📋 IB Content Statements (S2.4)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S2.4.1: Bonding is best described as a continuum between ionic, covalent, and metallic models.
- S2.4.2: The type of bonding present can be deduced from the position of the elements in the periodic table.
- S2.4.3: Polymers are large molecules made from repeating subunits called monomers. The properties of polymers depend on the type of intermolecular forces.
- S2.4.4: Addition polymers form from unsaturated monomers. The repeating unit can be deduced from the monomer structure.
- S2.4.5: Condensation polymers form when monomers join with the loss of a small molecule (e.g., water). Polyesters and polyamides are key examples.
- S2.4.6: Alloys and composites are examples of mixtures with useful properties.
🔹 The Bonding Continuum
Key Insight
Bonding is not three separate categories — it exists on a continuum. Most bonds have some ionic character AND some covalent character. The position on the continuum depends on the electronegativity difference between the atoms.
| $\Delta\chi$ Range | Bond Character | Example | Structure Type |
|---|---|---|---|
| 0 | Pure covalent (non-polar) | $H_2$, $Cl_2$ | Simple molecular |
| 0.1 – 0.4 | Non-polar covalent | $C-H$ | Simple molecular |
| 0.5 – 1.7 | Polar covalent | $H-Cl$, $O-H$ | Simple molecular |
| > 1.8 | Ionic | $NaCl$, $MgO$ | Giant ionic lattice |
| Metal + Metal | Metallic | $Cu$, $Fe$ | Giant metallic lattice |
Triangle of bonding: A triangular diagram with ionic, covalent, and metallic bonding at the three vertices. Any compound can be plotted within the triangle based on its bonding character. For example, $NaCl$ is near the ionic vertex, while $HCl$ is between ionic and covalent.
🔗 Polymers
📺 S2.4.4 — Common Properties of Plastics
Definition
Polymers are large molecules (macromolecules) made from many repeating structural units called monomers, joined by covalent bonds.
Addition Polymers
Formed from unsaturated monomers (containing C=C double bonds). The double bond opens and monomers link together. No atoms are lost.
| Monomer | Polymer | Use |
|---|---|---|
| Ethene ($CH_2=CH_2$) | Polyethene (PE) | Plastic bags, bottles |
| Propene ($CH_2=CHCH_3$) | Polypropene (PP) | Rope, packaging |
| Chloroethene ($CH_2=CHCl$) | PVC | Pipes, window frames |
| Tetrafluoroethene ($CF_2=CF_2$) | PTFE (Teflon) | Non-stick pans |
Condensation Polymers
Formed from monomers with two functional groups. Each bond forms with the loss of a small molecule (usually $H_2O$).
| Type | Monomer Types | Bond Formed | Example |
|---|---|---|---|
| Polyester | Diol + Dicarboxylic acid | Ester bond ($-COO-$) | PET (drink bottles) |
| Polyamide | Diamine + Dicarboxylic acid | Amide bond ($-CONH-$) | Nylon (clothing, rope) |
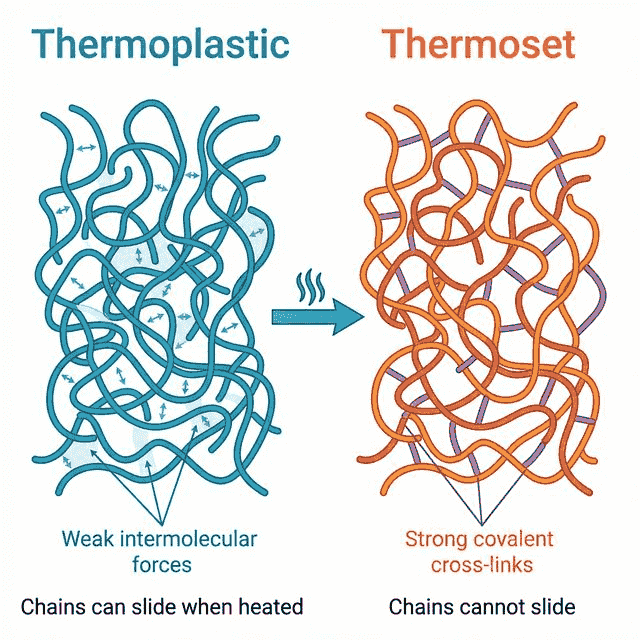
Thermoplastics vs Thermosets
| Feature | Thermoplastics | Thermosets |
|---|---|---|
| Structure | Weak intermolecular forces between chains | Strong covalent cross-links between chains |
| On heating | Soften and can be remoulded | Do not soften; decompose at high temperatures |
| Recyclable? | Yes — can be melted and reshaped | No — cross-links prevent melting |
| Examples | Polyethene, PVC, polystyrene | Bakelite, melamine, epoxy resin |
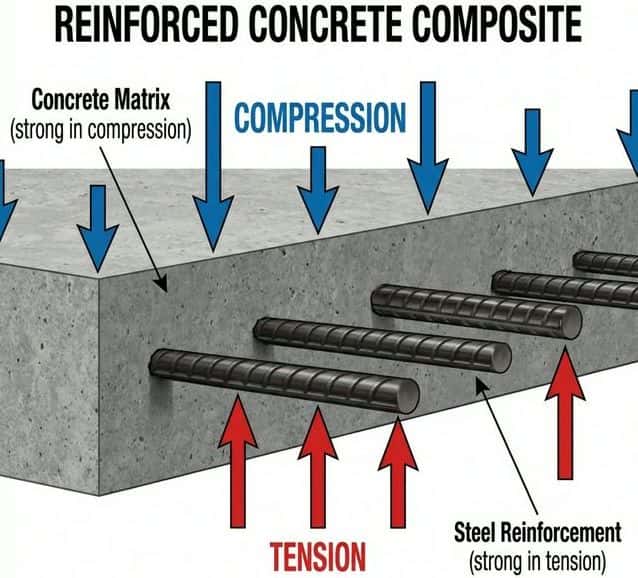
🏗️ Composites
Definition
A composite is a material made from two or more different substances with significantly different properties. Together, they create a material with properties superior to either component alone.
Composites consist of: a Matrix (binder) and Reinforcement (fibers/particles).
| Composite | Matrix | Reinforcement | Key Advantage |
|---|---|---|---|
| Reinforced concrete | Concrete (strong in compression) | Steel rods (strong in tension) | Handles both compression and tension forces |
| Fiberglass | Plastic resin | Glass fibers | Lightweight, strong, waterproof |
| Carbon fiber composite | Epoxy resin | Carbon fibers | Extremely strong, very lightweight |
🔬 Nanotechnology
The Nanoscale (1–100 nm)
At the nanoscale, the surface area to volume ratio becomes extremely high. This dramatically changes the properties of materials compared to their bulk forms.
| Material | Bulk Properties | Nano Properties | Application |
|---|---|---|---|
| Gold | Yellow, inert, unreactive | Red/purple, catalytically active | Medical diagnostics, drug delivery |
| Silver | Conductive, lustrous | Strong antimicrobial properties | Wound dressings, water purification |
| Carbon nanotubes | Graphite: soft, lubricant | 100× stronger than steel, excellent conductor | Electronics, drug delivery, composites |
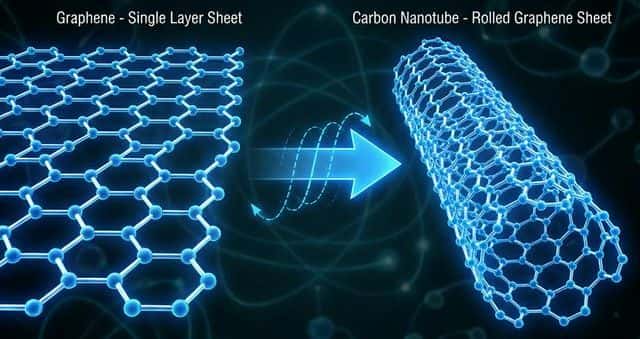
Carbon Allotropes at the Nanoscale
| Allotrope | Structure | Key Property |
|---|---|---|
| Graphene | Single layer of hexagonal carbon rings (2D sheet) | Strongest material known, excellent conductor |
| Carbon nanotube | Graphene sheet rolled into a cylinder | High tensile strength, conducts electricity |
| Fullerene ($C_{60}$) | Spherical cage of 60 carbon atoms (soccer ball) | Hollow — can trap atoms inside for drug delivery |
🧠 Memory Aids
🔤 Addition vs Condensation — "ACE"
- Addition = All atoms kept (nothing lost)
- Condensation = small molecule Comes out (usually water)
- Ester/amide bonds formed in condensation
🔤 Polyester vs Polyamide — "DADA"
- Diol + Acid → Polyester (ester bond $-COO-$)
- Diamine + Acid → Polyamide (amide bond $-CONH-$)
🔤 Nano Rule — "SAV changes everything"
At the nanoscale, the Surface Area to Volume ratio becomes huge → new properties emerge (colour, reactivity, strength).
🔤 Thermosets — "Cross = Can't Come Back"
Thermosets have cross-links (strong covalent bonds between chains) → cannot be melted or reshaped → not recyclable. The "cross" locks them permanently.
🌍 Real-World Applications
🥤 PET Bottles — Condensation Polymer in Daily Life
Context: Most plastic drink bottles are made of PET (polyethylene terephthalate), a condensation polyester.
Science: PET is formed from ethylene glycol (diol) and terephthalic acid (dicarboxylic acid). Each bond forms with the loss of water, creating ester linkages ($-COO-$) throughout the chain. The resulting polymer is lightweight, transparent, and has a low gas permeability.
Impact: PET is one of the most recycled plastics (recycling code #1). It can be melted and reformed because it is a thermoplastic — no cross-links.
🏎️ Carbon Fiber in Formula 1 Cars
Context: F1 car chassis are made from carbon fiber reinforced polymer (CFRP) composite.
Science: Carbon fibers (from graphite) provide extreme tensile strength, while the epoxy resin matrix binds them together. The composite is 5× stronger than steel but only 1/5 the weight.
Impact: The survival cell in F1 cars can withstand 200+ km/h crashes while protecting the driver. This technology has also improved aircraft, bicycles, and medical prosthetics.
🩺 Gold Nanoparticles in Medical Tests
Context: COVID-19 lateral flow tests use gold nanoparticles to produce the visible colored line.
Science: Gold nanoparticles (~40 nm) appear red/purple (unlike bulk gold which is yellow). When antibodies on the test strip bind to the virus, they aggregate the gold nanoparticles, producing a visible red line.
Impact: Fast, cheap, at-home diagnostics made possible by different optical properties at the nanoscale. The same principle is used in pregnancy tests.
⚠️ Common Mistakes
- ❌ Confusing addition and condensation → ✅ Addition: C=C opens, nothing lost. Condensation: two functional groups react, small molecule (usually $H_2O$) lost.
- ❌ "Thermosets can be recycled by melting" → ✅ Thermosets have covalent cross-links. Heating decomposes them — they cannot be melted and reshaped. Only thermoplastics can be recycled by melting.
- ❌ "Nanoparticles have different properties because they are small" → ✅ Must explain the mechanism: at the nanoscale, the very high surface area to volume ratio creates more active sites, changing reactivity and optical properties.
- ❌ Drawing the wrong repeating unit → ✅ For addition polymers: open the C=C double bond → show brackets with $n$. For condensation: show the ester or amide linkage and the loss of $H_2O$.
- ❌ "Composites are alloys" → ✅ Composites have two distinct phases (matrix + reinforcement). Alloys are homogeneous mixtures of metals. Different concepts.
📝 Exam-Style Questions
Question 1: Explain why thermosetting plastics cannot be recycled by melting. [2 marks]
Mark Scheme:
- [1 mark] They contain strong covalent cross-links between polymer chains.
- [1 mark] Heating causes the bonds to break (chemical decomposition) rather than allowing chains to slide (melting).
Question 2: State the difference between addition and condensation polymerization. [2 marks]
Mark Scheme:
- [1 mark] Addition: unsaturated monomers (C=C) join; no atoms lost.
- [1 mark] Condensation: monomers with two functional groups join; small molecule (usually water) is eliminated.
Question 3: Explain why the properties of gold nanoparticles differ from bulk gold. [2 marks]
Mark Scheme:
- [1 mark] At the nanoscale, the surface area to volume ratio is very high.
- [1 mark] This creates more reactive surface atoms → different optical properties (red/purple vs yellow) and higher catalytic activity.
Question 4: Identify the matrix and reinforcement in carbon fiber composite. [2 marks]
Mark Scheme:
- [1 mark] Matrix: Epoxy resin (polymer).
- [1 mark] Reinforcement: Carbon fibers.
Question 5: State the monomers needed to form a polyester and name the bond formed. [2 marks]
Mark Scheme:
- [1 mark] A diol and a dicarboxylic acid.
- [1 mark] Ester bond ($-COO-$) formed with loss of $H_2O$.
Question 6: Suggest one application of carbon nanotubes and explain why they are suitable. [2 marks]
Mark Scheme:
- [1 mark] Any valid application: drug delivery / electronics / reinforcing composites.
- [1 mark] Suitable because: hollow structure (drug delivery) / high conductivity (electronics) / high tensile strength (composites).
Question 7: Contrast the structure of thermoplastics and thermosets. [2 marks]
Mark Scheme:
- [1 mark] Thermoplastics: chains held by weak intermolecular forces → can slide when heated.
- [1 mark] Thermosets: chains held by strong covalent cross-links → rigid, cannot be reshaped.
Question 8: Explain why bonding is described as a continuum rather than three distinct categories. [2 marks]
Mark Scheme:
- [1 mark] Most bonds have partial ionic and partial covalent character.
- [1 mark] The degree of each depends on the electronegativity difference between the atoms — there is no sharp boundary.