📋 IB Content Statements (S3.2)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S3.2.1: A homologous series is a family of compounds with the same general formula and similar chemical properties. Each successive member differs by $CH_2$.
- S3.2.2: Structural formulas can be represented as full, condensed, or skeletal formulas.
- S3.2.3: Functional groups give organic molecules their characteristic chemical properties.
- S3.2.4: IUPAC naming follows a systematic set of rules based on the longest carbon chain, substituents, and functional groups.
- S3.2.5: Structural isomers have the same molecular formula but different structural arrangements.
HL Extension
- S3.2.6 (HL): Stereoisomers have the same structural formula but different arrangements in space (cis-trans and optical isomers).
🔹 Homologous Series
Definition
A homologous series is a family of organic compounds that:
- Have the same general formula
- Differ by $CH_2$ between successive members
- Show a gradual change in physical properties (e.g., boiling point increases with chain length)
- Have similar chemical properties (same functional group)
| Homologous Series | General Formula | Functional Group | Example (3C) | Suffix/Prefix |
|---|---|---|---|---|
| Alkanes | $C_nH_{2n+2}$ | None (C–C, C–H only) | Propane ($C_3H_8$) | -ane |
| Alkenes | $C_nH_{2n}$ | C=C double bond | Propene ($C_3H_6$) | -ene |
| Alcohols | $C_nH_{2n+1}OH$ | $-OH$ (hydroxyl) | Propan-1-ol ($C_3H_7OH$) | -ol |
| Carboxylic acids | $C_nH_{2n+1}COOH$ | $-COOH$ (carboxyl) | Propanoic acid ($C_2H_5COOH$) | -oic acid |
| Aldehydes | $C_nH_{2n}O$ | $-CHO$ | Propanal ($C_2H_5CHO$) | -al |
| Ketones | $C_nH_{2n}O$ | $-CO-$ | Propanone ($CH_3COCH_3$) | -one |
| Esters | — | $-COO-$ | Methyl ethanoate | -oate |
| Amines | $C_nH_{2n+3}N$ | $-NH_2$ | Propylamine ($C_3H_7NH_2$) | -amine |
| Halogenoalkanes | — | $-X$ (F, Cl, Br, I) | 1-chloropropane ($C_3H_7Cl$) | halo- prefix |
📋 IUPAC Nomenclature Rules
Step-by-Step Naming
- Find the longest continuous carbon chain → root name (meth-, eth-, prop-, but-, pent-, hex-...)
- Identify the principal functional group → suffix (-ol, -al, -one, -oic acid, -amine)
- Number the chain so the functional group gets the lowest possible number
- Name substituents as prefixes (methyl-, ethyl-, chloro-) with position numbers
- Alphabetize substituent prefixes (ignore di-, tri-)
| Carbons | Root | Carbons | Root |
|---|---|---|---|
| 1 | meth- | 6 | hex- |
| 2 | eth- | 7 | hept- |
| 3 | prop- | 8 | oct- |
| 4 | but- | 9 | non- |
| 5 | pent- | 10 | dec- |
🧪 Key Functional Groups & Reactions
The functional group determines the chemical behaviour of the molecule. The carbon chain provides the "skeleton" but the functional group gives the molecule its reactivity.
Priority Order for Naming (highest → lowest)
Carboxylic acid > Ester > Amide > Aldehyde > Ketone > Alcohol > Amine > Alkene > Alkane
Degrees of Unsaturation (IHD)
Index of Hydrogen Deficiency
$$IHD = \frac{2C + 2 + N - H - X}{2}$$
Where C = carbons, N = nitrogens, H = hydrogens, X = halogens. Each IHD represents one ring or one double bond. Two IHD could mean one triple bond.
🔀 Isomerism
Types of Isomerism
Isomers are molecules with the same molecular formula but different arrangements of atoms.
| Type | Definition | Example ($C_4H_{10}$) |
|---|---|---|
| Chain isomers | Different arrangement of the carbon skeleton | Butane vs 2-methylpropane |
| Position isomers | Same functional group at different positions | Butan-1-ol vs Butan-2-ol |
| Functional group isomers | Different functional groups (same formula) | Propanal ($C_3H_6O$) vs Propanone ($C_3H_6O$) |
HL: Stereoisomers
Same structural formula, different spatial arrangement.
| Type | Requirement | Example |
|---|---|---|
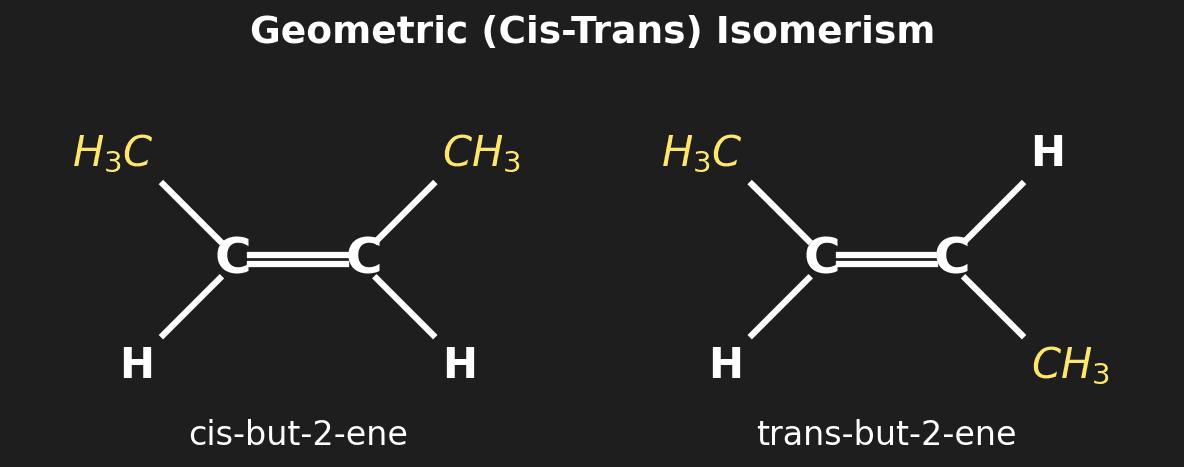
| Cis-trans (geometric) | Restricted rotation (C=C or ring) + two different groups on each carbon | cis-but-2-ene vs trans-but-2-ene |
| Optical (enantiomers) | Chiral centre (carbon with 4 different groups attached) | D-alanine vs L-alanine |
📊 Physical Properties & Trends
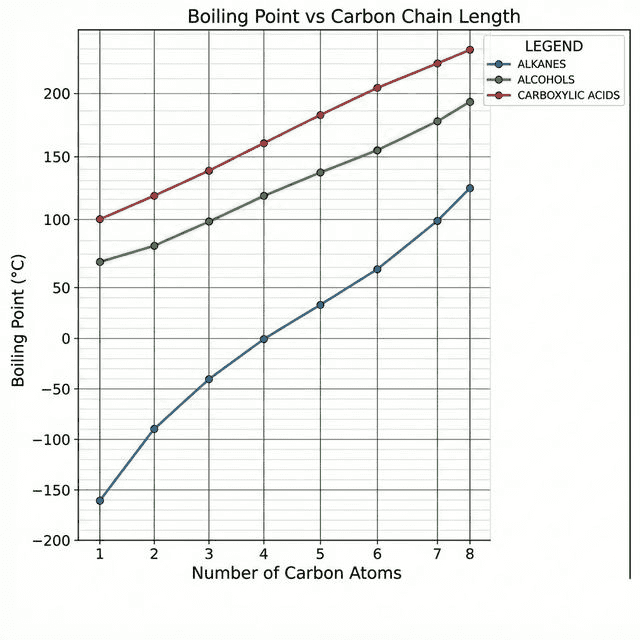
Boiling Point Trends
| Factor | Effect on BP | Explanation |
|---|---|---|
| Longer chain | Higher BP | More electrons → stronger London dispersion forces (more surface contact) |
| Branching | Lower BP | More compact shape → less surface contact → weaker LDFs |
| Hydrogen bonding | Much higher BP | $-OH$ and $-COOH$ groups form H-bonds → much more energy to overcome |
| Polarity | Higher BP | Polar molecules (dipole-dipole) have stronger IMFs than non-polar |
Solubility Trends
- Short-chain alcohols and carboxylic acids → soluble in water (H-bonding with water dominates).
- Longer chains → less soluble (non-polar hydrocarbon chain dominates over the polar functional group).
- Alkanes → insoluble in water (non-polar, cannot H-bond).
🧠 Memory Aids
🔤 Carbon Roots — "Monkeys Eat Peanut Butter"
- Meth = 1C
- Eth = 2C
- Prop = 3C
- But = 4C
Then: Pent (5), Hex (6), Hept (7), Oct (8), Non (9), Dec (10).
🔤 IUPAC Naming — "Find, Identify, Number, Name, Alphabetize" (FINNA)
- Find the longest chain
- Identify the principal functional group (suffix)
- Number from the end closest to the functional group
- Name substituents (prefixes)
- Alphabetize prefixes
🔤 Three Types of Structural Isomers — "ChaPoFu"
- Chain isomers (different carbon skeleton)
- Position isomers (same group, different position)
- Functional group isomers (different functional group entirely)
🔤 BP Trend — "LDF + HB = Higher BP"
Longer chains = more London Dispersion Forces. $-OH$ or $-COOH$ = Hydrogen Bonding. Both increase BP. Branching = lower surface contact = lower LDFs = lower BP.
🌍 Real-World Applications
⛽ Petroleum Fractions — Homologous Series in Action
Context: Crude oil is separated by fractional distillation into fractions of different chain lengths.
Science: Shorter alkanes (1–4C: natural gas) have lower boiling points due to weaker London forces. Longer chains (diesel, lubricants, bitumen) have progressively higher boiling points. This is the homologous series trend in action.
Impact: Each fraction has different uses — from LPG cooking gas to petrol, kerosene (jet fuel), diesel, and bitumen (road surfaces).
💊 Thalidomide — Optical Isomerism Matters
Context: Thalidomide was prescribed in the 1960s for morning sickness. One optical isomer was a safe sedative; the other caused severe birth defects.
Science: The two enantiomers (mirror images) of thalidomide interact differently with biological receptors. Even though they have identical molecular and structural formulas, their 3D shape matters for biological activity.
Impact: This tragedy led to much stricter drug testing regulations and highlighted the importance of chirality in pharmaceutical chemistry. Today, many drugs are sold as single enantiomers.
🧴 Ethanol — Alcohol in Daily Life
Context: Ethanol ($C_2H_5OH$) is used as a fuel additive (E10 petrol), in alcoholic beverages, and as a solvent in hand sanitizers.
Science: The $-OH$ group makes ethanol polar and able to hydrogen-bond with water → completely miscible. It also has a lower boiling point (78°C) than water (100°C) because the non-polar ethyl group reduces overall IMF strength compared to pure water.
Impact: Ethanol's dual nature (polar OH + non-polar chain) makes it an excellent solvent that dissolves both polar and non-polar substances.
⚠️ Common Mistakes
- ❌ "Isomers have different molecular formulas" → ✅ Isomers have the same molecular formula but different arrangements of atoms. If the formula is different, they are different compounds, not isomers.
- ❌ Not finding the longest continuous chain → ✅ The longest chain may not be drawn horizontally — it might zigzag. Always trace every possible path to find the longest chain.
- ❌ "Alkanes are unsaturated" → ✅ Alkanes are saturated (all single bonds, maximum H atoms). Alkenes are unsaturated (contain C=C double bond).
- ❌ Using wrong suffix/prefix → ✅ For alcohols use -ol, for aldehydes use -al, for ketones use -one. Don't confuse them. If $-OH$ is on the first carbon with $-CHO$, it's an aldehyde (suffix -al), not an alcohol.
- ❌ Numbering from the wrong end → ✅ Always number so the principal functional group gets the lowest number. If there's no functional group, the first branching point gets the lowest number.
📝 Exam-Style Questions
Question 1: Define a homologous series and state two features. [2 marks]
Mark Scheme:
- [1 mark] A family of compounds with the same general formula that differ by $CH_2$ between successive members.
- [1 mark] Any one: same functional group / similar chemical properties / gradual change in physical properties.
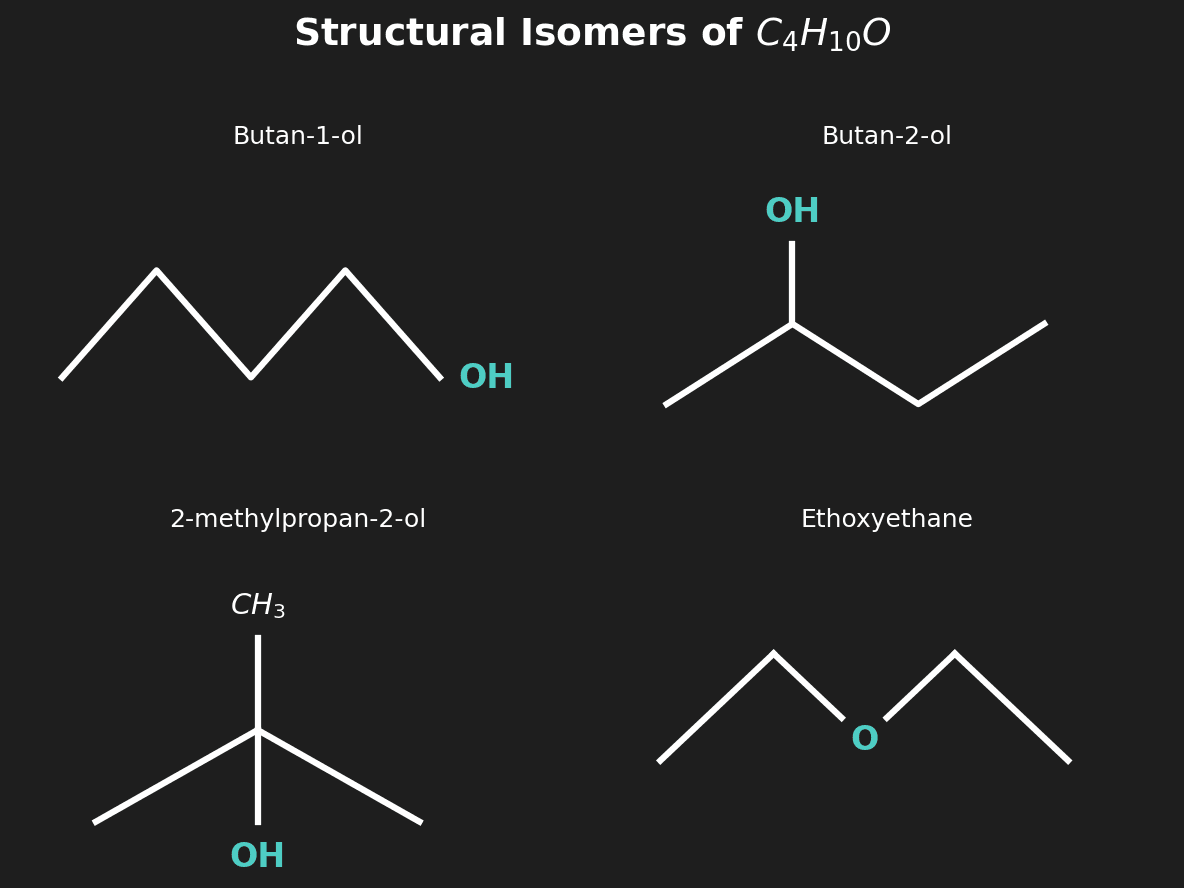
Question 2: Draw structural isomers of $C_4H_{10}O$ that are alcohols. [2 marks]
Mark Scheme:
- [1 mark] Butan-1-ol ($CH_3CH_2CH_2CH_2OH$).
- [1 mark] Butan-2-ol ($CH_3CH(OH)CH_2CH_3$) or 2-methylpropan-1-ol or 2-methylpropan-2-ol.
Question 3: Name the compound: $CH_3CH(CH_3)CH_2CH_2OH$. [1 mark]
Mark Scheme:
- [1 mark] 3-methylbutan-1-ol.
Question 4: Explain why butan-1-ol has a higher boiling point than butane. [2 marks]
Mark Scheme:
- [1 mark] Butan-1-ol can form hydrogen bonds (due to the $-OH$ group).
- [1 mark] Butane can only form London dispersion forces → hydrogen bonds are stronger → more energy needed to overcome them.
Question 5: Explain why pentane has a higher boiling point than 2,2-dimethylpropane (same molecular formula $C_5H_{12}$). [2 marks]
Mark Scheme:
- [1 mark] Pentane is a longer, straighter chain → greater surface area for contact between molecules.
- [1 mark] 2,2-dimethylpropane is more spherical/compact → less surface contact → weaker London dispersion forces.
Question 6 (HL): State the conditions needed for geometric (cis-trans) isomerism. [2 marks]
Mark Scheme:
- [1 mark] Restricted rotation (usually a C=C double bond or ring).
- [1 mark] Two different groups/atoms attached to each carbon of the double bond.
Question 7: Explain why methanol is miscible with water but hexan-1-ol has limited solubility. [2 marks]
Mark Scheme:
- [1 mark] Methanol: small hydrocarbon chain → the $-OH$ group dominates → extensive hydrogen bonding with water.
- [1 mark] Hexan-1-ol: long non-polar carbon chain dominates → not enough H-bonding to overcome the hydrophobic chain.
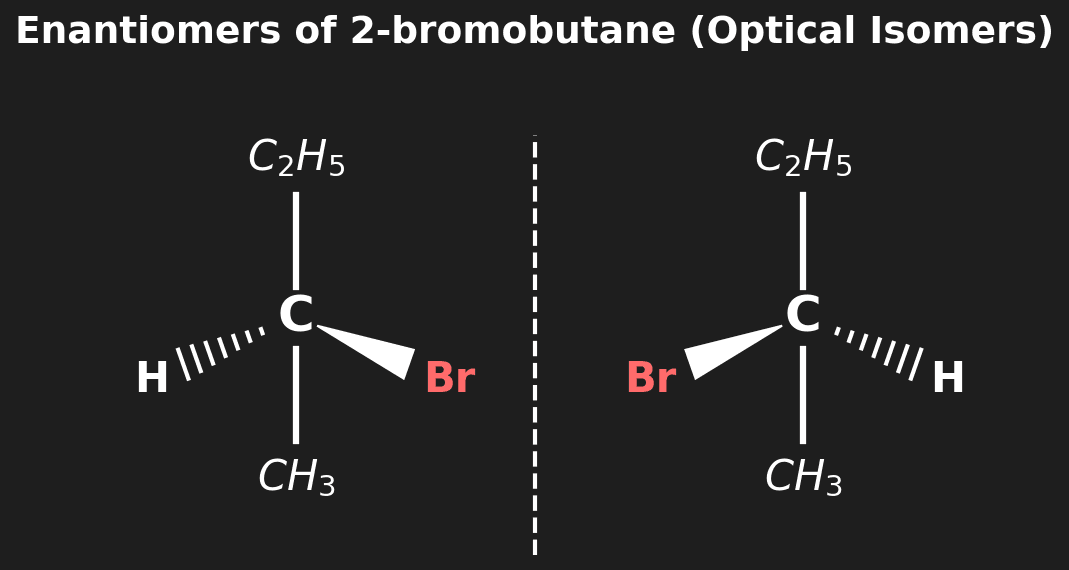
Question 8 (HL): Identify the chiral centre in 2-bromobutane and explain why it shows optical isomerism. [2 marks]
Mark Scheme:
- [1 mark] C-2 is the chiral centre (it has four different groups: $-H$, $-Br$, $-CH_3$, $-CH_2CH_3$).
- [1 mark] This produces two non-superimposable mirror images (enantiomers) that rotate plane-polarized light in opposite directions.