📋 IB Content Statements (S3.1)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S3.1.1: The periodic table is arranged into groups and periods. Elements in a group have the same number of valence electrons.
- S3.1.2: Trends in properties across a period and down a group can be explained by the concepts of nuclear charge, electron shielding, and atomic/ionic radius.
- S3.1.3: Trends in metallic/non-metallic character, ionization energy, electron affinity, and electronegativity can be explained by the same concepts.
- S3.1.4: Oxides of Period 3 change character from basic (metal oxides) through amphoteric ($Al_2O_3$) to acidic (non-metal oxides).
HL Extension
- S3.1.5 (HL): First ionization energy data for successive elements across a period provides evidence for sub-shells.
- S3.1.6 (HL): Transition metals show variable oxidation states, form complex ions, and form colored compounds.
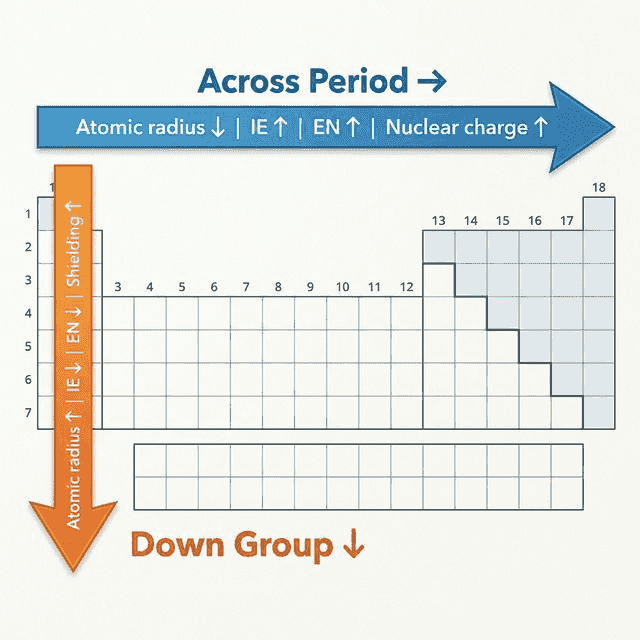
🔹 Understanding Periodic Trends
Three Key Factors (E.N.S.)
All periodic trends can be explained using just three factors:
- Effective Nuclear Charge ($Z_{eff}$): The net positive charge experienced by valence electrons. Increases across a period (more protons, same shielding).
- Number of Electron Shells: Increases down a group → more distance between nucleus and valence electrons.
- Shielding (screening): Inner electrons repel valence electrons, reducing the pull of the nucleus. Shielding increases down a group.
📐 Atomic & Ionic Radius
| Trend | Direction | Explanation |
|---|---|---|
| Across a period | Radius decreases → | $Z_{eff}$ increases (more protons, same shielding), so electrons are pulled closer to the nucleus. |
| Down a group | Radius increases ↓ | More electron shells are added, increasing the distance. Increased shielding also reduces the pull on valence electrons. |
Ionic Radius Rules
- Cations are smaller than their parent atoms (lost electrons → fewer shells or reduced repulsion, remaining electrons pulled in tighter).
- Anions are larger than their parent atoms (gained electrons → more repulsion, electron cloud expands).
- Isoelectronic species (same number of electrons): more protons = smaller radius. E.g., $O^{2-} > F^- > Na^+ > Mg^{2+} > Al^{3+}$ (all have 10 electrons).
⚡ Ionization Energy
Definition
The first ionization energy ($IE_1$) is the minimum energy needed to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous $1+$ ions.
$$X(g) \rightarrow X^+(g) + e^-$$
| Trend | Direction | Explanation |
|---|---|---|
| Across a period | IE increases → | $Z_{eff}$ increases, electrons are more tightly held. Smaller atomic radius means electrons are closer to the nucleus. |
| Down a group | IE decreases ↓ | More shells → more distance and shielding → valence electrons are easier to remove. |
HL: Anomalies in IE across Period 2 & 3
Two "dips" in the otherwise increasing trend:
- Group 2 → Group 13 dip: $Be → B$ (or $Mg → Al$). Group 13 loses a $2p$ electron (higher energy, further from nucleus) vs a $2s$ electron in Group 2 → easier to remove.
- Group 15 → Group 16 dip: $N → O$ (or $P → S$). Group 16 has a paired electron in one $p$-orbital. Electron-electron repulsion in the pair makes it easier to remove.
🧲 Electronegativity
Definition
Electronegativity ($\chi$) is the ability of an atom in a covalent bond to attract the shared pair of electrons towards itself.
Measured on the Pauling scale: Fluorine = 4.0 (most electronegative), Francium is the least.
| Trend | Direction | Explanation |
|---|---|---|
| Across a period | EN increases → | Smaller radius + higher $Z_{eff}$ → stronger pull on shared electrons. |
| Down a group | EN decreases ↓ | Larger radius + more shielding → weaker pull on shared electrons. |
Noble gases have no electronegativity values because they do not normally form covalent bonds. Don't include them in trend discussions.
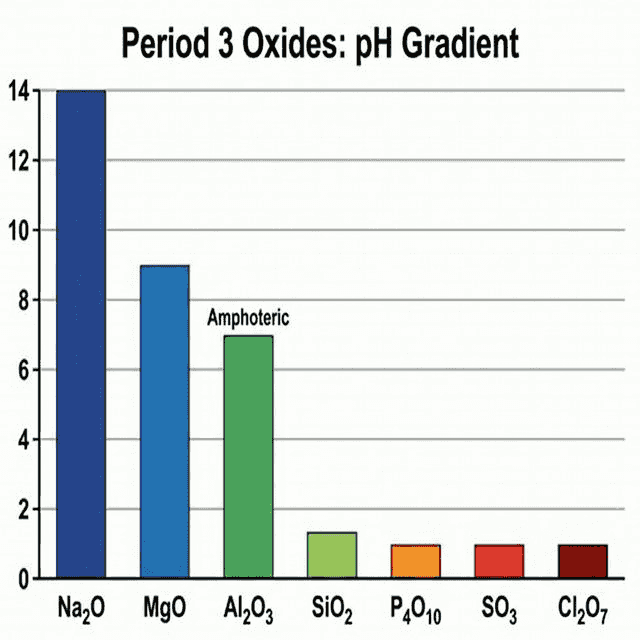
🧪 Period 3 Oxides
The oxides of Period 3 show a clear transition from basic to acidic character:
| Element | Oxide | Bonding | Character | pH of solution |
|---|---|---|---|---|
| $Na$ | $Na_2O$ | Ionic | Strongly basic | ~14 |
| $Mg$ | $MgO$ | Ionic | Basic | ~9 |
| $Al$ | $Al_2O_3$ | Ionic with covalent character | Amphoteric | Insoluble |
| $Si$ | $SiO_2$ | Giant covalent | Weakly acidic | Insoluble |
| $P$ | $P_4O_{10}$ | Simple covalent | Acidic | ~1 |
| $S$ | $SO_3$ | Simple covalent | Strongly acidic | ~1 |
| $Cl$ | $Cl_2O_7$ | Simple covalent | Strongly acidic | ~1 |
Why the change?
Metal oxides contain oxide ions ($O^{2-}$) which act as bases in water: $O^{2-} + H_2O \rightarrow 2OH^-$
Non-metal oxides form acids when dissolved in water: $SO_3 + H_2O \rightarrow H_2SO_4$
$Al_2O_3$ is amphoteric — it can react with both acids AND bases.
🧠 Memory Aids
🔤 "ENS" — Three factors for ALL trends
- Effective nuclear charge ($Z_{eff}$)
- Number of electron shells
- Shielding (by inner electrons)
Every single periodic trend answer uses these three factors.
🔤 Across a Period — "SIR" (Small, Ionize, Reactive non-metals)
- Smaller atomic radius
- Increased IE and EN
- Rising non-metallic character
🔤 Cations vs Anions — "Cats are small, Ans are large"
Cations (positive) are smaller than the parent atom. Anions (negative) are larger. Cat = small domestic animal. An(aconda) = large animal.
🔤 Period 3 Oxides — "BAA" (Basic → Amphoteric → Acidic)
Reading from left to right across Period 3 oxides: Basic metals → Amphoteric (aluminium) → Acidic non-metals. Like a sheep: "BAA" 🐑
🔤 HL IE Anomalies — "2p then Pair"
Two dips: (1) Losing a 2p electron is easier than 2s → Gp 2→13 dip. (2) A paired electron is easier to remove than unpaired → Gp 15→16 dip.
🌍 Real-World Applications
🔥 Alkali Metals in Water — Group 1 Reactivity
Context: Sodium, potassium, and cesium react increasingly vigorously with water, from fizzing (Na) to explosive (Cs).
Science: Down Group 1, IE decreases because the valence electron is further from the nucleus with more shielding. This means the electron is lost more easily → faster, more vigorous reaction.
Impact: This trend is used to predict reactivity of any group in the periodic table. Metals get more reactive going down; non-metals get more reactive going up.
🏭 Acid Rain — Non-Metal Oxides
Context: Burning fossil fuels releases sulfur dioxide ($SO_2$) and nitrogen oxides ($NO_x$), which dissolve in rainwater to form sulfuric and nitric acid.
Science: These are non-metal oxides → acidic when dissolved in water. $SO_2 + H_2O \rightarrow H_2SO_3$ and $2SO_2 + O_2 \rightarrow 2SO_3$, $SO_3 + H_2O \rightarrow H_2SO_4$
Impact: Acid rain damages buildings (especially limestone: $CaCO_3 + H_2SO_4 \rightarrow CaSO_4 + H_2O + CO_2$), kills aquatic life, and destroys forests.
🦷 Fluorine in Toothpaste — Highest Electronegativity
Context: Fluoride ions ($F^-$) in toothpaste replace hydroxide ions in tooth enamel to form fluorapatite.
Science: Fluorine has the highest electronegativity (4.0) due to its small atomic radius and high nuclear charge. The resulting $F^-$ ion forms very strong ionic bonds in the enamel lattice.
Impact: Fluorapatite is more resistant to acid attack than hydroxyapatite, reducing tooth decay. The strong bonding of $F^-$ is a direct consequence of fluorine's electronegativity.
⚠️ Common Mistakes
- ❌ "Nuclear charge increases down a group, so IE increases" → ✅ True that nuclear charge increases, but shielding and distance increase more → IE decreases. Always mention all three factors (ENS).
- ❌ "Atomic radius increases across a period" → ✅ It decreases across a period. More protons with same shielding → electrons pulled closer.
- ❌ Confusing electronegativity with electron affinity → ✅ Electronegativity is the ability to attract shared electrons in a bond. Electron affinity is the energy change when an atom gains an electron.
- ❌ "Al₂O₃ is acidic because aluminium is a metal" → ✅ $Al_2O_3$ is amphoteric — it reacts with both acids and bases. This is a key exam point.
- ❌ Saying noble gases have low electronegativity → ✅ Noble gases have no electronegativity values because they do not form covalent bonds (usually). Do not plot them on EN trend graphs.
📝 Exam-Style Questions
Question 1: Explain the trend in first ionization energy across Period 3. [3 marks]
Mark Scheme:
- [1 mark] General increase across the period.
- [1 mark] Nuclear charge / number of protons increases across the period.
- [1 mark] Shielding remains approximately constant (same number of inner shells) → greater effective nuclear charge → electrons held more tightly.
Question 2: Explain why the atomic radius of sodium is larger than the atomic radius of chlorine. [2 marks]
Mark Scheme:
- [1 mark] Chlorine has more protons (17 vs 11), creating a greater nuclear charge.
- [1 mark] With similar shielding (both have 2 inner shells), the valence electrons in Cl are pulled closer to the nucleus.
Question 3: Explain why $Na_2O$ is basic while $SO_3$ is acidic. [3 marks]
Mark Scheme:
- [1 mark] $Na_2O$ contains $O^{2-}$ ions which react with water: $O^{2-} + H_2O → 2OH^-$ (producing hydroxide ions).
- [1 mark] $SO_3$ reacts with water to form sulfuric acid: $SO_3 + H_2O → H_2SO_4$.
- [1 mark] Metal oxides are basic; non-metal oxides are acidic.
Question 4: Rank the following in order of increasing radius: $Na^+$, $F^-$, $Ne$, $Mg^{2+}$. [2 marks]
Mark Scheme:
- [1 mark] All are isoelectronic (10 electrons).
- [1 mark] Order: $Mg^{2+} < Na^+ < Ne < F^-$. More protons=smaller radius (tighter pull on the same number of electrons).
Question 5: Explain why fluorine has the highest electronegativity. [2 marks]
Mark Scheme:
- [1 mark] Very small atomic radius.
- [1 mark] High nuclear charge relative to its size → bonding electrons are strongly attracted to the nucleus.
Question 6 (HL): Explain the decrease in first ionization energy from nitrogen to oxygen. [2 marks]
Mark Scheme:
- [1 mark] Oxygen has a paired electron in one of its $2p$ orbitals.
- [1 mark] Electron-electron repulsion in the pair makes this electron easier to remove than an unpaired electron in nitrogen's half-filled $2p$ subshell.
Question 7: State what is meant by "amphoteric" and give one example. [2 marks]
Mark Scheme:
- [1 mark] Amphoteric means a substance can react with both acids and bases.
- [1 mark] Example: $Al_2O_3$ (aluminium oxide).
Question 8: Explain why a chloride ion ($Cl^-$) is larger than a chlorine atom ($Cl$). [2 marks]
Mark Scheme:
- [1 mark] $Cl^-$ has gained one electron → 18 electrons but only 17 protons.
- [1 mark] Greater electron-electron repulsion → electron cloud expands → larger radius.