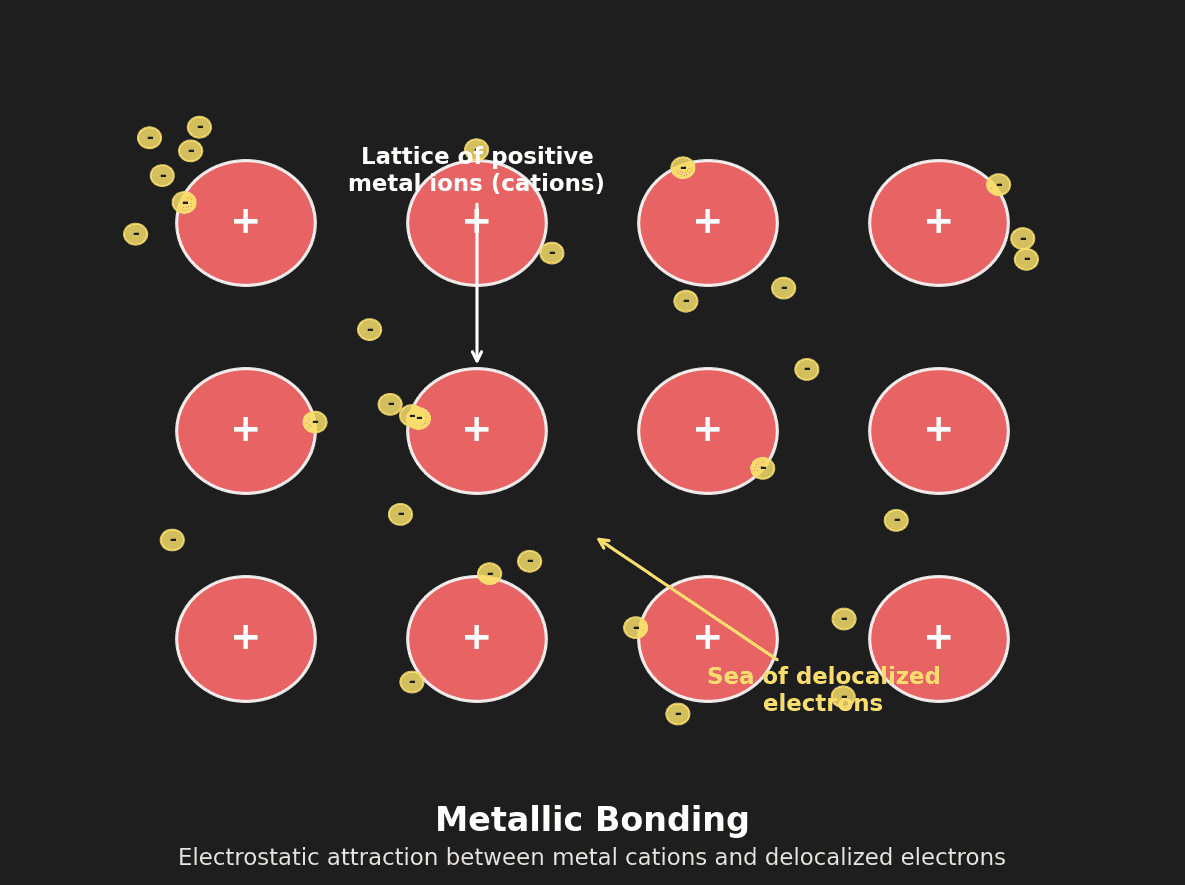
The electrostatic attraction between a lattice of positive metal ions (cations) and a sea of delocalized electrons.
When metal atoms pack together, their valence electrons become detached from individual atoms and are free to move throughout the entire structure. The atoms become positive ions (cations) arranged in a regular lattice.
Structure: Cations fixed in lattice positions, electrons free to move throughout the structure.
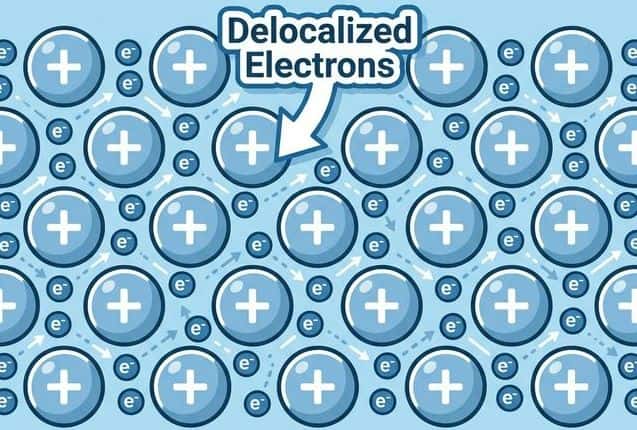
Key point for exams: Metallic bonding is non-directional — the attraction is equal in all directions. This is why layers can slide past each other (malleability) without breaking the bonding.
⚡ Properties of Metals
📺 S2.3.1 — Electrical, Thermal Conductivity and Malleability
| Property | Observation | Explanation (Metallic Bonding) |
|---|---|---|
| Electrical conductivity | Good conductors (solid and liquid) | Delocalized electrons are free to move and carry charge through the lattice |
| Thermal conductivity | Good heat conductors | Delocalized electrons transfer kinetic energy rapidly through the structure |
| Malleability | Can be hammered into sheets | Layers of cations can slide over each other without breaking the non-directional bonding — electrons adjust position |
| Ductility | Can be drawn into wires | Same as malleability — layers slide; the electron sea maintains bonding in the new shape |
| Lustre (shine) | Shiny when polished | Delocalized electrons on the surface absorb and re-emit light at all visible wavelengths |
| High melting/boiling points | Generally high (exceptions: Hg, Ga, Cs) | Strong electrostatic attraction between cations and electron sea requires a lot of energy to overcome |
⚠️ Contrast with Ionic: Ionic compounds are brittle because sliding layers brings like-charged ions next to each other → electrostatic repulsion → the crystal shatters. Metals are malleable because the bonding is non-directional.
💪 Strength of the Metallic Bond
The strength of metallic bonding (and therefore melting point) depends on two factors:
| Factor | Effect | Example |
|---|---|---|
| Number of delocalized electrons | More electrons per atom → stronger attraction | $Mg$ (2 e⁻) has higher MP than $Na$ (1 e⁻) |
| Charge density of cation | Smaller cation radius + higher charge → stronger attraction | $Al^{3+}$ (small, 3+) is stronger than $K^+$ (large, 1+) |
Trends:
- Across Period 3 (Na → Al): MP increases. Charge increases ($1+ → 2+ → 3+$), radius decreases → stronger attraction.
- Down Group 1 (Li → Cs): MP decreases. Cation gets bigger → charge density decreases → weaker attraction.
| Metal | Cation | Delocalized e⁻ | MP (°C) | Explanation |
|---|---|---|---|---|
| $Na$ | $Na^+$ | 1 | 98 | Large cation, only 1 e⁻ → weak bonding |
| $Mg$ | $Mg^{2+}$ | 2 | 650 | Smaller cation, 2 e⁻ → stronger |
| $Al$ | $Al^{3+}$ | 3 | 660 | Smallest cation, 3 e⁻ → strongest in period |
| $K$ | $K^+$ | 1 | 64 | Larger than Na⁺ → weaker than Na |
🔩 Alloys
Definition
An alloy is a mixture of a metal with one or more other elements (metals or non-metals). Alloys are generally harder and stronger than pure metals.
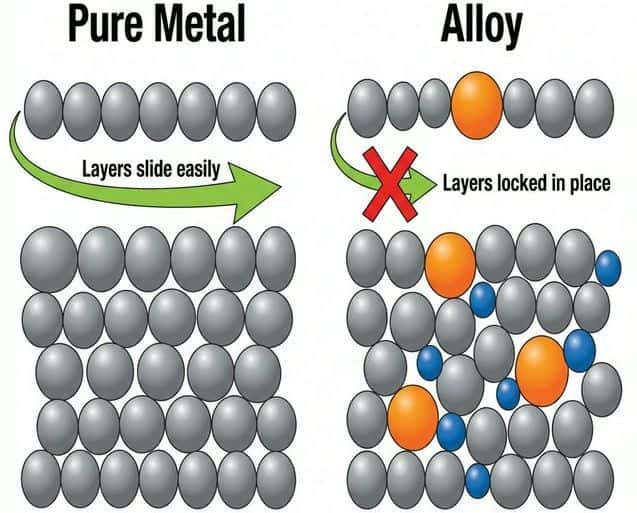
Why are alloys harder?
Different-sized atoms disrupt the regular lattice. This prevents layers of ions from sliding over each other easily. The irregularity "locks" the layers in place.
| Alloy | Components | Key Property | Use |
|---|---|---|---|
| Steel | Iron + Carbon (0.2–2%) | Much harder and stronger than pure iron | Construction, bridges, buildings |
| Stainless Steel | Iron + Chromium + Nickel | Corrosion resistant | Cutlery, medical instruments |
| Brass | Copper + Zinc | Corrosion resistant, good acoustic properties | Musical instruments, fittings |
| Bronze | Copper + Tin | Hard, resistant to saltwater corrosion | Sculptures, medals, ship propellers |
| Solder | Tin + Lead (or Tin + Silver) | Low melting point | Joining electronic components |
🧠 Memory Aids
🔤 "SEA" — What makes a metallic bond?
- Sea of delocalized electrons
- Electrostatic attraction
- Array of cations (positive ions in a lattice)
🔤 "MEDALS" — Properties of Metals
- Malleable (hammered into sheets)
- Electrically conductive
- Ductile (drawn into wires)
- Attractive lustre (shiny)
- Large melting points (generally)
- Sonorous (ring when hit)
🔤 MP Trends — "Charge Up, Size Down"
Higher charge + smaller radius = stronger metallic bond = higher melting point. Think: "More electrons, tighter grip, higher melt."
🔤 Alloys — "Different sizes = can't slide"
Imagine trying to slide a tray of identically-sized oranges (pure metal) vs a tray of mixed oranges and apples (alloy). The mixed sizes jam up and can't slide.
🌍 Real-World Applications
🏗️ Steel in Construction
Context: Modern skyscrapers and bridges rely on steel (iron + carbon alloy) for their structural frameworks.
Science: Carbon atoms are smaller than iron atoms and fit into the gaps in the iron lattice. This disrupts the regular arrangement, preventing layers from sliding → dramatically increasing hardness and tensile strength.
Impact: Steel is ~1000× harder than pure iron, making it the backbone of modern infrastructure. Different carbon percentages create different types: mild steel (0.2%), medium carbon steel (0.3–0.6%), high carbon steel (0.6–2%).
💻 Copper Wiring in Electronics
Context: Virtually all electrical wiring in homes and electronics uses copper.
Science: Copper has excellent electrical conductivity (second only to silver) because its single delocalized electron per atom moves very freely. Copper is also highly ductile — it can be drawn into thin wires without breaking.
Impact: The combination of conductivity + ductility + relatively low cost makes copper the ideal wiring material. Silver conducts better but is too expensive.
✈️ Aluminium Alloys in Aviation
Context: Aircraft fuselages are built from aluminium alloys (duralumin: Al + Cu + Mg + Mn).
Science: Pure aluminium is lightweight but too soft for aircraft. Adding copper, magnesium, and manganese atoms of different sizes disrupts the lattice, making duralumin 3× stronger while retaining aluminium's low density.
Impact: Aircraft need high strength-to-weight ratio. Duralumin provides this — it's as strong as steel but only one-third the weight.
⚠️ Common Mistakes
- ❌ "Metallic bonds are between atoms" → ✅ Metallic bonds are between cations (positive ions) and a sea of delocalized electrons. The atoms have lost their valence electrons.
- ❌ "Metals conduct because electrons move between atoms" → ✅ Must say "delocalized electrons are free to move through the lattice, carrying charge" (or kinetic energy for thermal conductivity).
- ❌ "Alloys are harder because they have stronger bonds" → ✅ Alloys are harder because different-sized atoms disrupt the regular lattice, preventing layers from sliding. The bonding itself is not necessarily stronger.
- ❌ Forgetting to mention "non-directional" → ✅ Always state that metallic bonding is non-directional when explaining malleability. This is what distinguishes it from ionic bonding (where sliding causes shattering).
- ❌ "Metals have high MP because of strong bonds" → ✅ Must explain why the bonds are strong: charge of cation, number of delocalized electrons, and ionic radius.
📝 Exam-Style Questions
Question 1: Describe metallic bonding. [2 marks]
Mark Scheme:
- [1 mark] Electrostatic attraction between a lattice of positive ions (cations)...
- [1 mark] ...and delocalized electrons.
Question 2: Explain why Magnesium has a higher melting point than Sodium. [3 marks]
Mark Scheme:
- [1 mark] Mg has a higher ionic charge ($2+$) than Na ($1+$).
- [1 mark] Mg contributes two delocalized electrons per atom, Na only one.
- [1 mark] Stronger electrostatic attraction between the cations and the electron sea in Mg → more energy required to break the bonds.
Question 3: Explain why metals are malleable while ionic compounds are brittle. [3 marks]
Mark Scheme:
- [1 mark] In metals, bonding is non-directional.
- [1 mark] Layers of cations can slide over each other while the delocalized electrons adjust position and maintain bonding.
- [1 mark] In ionic compounds, sliding layers brings ions of like charge together, causing electrostatic repulsion → crystal shatters.
Question 4: Explain why adding carbon to iron to make steel increases its hardness. [2 marks]
Mark Scheme:
- [1 mark] Carbon atoms are a different size to iron atoms.
- [1 mark] This disrupts the regular lattice, preventing layers from sliding over each other.
Question 5: Predict and explain which has a higher melting point: Aluminium or Sodium. [3 marks]
Mark Scheme:
- [1 mark] Aluminium has a higher melting point.
- [1 mark] $Al^{3+}$ has a higher charge and smaller radius than $Na^+$, giving it higher charge density.
- [1 mark] Al contributes 3 delocalized electrons vs 1 for Na → stronger electrostatic attraction.
Question 6: Suggest why Aluminium is a better electrical conductor than Magnesium. [1 mark]
Mark Scheme:
- [1 mark] Al has more delocalized electrons per atom (3 vs 2), meaning more charge carriers are available to conduct electricity.
Question 7: Explain why the melting point of metals decreases down Group 1. [2 marks]
Mark Scheme:
- [1 mark] The cation radius increases down the group.
- [1 mark] Larger cation → lower charge density → weaker electrostatic attraction between the cation and the delocalized electrons.
Question 8: Explain why metals can conduct heat but ionic solids cannot. [2 marks]
Mark Scheme:
- [1 mark] In metals, delocalized electrons can transfer kinetic energy rapidly through the structure.
- [1 mark] In ionic solids, there are no free-moving electrons — ions are fixed in position and can only transfer energy slowly via vibrations.