📋 IB Content Statements (S2.2)
This topic covers the following syllabus points from the IB Chemistry 2025 guide:
- S2.2.1: A covalent bond is formed by the electrostatic attraction between a shared pair of electrons and the positively charged nuclei.
- S2.2.2: Single, double, and triple covalent bonds involve one, two, and three shared pairs of electrons respectively.
- S2.2.3: A coordinate (dative) bond is a covalent bond in which both the electrons in the shared pair come from the same atom.
- S2.2.4: The valence shell electron pair repulsion (VSEPR) model enables the shapes of molecules to be predicted from the repulsion of electron domains around a central atom.
- S2.2.5: Bond polarity results from differences in electronegativity between bonded atoms.
- S2.2.6: Molecular polarity depends on both the polarity of the bonds and the shape of the molecule.
- S2.2.7: Resonance structures contribute to the stability of some molecules and polyatomic ions.
HL Extensions
- S2.2.8: Sigma ($\sigma$) bonds are formed by the head-on overlap of atomic orbitals. Pi ($\pi$) bonds are formed by the sideways overlap of parallel p orbitals.
- S2.2.9: Hybridization of atomic orbitals ($sp^3$, $sp^2$, $sp$) explains molecular geometries.
- S2.2.10: Expanded octets occur in period 3+ elements using d-orbitals.
🔹 Covalent Bonding
Key Definition
A covalent bond is the electrostatic attraction between a shared pair of electrons and the positively charged nuclei of the bonded atoms. Covalent bonds typically form between non-metal atoms.
Types of Covalent Bonds
| Bond Type | Shared Pairs | Bond Order | Strength | Length | Example |
|---|---|---|---|---|---|
| Single | 1 | 1 | Weakest | Longest | C–C in ethane, H–H |
| Double | 2 | 2 | Stronger | Shorter | O=O, C=C in ethene |
| Triple | 3 | 3 | Strongest | Shortest | N≡N, C≡C in ethyne |
Key Trend: As bond order increases → bond strength increases → bond length decreases. A triple bond is shorter and stronger than a double bond, which is shorter and stronger than a single bond.
Coordinate (Dative) Bonding
A coordinate bond (dative bond) is a covalent bond where both electrons come from the same atom. Once formed, it is indistinguishable from a regular covalent bond. Represented by an arrow (→) pointing from donor to acceptor.
Examples:
- $NH_4^+$: The lone pair on nitrogen donates to $H^+$ → coordinate bond
- $CO$: Carbon monoxide contains a triple bond: one σ, one π, and one coordinate bond from the oxygen lone pair
- $H_3O^+$: Water donates a lone pair to $H^+$
- $Al_2Cl_6$: Chlorine donates a lone pair to aluminum (which has an empty orbital)
⚡ Bond Polarity and Molecular Polarity
Electronegativity and Bond Polarity
Electronegativity ($\chi$) is the ability of an atom to attract the shared pair of electrons in a covalent bond. The difference in electronegativity ($\Delta\chi$) determines bond polarity:
| $\Delta\chi$ Range | Bond Type | Example |
|---|---|---|
| 0.0 – 0.4 | Non-polar covalent | $H_2$, $Cl_2$, $C-H$ |
| 0.5 – 1.7 | Polar covalent | $H-Cl$, $O-H$, $N-H$ |
| > 1.8 | Ionic | $NaCl$, $MgO$ |
Bond Polarity → Molecular Polarity
A molecule is polar (has a dipole moment) if:
- It contains polar bonds, AND
- The bond dipoles do NOT cancel due to molecular shape
| Molecule | Shape | Polar Bonds? | Dipoles Cancel? | Overall Polar? |
|---|---|---|---|---|
| $CO_2$ | Linear | Yes (C=O) | Yes (symmetrical) | Non-polar |
| $H_2O$ | Bent | Yes (O–H) | No (asymmetrical) | Polar |
| $CCl_4$ | Tetrahedral | Yes (C–Cl) | Yes (symmetrical) | Non-polar |
| $CHCl_3$ | Tetrahedral | Yes (C–Cl, C–H) | No (asymmetrical) | Polar |
| $NH_3$ | Trigonal pyramidal | Yes (N–H) | No (lone pair) | Polar |
| $BF_3$ | Trigonal planar | Yes (B–F) | Yes (symmetrical) | Non-polar |
Quick Rule: If all positions around the central atom have identical atoms/groups AND there are no lone pairs, dipoles cancel → non-polar. If there are lone pairs or different groups, the molecule is likely polar.
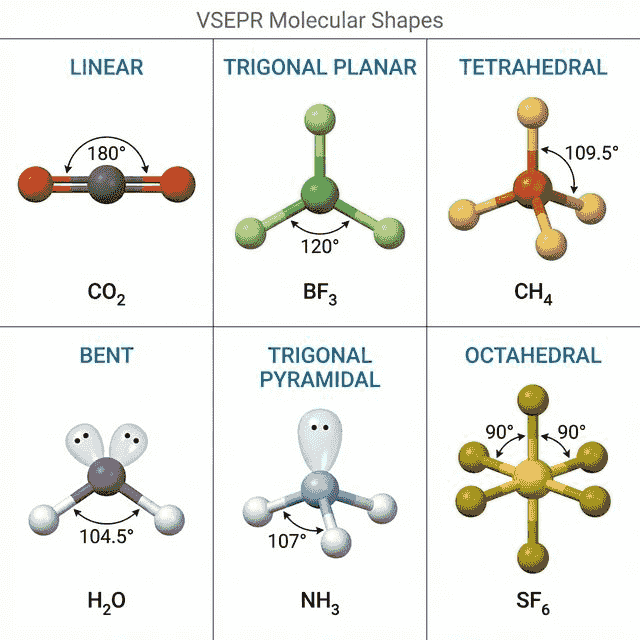
🔷 VSEPR Theory (Molecular Shapes)
Valence Shell Electron Pair Repulsion
Electron domains (bonding pairs and lone pairs) repel each other and orient themselves as far apart as possible to minimize repulsion. The molecular shape is determined by the arrangement of atoms (not electron domains).
Order of Repulsion: Lone Pair–Lone Pair > Lone Pair–Bonding Pair > Bonding Pair–Bonding Pair
The Shapes You Must Know
| Electron Domains | Lone Pairs | Shape | Bond Angle | Example |
|---|---|---|---|---|
| 2 | 0 | Linear | 180° | $CO_2$, $BeCl_2$ |
| 3 | 0 | Trigonal Planar | 120° | $BF_3$, $NO_3^-$ |
| 3 | 1 | Bent (V-shape) | ~117° | $SO_2$, $O_3$ |
| 4 | 0 | Tetrahedral | 109.5° | $CH_4$, $CCl_4$ |
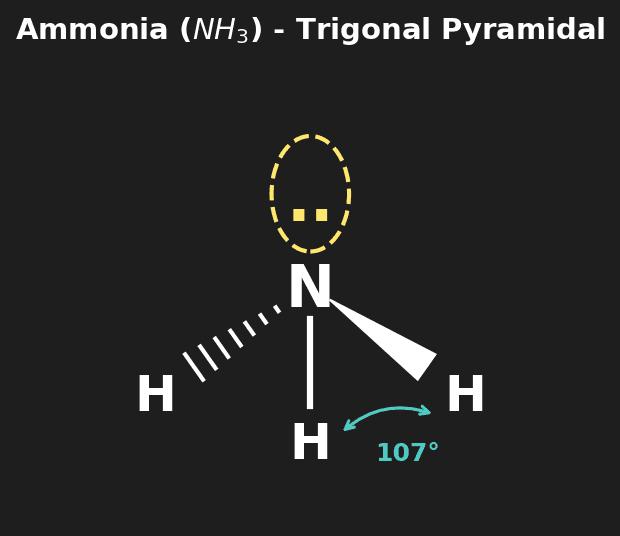
| 4 | 1 | Trigonal Pyramidal | ~107° | $NH_3$, $PCl_3$ |
| 4 | 2 | Bent (V-shape) | ~104.5° | $H_2O$, $H_2S$ |
Why do lone pairs reduce bond angle? Lone pairs occupy more space than bonding pairs (they are held closer to the nucleus and spread out more). Each lone pair squeezes the bonding pairs closer together, reducing the bond angle by approximately 2.5° each.
Interactive 3D Shapes Simulation
AvailableExplore VSEPR molecular geometries in 3D — rotate, zoom, and compare shapes.
→ Launch Simulation🔗 Sigma (σ) and Pi (π) Bonds
Orbital Overlap
Covalent bonds form when atomic orbitals overlap. The type of overlap determines whether the bond is sigma or pi.
| Feature | Sigma (σ) Bond | Pi (π) Bond |
|---|---|---|
| Overlap type | Head-on (end-to-end) | Sideways (parallel p orbitals) |
| Electron density | Along the bond axis | Above and below the bond axis |
| Rotation | Free rotation possible | No rotation (restricts geometry) |
| Strength | Stronger (greater overlap) | Weaker (less overlap) |
| In single bond | 1σ | 0π |
| In double bond | 1σ | 1π |
| In triple bond | 1σ | 2π |
Hybridization
| Hybridization | Orbitals Mixed | Geometry | Bond Angle | Unhybridized p-orbitals | Example |
|---|---|---|---|---|---|
| $sp^3$ | 1s + 3p | Tetrahedral | 109.5° | 0 | $CH_4$, $H_2O$, $NH_3$ |
| $sp^2$ | 1s + 2p | Trigonal Planar | 120° | 1 (forms π bond) | $C_2H_4$, $BF_3$ |
| $sp$ | 1s + 1p | Linear | 180° | 2 (form 2π bonds) | $C_2H_2$, $CO_2$ |
🔁 Resonance Structures
What is Resonance?
Resonance occurs when more than one valid Lewis structure can be drawn for a molecule or ion. The actual structure is a resonance hybrid — an average of all contributing structures. Delocalized electrons are shared across multiple atoms rather than being localized between two atoms.
Key Examples
| Species | Number of Resonance Structures | Effect on Bond Length/Order |
|---|---|---|
| Ozone ($O_3$) | 2 structures | All O–O bonds equal: bond order 1.5 |
| Carbonate ($CO_3^{2-}$) | 3 structures | All C–O bonds equal: bond order 1.33 |
| Benzene ($C_6H_6$) | 2 Kekulé structures | All C–C bonds equal: bond order 1.5 (between single and double) |
| Nitrate ($NO_3^-$) | 3 structures | All N–O bonds equal: bond order 1.33 |
Why resonance stabilizes molecules: Delocalization of electrons lowers the overall energy of the molecule. Resonance hybrids are more stable than any single contributing structure. This is why benzene is unusually stable and does not undergo addition reactions like typical alkenes.
Interactive Resonance Simulation
AvailableVisualize how electron delocalization creates resonance hybrids.
→ Launch Simulation⚛️ Expanded Octet & Hybridization (HL)
5 & 6 Electron Domains
Elements in Period 3 and below can accommodate more than 8 electrons because they have empty d-orbitals available for bonding. This allows 5 or 6 electron domains.
| Domains | Lone Pairs | Shape | Bond Angle | Example |
|---|---|---|---|---|
| 5 | 0 | Trigonal Bipyramidal | 90° and 120° | $PCl_5$ |
| 5 | 1 | Seesaw | ~87° and ~117° | $SF_4$ |
| 5 | 2 | T-shape | ~87° | $ClF_3$ |
| 5 | 3 | Linear | 180° | $XeF_2$ |
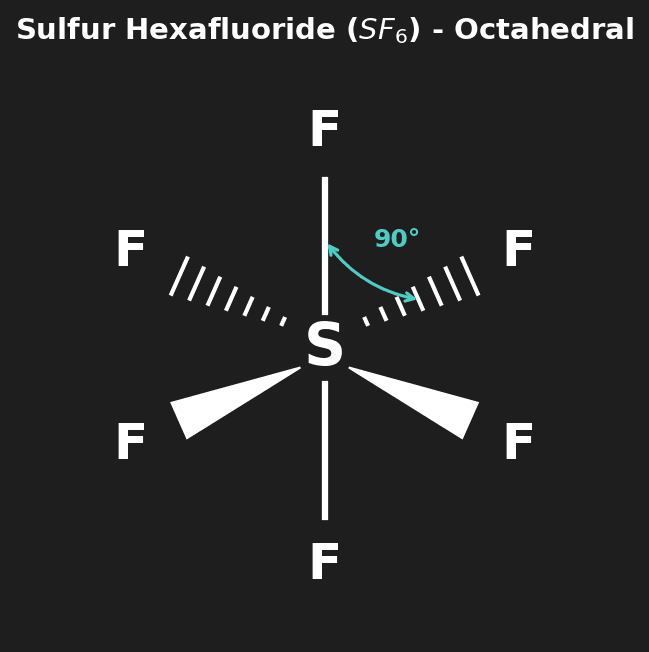
| 6 | 0 | Octahedral | 90° | $SF_6$ |
| 6 | 1 | Square Pyramidal | ~85° | $BrF_5$ |
| 6 | 2 | Square Planar | 90° | $XeF_4$ |
Interactive Expanded Octet Simulation
AvailableExplore expanded octet geometries in 3D.
→ Launch Simulation🧠 Memory Aids & Mnemonics
🔤 VSEPR Shape Order (2-4 domains, no lone pairs)
"Lone Teens Tet" → Linear, Trigonal planar, Tetrahedral
180° → 120° → 109.5°. As domains increase, bond angle decreases.
🔤 Lone Pair Effect on Bond Angle
"Lone pairs are space hogs" → Each lone pair reduces the bond angle by ~2.5°
Tetrahedral (109.5°) → Trigonal Pyramidal (~107°) → Bent (~104.5°). One lone pair = −2.5°, two lone pairs = −5°.
🔤 Sigma and Pi Bond Counting
"Sigma First, then Pi fills the rest"
The first bond between any two atoms is ALWAYS sigma. Any additional bonds (in double or triple bonds) are pi. Single = 1σ. Double = 1σ + 1π. Triple = 1σ + 2π.
🔤 Molecular Polarity Quick Test
"Symmetry Kills Polarity"
If a molecule is perfectly symmetrical with identical atoms around the central atom and NO lone pairs → non-polar (dipoles cancel). Asymmetry (lone pairs or different groups) → polar.
🔤 Hybridization Quick Determination
Number of electron domains = number in $sp^x$
2 domains → $sp$. 3 domains → $sp^2$. 4 domains → $sp^3$. Count ALL electron domains (bonding + lone pairs).
🌍 Real-World Applications
💊 Drug Design and Molecular Shape
Context: Why does a drug work on one receptor but not another? The answer lies in molecular shape — the foundation of pharmacology.
Science: Drugs function through a "lock and key" mechanism: the drug molecule must fit precisely into the active site of an enzyme or receptor. This fit depends on VSEPR geometry, bond angles, and polarity. For example, ibuprofen has a tetrahedral carbon center whose 3D shape allows it to block the active site of the COX enzyme (which produces pain-causing prostaglandins). The (S)-enantiomer is active, while the (R)-enantiomer is inactive — same atoms, different 3D shape.
Impact: Understanding molecular geometry is essential for rational drug design. Computational chemistry now uses VSEPR predictions and molecular modeling to design drugs virtually before synthesis, saving years of research.
🌐 Ozone Depletion and Bond Polarity
Context: The ozone layer protects Earth from harmful UV radiation. Its destruction by CFCs (chlorofluorocarbons) is directly related to bond polarity and molecular structure.
Science: CFCs like $CFCl_3$ contain polar C–Cl bonds. UV light provides enough energy to break the weaker C–Cl bond (homolytic fission), releasing a chlorine radical ($Cl\cdot$). This radical attacks ozone ($O_3$): $Cl\cdot + O_3 \rightarrow ClO\cdot + O_2$. The $ClO\cdot$ then reacts with atomic oxygen: $ClO\cdot + O \rightarrow Cl\cdot + O_2$. The chlorine radical is regenerated and can destroy ~100,000 ozone molecules before being removed.
Impact: The Montreal Protocol (1987) banned CFCs, and the ozone hole is slowly recovering. Understanding bond polarity and bond energy was key to identifying why CFCs were so destructive and designing safer alternatives (HFCs, HFOs).
🏗️ Carbon Allotropes — Same Element, Different Bonding
Context: Diamond and graphite are both pure carbon, yet one is the hardest natural material and the other is one of the softest. The difference is entirely in bonding and structure.
Science: In diamond, each carbon is $sp^3$ hybridized with 4 sigma bonds in a tetrahedral arrangement — creating a rigid 3D network. In graphite, each carbon is $sp^2$ hybridized with 3 sigma bonds in trigonal planar layers. The unhybridized p orbital forms delocalized π bonds across the layer, allowing electrons to move freely (electrical conductivity). Layers are held together by weak London dispersion forces, allowing them to slide (why graphite is a lubricant and pencil material).
Impact: Graphene (a single layer of graphite) is 200× stronger than steel, conducts electricity better than copper, and is nearly transparent. It represents the future of electronics, composite materials, and water filtration.
⚠️ Common Mistakes & Exam Pitfalls
🚫 Mistakes Students Make in This Topic
- Drawing lone pairs on the wrong atom in Lewis structures: Always check that each atom has a full octet (or duet for H). The most common error is forgetting lone pairs on terminal atoms (e.g., O in $H_2O$ has 2 lone pairs).
- Confusing electron domain geometry with molecular geometry: Electron domain geometry counts ALL domains (bonding + lone pairs). Molecular geometry describes only the atom positions. $NH_3$ has tetrahedral electron geometry but trigonal pyramidal molecular geometry.
- Saying "$CO_2$ is polar because it has polar bonds": $CO_2$ has polar C=O bonds but is linear and symmetrical, so dipoles cancel → overall non-polar. Shape matters!
- Forgetting that coordinate bonds look the same as regular covalent bonds once formed: They have the same bond length and strength. The distinction is only in how the bond was formed.
- Wrong bond angles from incorrect lone pair counting: Count ALL electron domains, including lone pairs. Students often forget to count lone pairs when predicting shapes, leading to wrong bond angles.
- Confusing bond order in resonance structures: In benzene, the C–C bond order is 1.5, NOT alternating 1 and 2. The resonant hybrid is an average, and all bonds are identical.
📝 IB-Style Exam Questions
Question 1: Predict the shape and bond angle of the Ammonia molecule ($NH_3$). [3 marks]
Mark Scheme:
- [1 mark] 4 electron domains (3 bonding pairs, 1 lone pair).
- [1 mark] Trigonal Pyramidal molecular geometry.
- [1 mark] Bond angle approx 107° (less than 109.5° due to lone pair repulsion).
Question 2: Explain why the $C-O$ bond length in Carbon Monoxide is shorter than in Carbon Dioxide. [2 marks]
Mark Scheme:
- [1 mark] CO has a triple bond (bond order 3).
- [1 mark] $CO_2$ has double bonds (bond order 2). Triple bonds are shorter and stronger.
Question 3: Deduce the Lewis structure and molecular geometry of $SF_6$. [2 marks]
Mark Scheme:
- [1 mark] Sulphur in center bonded to 6 Fluorines (expanded octet).
- [1 mark] Octahedral geometry (90° bond angles).
Question 4: Compare the bond strength of the carbon-to-carbon bonds in Ethane ($C_2H_6$), Ethene ($C_2H_4$) and Benzene ($C_6H_6$). [3 marks]
Mark Scheme:
- [1 mark] Ethane (single) < Benzene (delocalized 1.5) < Ethene (double).
- [1 mark] Strength increases with bond order.
- [1 mark] Benzene has intermediate bond order between single and double due to resonance.
Question 5: State the hybridization of the carbon atoms in Ethene ($C_2H_4$). [1 mark]
Mark Scheme:
- [1 mark] $sp^2$ hybridized (trigonal planar geometry at each carbon).
Question 6: Explain why $CCl_4$ is non-polar despite containing polar bonds. [2 marks]
Mark Scheme:
- [1 mark] $CCl_4$ has a tetrahedral shape with 4 identical C–Cl bonds / no lone pairs on the central atom.
- [1 mark] The bond dipoles are arranged symmetrically and cancel each other out, giving a net dipole moment of zero.
Question 7: Explain why the bond angle in $H_2O$ (104.5°) is less than in $NH_3$ (107°). [2 marks]
Mark Scheme:
- [1 mark] $H_2O$ has 2 lone pairs and 2 bonding pairs; $NH_3$ has 1 lone pair and 3 bonding pairs.
- [1 mark] The extra lone pair in $H_2O$ exerts greater repulsion on the bonding pairs, compressing the bond angle further than in $NH_3$.
Question 8: Describe the bonding in a molecule of $N_2$ in terms of sigma and pi bonds. [2 marks]
Mark Scheme:
- [1 mark] $N_2$ contains a triple bond consisting of 1 sigma (σ) bond and 2 pi (π) bonds.
- [1 mark] The sigma bond is formed by head-on overlap of sp-hybridized orbitals. The pi bonds are formed by sideways overlap of unhybridized p-orbitals.